Which Of The Following Has The Highest Ionization Energy
Ever wondered what makes some atoms a bit more clingy than others when it comes to their electrons? It’s a question that might not seem to pop up in everyday conversation, but understanding it unlocks a fascinating peek into the fundamental building blocks of our universe. Today, we’re going to dive into the intriguing concept of ionization energy, and specifically, ponder which of a given set of elements might have the highest ionization energy. Think of it like a friendly puzzle about how much effort it takes to snatch an electron away from an atom. It’s not just for super-scientists; this knowledge can actually be quite fun and enlightening!
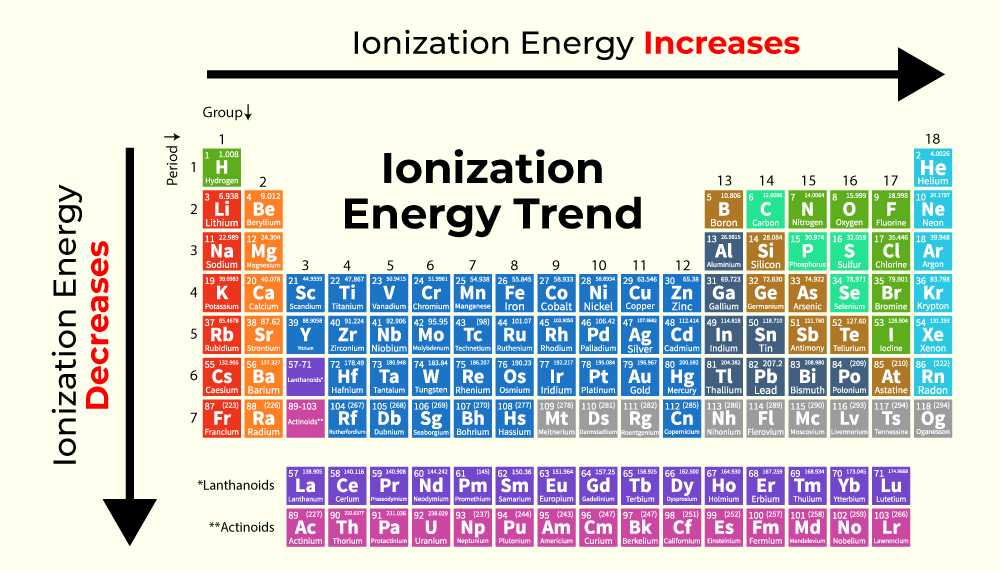
So, what exactly is ionization energy? In simple terms, it's the minimum energy required to remove the outermost electron from a neutral atom or molecule in its gaseous state. Imagine an atom as a tiny solar system, with electrons orbiting the nucleus. Ionization energy is like the force keeping those electrons in orbit. The higher the ionization energy, the more tightly bound that electron is, and the harder it is to pull it away. This property is incredibly useful in chemistry because it helps us predict how elements will behave. Understanding ionization energy is key to understanding chemical bonding, reactivity, and the very structure of the periodic table. It’s a fundamental property that explains why certain elements readily form compounds while others prefer to go it alone.
Where might you see this concept in action? Well, in education, it's a cornerstone of chemistry classes, helping students grasp the periodic trends and the underlying reasons for them. Beyond the classroom, ionization energy plays a role in fields like spectroscopy, a technique used to analyze the composition of materials by looking at how they interact with light. This has applications in everything from environmental monitoring to forensic science. Even in the world of materials science, understanding how easily electrons can be removed from atoms is crucial for developing new semiconductors or advanced batteries. It’s a behind-the-scenes player in a lot of the technology we rely on.
Curious to explore this yourself? You don’t need a fancy lab! A great way to start is by looking at a periodic table. Notice how ionization energy generally increases as you move from left to right across a period (row) and decreases as you move down a group (column). This pattern is your first clue! You can find charts online that list ionization energies for different elements. Try comparing elements from opposite ends of the periodic table. For instance, compare an alkali metal like sodium (which has a low ionization energy and readily loses an electron) with a noble gas like neon (which has a very high ionization energy and holds onto its electrons fiercely). You can also find interactive periodic table websites that let you click on elements to see their properties, including ionization energy. It’s a simple, visual way to start building your intuition about these fundamental atomic characteristics.
