Which Of The Following Has The Largest Standard Molar Entropy
Hey there, fellow curious minds! Ever find yourself wondering about the hidden world of molecules and their messy, energetic lives? Today, we're diving into something called standard molar entropy. Sounds a bit fancy, right? But stick with me, because it’s actually a super cool way to understand how much "oomph" or randomness a substance has. Think of it as a measure of how much a substance likes to jiggle, wiggle, and generally spread out.
So, what's this all about? Imagine you’ve got a bunch of tiny dancers. Some are doing a very precise, synchronized ballet, all in neat rows. Others are at a wild, spontaneous dance party, flinging their arms and legs everywhere. Entropy is kind of like measuring the chaos of that dance party. The more chaotic, the higher the entropy.
Now, we're going to tackle a question that might pop up on a chemistry quiz (or just in your own thoughtful musings): Which of the following has the largest standard molar entropy? This is where things get really interesting, because it's not always about what looks the biggest or the heaviest. It’s about how those molecules can arrange themselves and move around.
Let's Break Down What "Standard Molar Entropy" Even Means
Okay, deep breaths. We’ll keep it simple.
"Standard" just means we're talking about specific conditions – usually a temperature of 298 Kelvin (that's about room temperature, 25 degrees Celsius) and a pressure of 1 atmosphere. It's like setting up a fair playing field so we can compare apples to apples.
"Molar" means we’re looking at one mole of a substance. A mole is just a super-duper large number of tiny things – about 602,200,000,000,000,000,000,000 of them! It’s a chemist's way of counting. So, we’re talking about the entropy of a specific, massive amount of stuff.
"Entropy" (that’s the big one!) is our measure of disorder or randomness. Think of it this way: imagine your bedroom. When it’s perfectly tidy, with everything in its place, it has low entropy. When it’s a complete disaster zone after a week of studying, it has high entropy! Molecules are kind of the same.
Why Does Entropy Change? Three Big Reasons
So, what makes one substance more "disordered" than another? There are a few key players:

1. The State of Matter: Solid, Liquid, or Gas?
This is probably the most intuitive factor. Think about water.
In ice (solid water), the molecules are locked in a pretty rigid crystal structure. They can vibrate a bit, but they're not going anywhere. That’s like a very orderly march. Low entropy.
In liquid water, the molecules can slide past each other. They're still close, but they have more freedom to move around. This is like a crowded but flowing dance floor. Medium entropy.
In steam (gaseous water), the molecules are zipping around everywhere, bouncing off each other and the walls of their container. They’re totally spread out and moving chaotically. This is the ultimate, wild dance party! High entropy.
So, as a general rule, gases have much higher standard molar entropies than liquids, and liquids have higher entropies than solids. This is a big clue!

2. The Size and Complexity of the Molecule
Now, this is where it gets a bit more nuanced. Imagine comparing a tiny, single-person dance to a huge, multi-person rave.
A small, simple molecule, like a single atom of helium (He), doesn't have a whole lot of ways it can move. It can just translate (move from point A to point B). Relatively low entropy.
A larger molecule, like a long chain of carbon atoms (think of a tiny piece of plastic), has more ways it can move. The atoms can vibrate, rotate, and the whole molecule can move around. It’s like a whole group of dancers who can spin, wave their arms independently, and still move together. Higher entropy.
The more atoms in a molecule, and the more complex its structure, the more ways it can distribute its energy and move, leading to higher entropy.
3. Molecular Weight (Sometimes!)
While not as dominant as the state of matter or molecular complexity, molecular weight can play a small role. Generally, heavier molecules have slightly more ways to store and distribute energy, which can contribute to higher entropy. Think of it like a heavier dancer having more inertia, which might lead to more dramatic (and thus more disordered) movements.
.jpg)
Putting It All Together: The Big Entropy Question!
Now, let’s get back to our hypothetical quiz question: Which of the following has the largest standard molar entropy? We'd be given a list of substances. To figure it out, we'd use our knowledge above!
Let's imagine a few common suspects:
- Solid Sodium Chloride (NaCl): A salt. It's a solid, and its ions are arranged in a pretty neat, repeating crystal lattice. Definitely on the low entropy side.
- Liquid Water (H₂O): We already talked about water. It's a liquid, so it's more disordered than a solid.
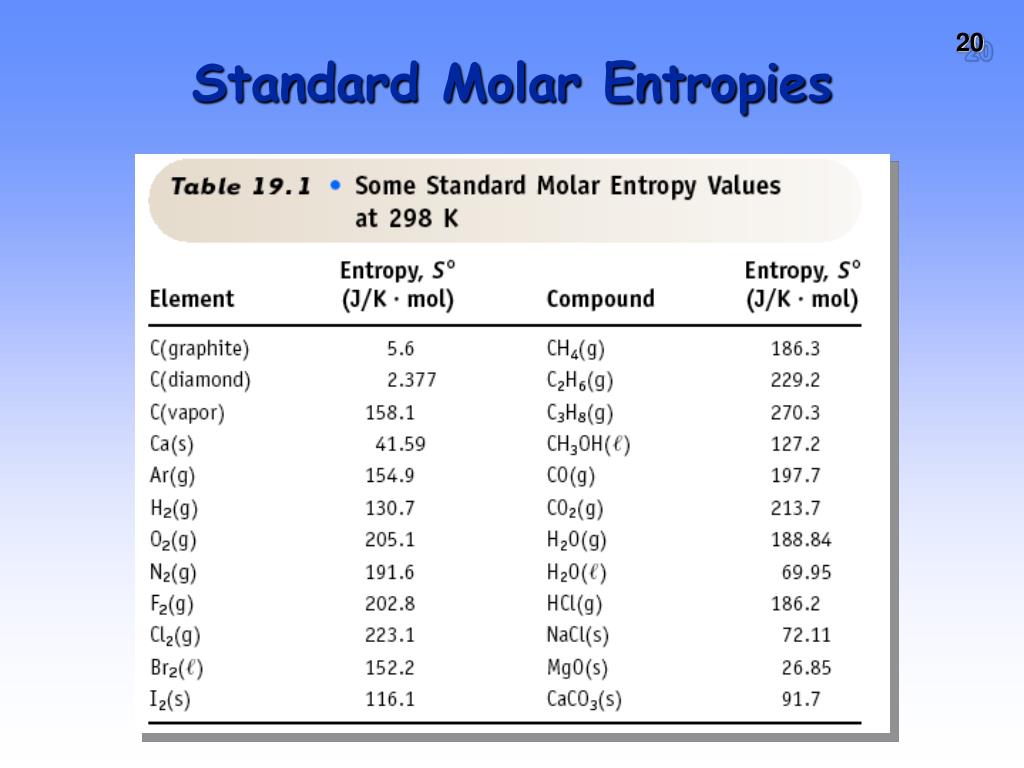
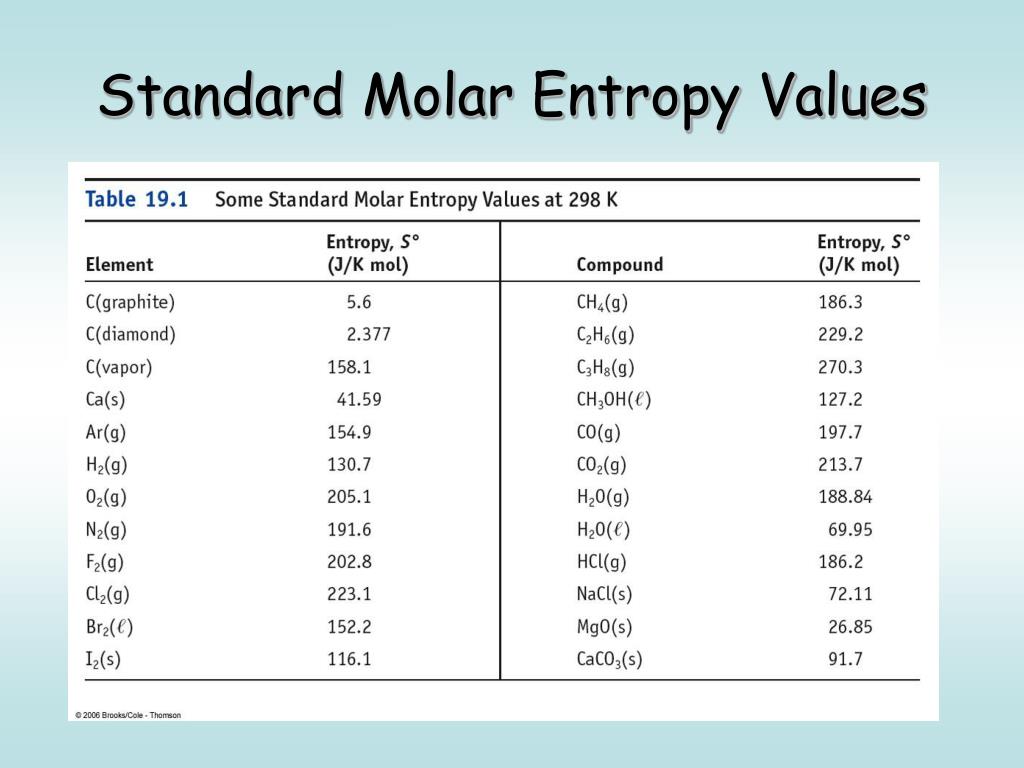
- Gaseous Oxygen (O₂): Oxygen is a gas, so it's going to have much higher entropy than a solid or a liquid. It's two atoms bonded together, so it's not super complex.
- Gaseous Carbon Dioxide (CO₂): Carbon dioxide is also a gas. This is where it gets interesting. CO₂ is made of three atoms (one carbon, two oxygens) arranged in a straight line. This means it has more ways to vibrate (bending and stretching modes) and rotate compared to O₂, which only has two atoms and fewer vibrational possibilities.
- Gaseous Methane (CH₄): Methane is a gas too. It's a relatively small molecule with one carbon and four hydrogens.
So, let's compare!
First, we eliminate the solids. Solid NaCl is out, as it’s in a highly ordered state.
Now we look at the gases. We have O₂, CO₂, and CH₄. All gases have high entropy compared to liquids and solids. But which one has the most?
We compare their complexity. O₂ is diatomic. CH₄ is tetrahedral. CO₂ is linear but has three atoms, allowing for more vibrational modes.
In this common scenario, gaseous substances will almost always have the highest standard molar entropy. When comparing gases, the one with the most complex molecular structure, meaning it has more atoms and more ways to vibrate and rotate, will typically have the highest standard molar entropy.
For example, if you were comparing O₂ (2 atoms) to CO₂ (3 atoms) and CH₄ (5 atoms), the CO₂ and CH₄ would generally have higher entropies than O₂. Between CO₂ and CH₄, their relative order can depend on the specific vibrational and rotational energy levels, but often, the one with more atoms and more complex internal motions wins the entropy game. Let's say, for argument's sake, CO₂ might have a slightly higher standard molar entropy than CH₄ in certain comparisons due to its specific vibrational modes.
It's All About Freedom!
Ultimately, the substance with the largest standard molar entropy is the one that has the most freedom to move, vibrate, and arrange itself in different ways. It’s the molecules that are living their best, most chaotic, widespread lives!
Isn't it cool how we can quantify something as abstract as disorder? It tells us so much about how substances behave and interact. So next time you see a gas happily filling a room, remember it's just embracing its incredibly high standard molar entropy!
Keep asking questions, keep wondering, and keep exploring the fascinating world around you!
