Which Of The Following Has The Lowest Ionization Energy
Ever find yourself staring at a bunch of options, trying to figure out which one is the least of something? Like, which pizza topping is the least likely to get you weird looks at a potluck? Or which streaming service has the lowest chance of making you fall asleep during the first five minutes? Well, buckle up, because we're about to dive into a similar kind of decision-making, but for atoms! Today's topic is all about figuring out which of the following has the lowest ionization energy. Sounds fancy, right? But trust me, it's more like picking the easiest chore on your to-do list.
So, what in the world is "ionization energy"? Think of it like this: atoms are these tiny little balls of fluff, right? And in the middle of that fluff, there's a nucleus – the grumpy old man in charge. Then, buzzing around the nucleus are these little electrons, like hyperactive toddlers. Ionization energy is basically the amount of energy it takes to convince one of those electrons to pack its bags and leave the family home. It's like bribing a teenager to finally do their laundry – sometimes it takes a lot of persuasion (energy), and sometimes they’re just itching to escape.
We're looking for the atom where it's easiest to snatch an electron. The one that's practically begging for someone to come and take it. It’s the electron that’s thinking, "Man, this place is crowded, and Dad's always on my case. I'm outta here!" We're talking about the atom with the lowest ionization energy.
Imagine you’ve got a bunch of balloons at a party. Some are tightly tied, like they’re super attached to their helium. Others? They’ve got a loose knot, practically wiggling free. Ionization energy is like trying to pull those balloons away. You’ll have a much easier time with the one that’s barely holding on, won't you? That's the one with the lowest ionization energy.
Now, the big question is, how do we figure out which atom is the most "easily-escapable"? It all comes down to a few things, but the most important players are the size of the atom and how strongly the nucleus is pulling on those electrons. Think of it like a tug-of-war. If the electron is really far away from the nucleus, and the nucleus isn't that strong a puller, then it’s a pretty easy win for the electron to break free. If the electron is super close to a really strong nucleus, it’s going to take a whole lot more convincing – or a lot more energy.
Let's break down some of the usual suspects you might find on a "Which of the following..." list. Often, you'll see elements from different parts of the periodic table thrown in, like little puzzle pieces you need to sort. You'll have some metals, some non-metals, and maybe even a noble gas chilling in the corner, thinking they're too good for all this fuss.
Consider the noble gases, like Helium (He) or Neon (Ne). These guys are the ultimate introverts of the atomic world. They're perfectly happy being alone, with their electrons all snug and settled. They’ve got a full outer shell, which is like having your own private room with all the snacks you need. They have absolutely no desire to give up an electron. It takes a ridiculous amount of energy to pry one away from them. They're the ones who politely decline every invitation to a party because they'd rather stay home and organize their sock drawer.
On the flip side, you have the alkali metals, like Lithium (Li) or Sodium (Na). These are the overly friendly, outgoing types. They've got just one electron hanging out in their outer shell, like an awkward guest who doesn't really fit in with the main group. This lone electron is basically waving a tiny white flag, saying, "Anyone want me? I'm getting lonely over here!" It takes very little energy to convince this electron to leave and go find a new atom to hang out with. They're the ones who are always the first to volunteer for everything, even if they have no idea what they're doing.
So, when you're looking at a list and trying to pick the one with the lowest ionization energy, you're essentially looking for the atom that’s the biggest and has the weakest grip on its outermost electron. It’s like trying to find the shabbiest, most neglected toy in a toy box – the one that’s practically begging to be discarded.
Let's imagine a scenario. You're at a really crowded party, and everyone’s packed in like sardines. There's the nucleus, the grumpy host, trying to keep everyone in line. The electrons are the party guests. Some guests are glued to the host's side, loving the attention. These are the electrons held tightly by a strong nucleus in a small atom. They're like the folks who are super invested in the host's gossip and won't leave their side. To get them to leave the party (ionize), you'd have to practically drag them out, which takes a lot of effort (high ionization energy).
Then, you have the guests on the fringes of the party. They're further away from the host, maybe a little bored. If the host's grip on them isn't that strong, they'll happily take the first opportunity to slip out the door. These are your atoms with low ionization energy. They're the ones who are already halfway out the door, looking for an excuse to leave.
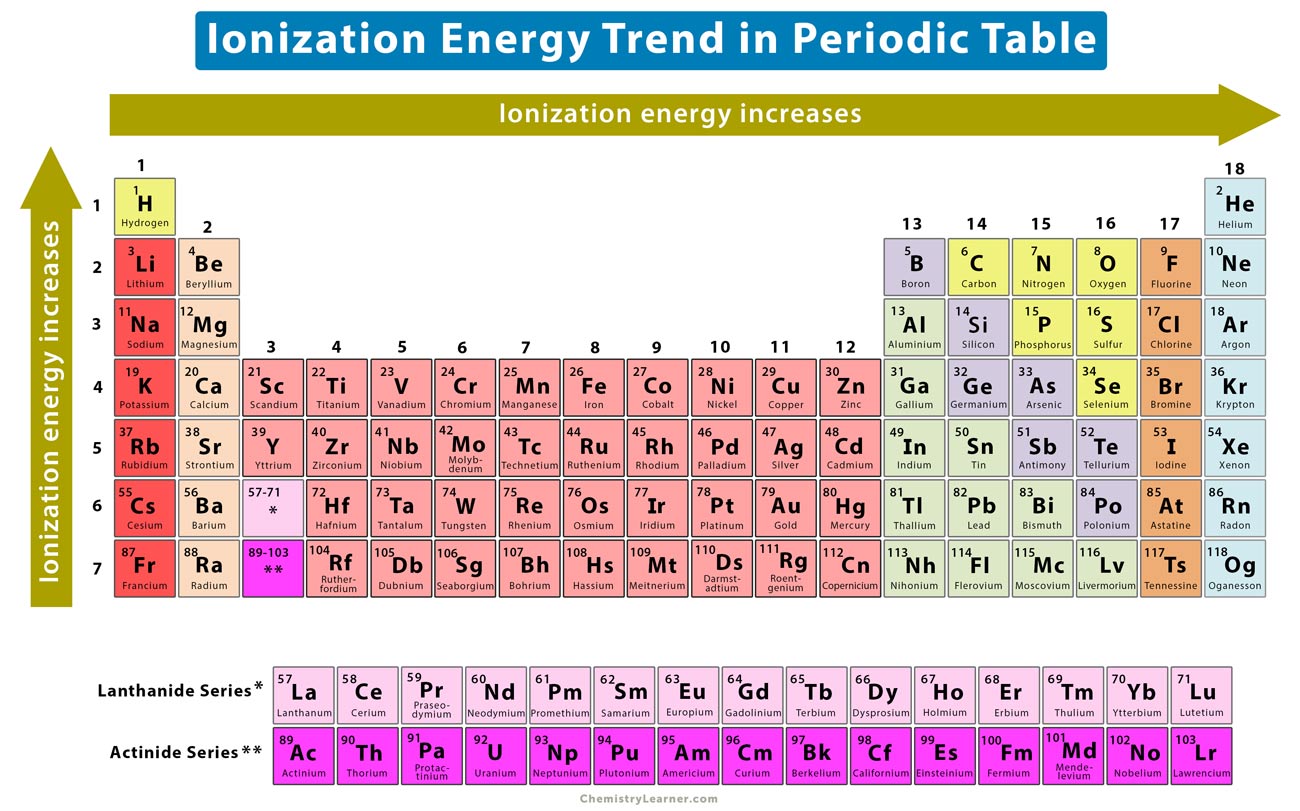
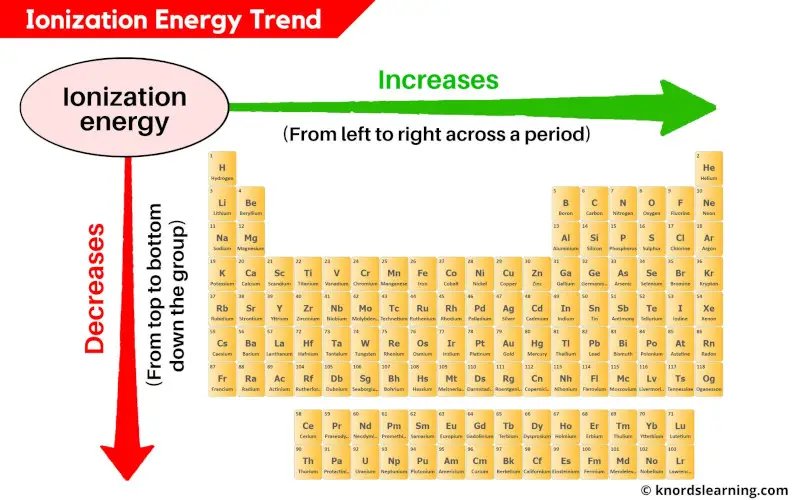
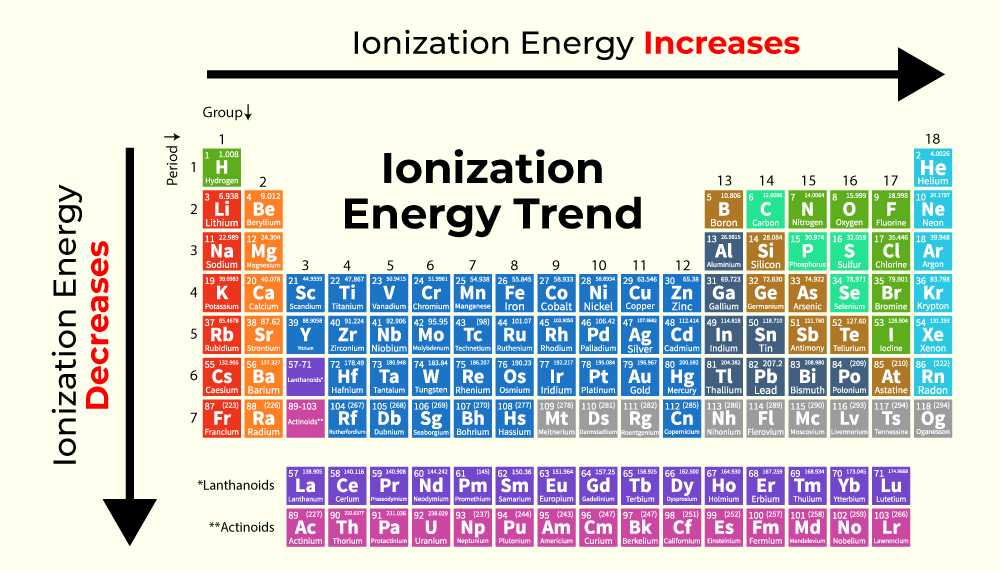
The periodic table is basically a cheat sheet for this whole atomic party situation. Generally, as you move down a group (that’s the up-and-down columns), the atoms get bigger. Think of it like adding more floors to a building. The electrons on the higher floors are further away from the ground floor nucleus. Plus, the inner layers of electrons act like a bit of a shield, weakening the nucleus's pull. So, elements lower down in a group tend to have lower ionization energies. They’re the ones who are living in the penthouse suite, feeling pretty detached.
Now, as you move across a period (that’s the left-to-right rows), things get a bit more interesting. The atoms generally get smaller. The nucleus is pulling harder on the electrons, and they’re all sort of squeezed together. It’s like a smaller party, but with more people trying to talk to the host. This means it takes more energy to pull an electron away. So, as you go from left to right across a period, the ionization energy generally increases. These are the folks crammed into the living room, practically breathing down the host's neck.
So, if you were given a list like:
- Lithium (Li)
- Beryllium (Be)
- Boron (B)
- Carbon (C)
What if the list looks like this?

- Sodium (Na)
- Potassium (K)
- Rubidium (Rb)
- Cesium (Cs)
Consider another scenario. You're at a family reunion. Your grandmother (the nucleus) is at the head of the table. Your cousins who live nearby (electrons in a small atom) are practically sitting on her lap, hanging on her every word. It takes a lot of effort to get them to leave and go play outside (high ionization energy).
But your cousins who live across the country (electrons in a large atom) are at the other end of the long dining table. They're a bit more independent. If Grandma just gives them a little nudge (a small amount of energy), they're happy to get up and go explore the backyard. These are your low ionization energy atoms. They’re the ones who are always checking their watch, wondering when they can leave.
The key takeaway is this: ionization energy is a measure of how tightly an atom holds onto its electrons. The lower the ionization energy, the easier it is to snatch an electron. And the main factors influencing this are the size of the atom and the strength of the nuclear pull.
So, when you see that question, "Which of the following has the lowest ionization energy?", don't panic. Just think about which atom is the biggest, the most spread out, and has an electron that's practically shouting, "Get me outta here!" It's about finding the atom that’s the least clingy, the most ready to embrace freedom. It's the atom that’s always looking for an excuse to say, "Peace out!"
Think of it as an atomic popularity contest, but instead of popularity, we're measuring how quickly an atom is willing to let go of its friends (electrons). The one that's the most eager to get rid of a friend is the winner – or rather, the one with the lowest ionization energy.
It's a bit like choosing between doing your taxes (high ionization energy – you're not letting go of that money easily!) and giving away a slightly-worn-out t-shirt you don't really like anymore (low ionization energy – easy peasy!). You're looking for the atomic equivalent of that old t-shirt.
So, next time you encounter this question, just picture those atoms. Picture the big, sprawly ones with electrons that are practically waving goodbye. Those are your winners. They’re the atoms that are easiest to "ionize," meaning they’re the most willing to lose an electron. It’s all about finding that electron that’s just dying for an adventure, and the nucleus that’s too tired or too far away to stop it. Easy, right?
