Which Of The Following Has The Highest First Ionization Energy
Ever found yourself staring at a periodic table, perhaps while waiting for your artisanal coffee to brew, and wondered about the hidden drama within those neat rows and columns? It’s not just about colors and symbols, oh no. There’s a whole universe of energy dynamics going on, and today, we’re diving into a particularly electrifying topic: first ionization energy. Think of it as the atomic version of a celebrity’s entourage – how much effort (energy) does it take to snatch away one of their most prized possessions (an electron)? It's a concept that sounds super science-y, but trust me, it has a cool, almost relatable vibe once you get into it. And the burning question, the one that might keep you up at night between binge-watching sessions: Which of the following has the highest first ionization energy? Let’s break it down, shall we?
So, what exactly is this "first ionization energy" we’re nattering on about? Imagine an atom chilling out, minding its own business. It’s got a nucleus at its core, like the VIP lounge, and electrons orbiting around it, like the eager fans. The first ionization energy is the minimum energy required to remove the outermost electron from a neutral atom in its gaseous state. Think of it like trying to get that one perfect selfie with a pop star – you need to put in some serious effort to break through the security and the crowd, right? For an atom, that effort is measured in energy units, usually kilojoules per mole (kJ/mol).
Now, why does this matter? Well, understanding ionization energy is key to understanding how elements behave. It tells us how readily an atom will give up an electron, which is pretty fundamental to forming chemical bonds – the glue that holds everything together, from the water you drink to the smartphone you’re probably reading this on. It’s like knowing a person’s willingness to lend a hand; it tells you a lot about their personality and how they’ll interact with the world.
Let’s talk about the players in this energy game. We’re often presented with a list of elements and asked to identify the champ of high ionization energy. These lists usually include elements from different parts of the periodic table, and that’s where the magic happens. The periodic table isn't just a pretty chart; it's a masterpiece of organization that reveals trends in atomic properties. It’s like the Dewey Decimal System for atoms – everything has its place and its predictable neighbors.
Two major trends dictate ionization energy: atomic radius and effective nuclear charge. Keep these two in your mental toolkit, and you’ll be navigating ionization energy like a pro.
The Dance of Atomic Radius
First up, let’s get cozy with atomic radius. This refers to the size of an atom. As you move down a group (the vertical columns) on the periodic table, the atomic radius generally increases. Why? Because each new period adds another electron shell, pushing the outermost electrons further away from the nucleus. Think of it like adding more floors to a building; the top floors are further from the ground. These outermost electrons, being further away, are less tightly held by the positive pull of the nucleus. It’s like trying to hold onto a balloon when you’re standing on the ground versus when you’re on the roof – much easier to let go from higher up!
Conversely, as you move across a period (the horizontal rows) from left to right, the atomic radius generally decreases. Even though you’re adding more electrons, they are being added to the same electron shell. The number of protons in the nucleus increases, making the positive charge stronger. This stronger pull draws the electron cloud closer. Imagine a stronger magnet attracting a piece of metal; it pulls it closer. So, atoms get smaller as you go across a period.

Now, how does this tie back to ionization energy? Simple! Smaller atoms have higher ionization energies. Why? Because the outermost electron is closer to the positively charged nucleus and is therefore more strongly attracted. It takes a lot more energy to pry that electron loose. It's like trying to steal a cookie from a baby sitting right next to you versus trying to snag one from a toddler across a vast playground. Much harder with the closer, more watchful gaze!
The Power of Effective Nuclear Charge
Next on our elemental exploration is the concept of effective nuclear charge. This is the net positive charge experienced by an electron in an atom. While the nucleus has a full positive charge, the inner electrons act as a shield, or "screen," reducing the attraction felt by the outer electrons. So, the effective nuclear charge is the actual pull felt by the valence electron, taking into account this shielding effect.
As you move across a period from left to right, the number of protons in the nucleus increases, leading to a stronger nuclear charge. Even though there are more electrons, the shielding effect doesn’t perfectly counteract the increased nuclear pull. The result? The effective nuclear charge increases. This stronger pull makes the outermost electrons harder to remove, thus increasing the ionization energy.
As you move down a group, the added electron shells provide more shielding for the outermost electrons. While the nuclear charge increases, the increased distance and shielding mean the effective nuclear charge experienced by the valence electrons actually decreases. This weaker pull makes it easier to remove the outermost electron, leading to lower ionization energies.
Putting it All Together: The Periodic Table as Your Guide
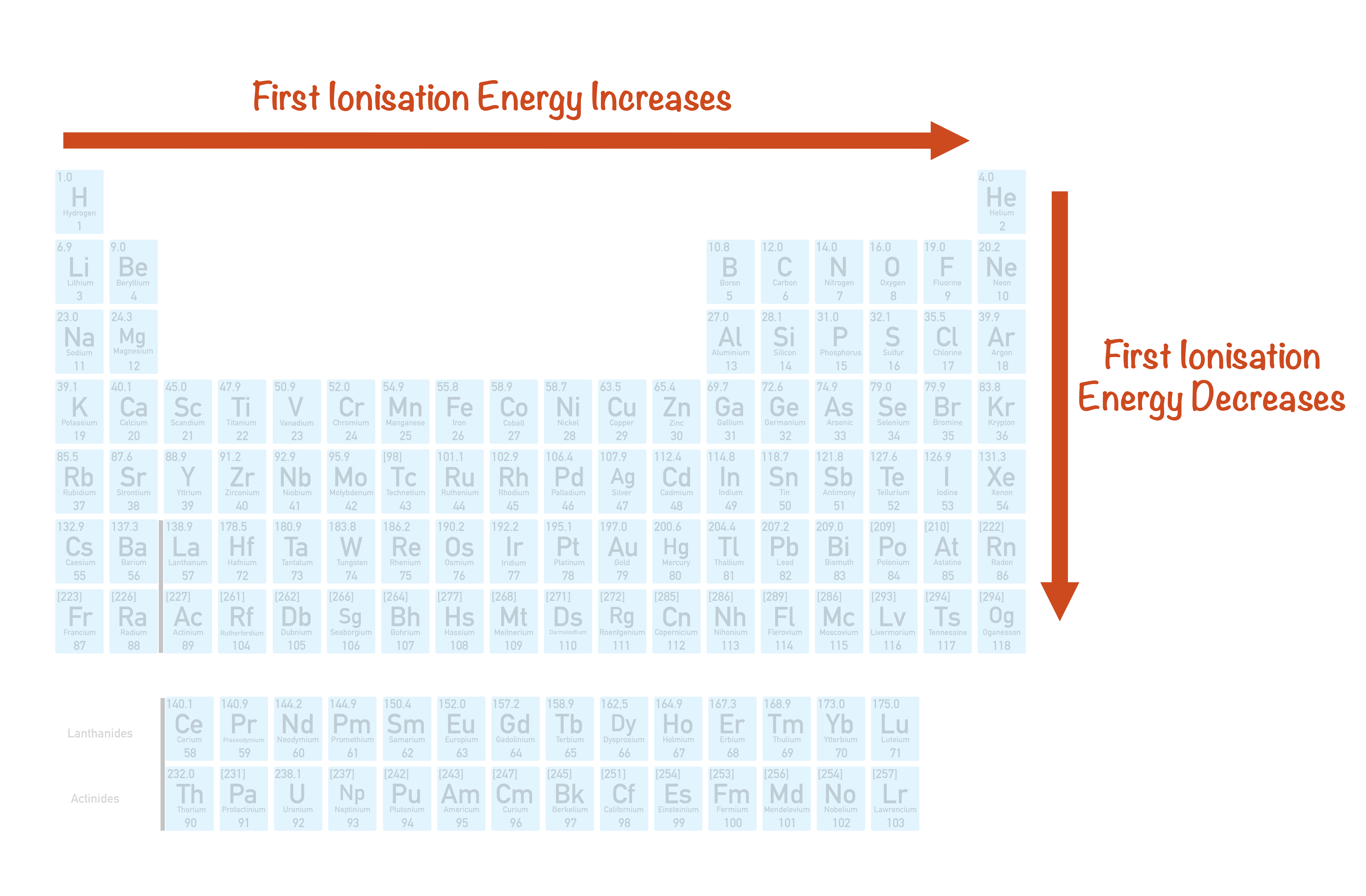
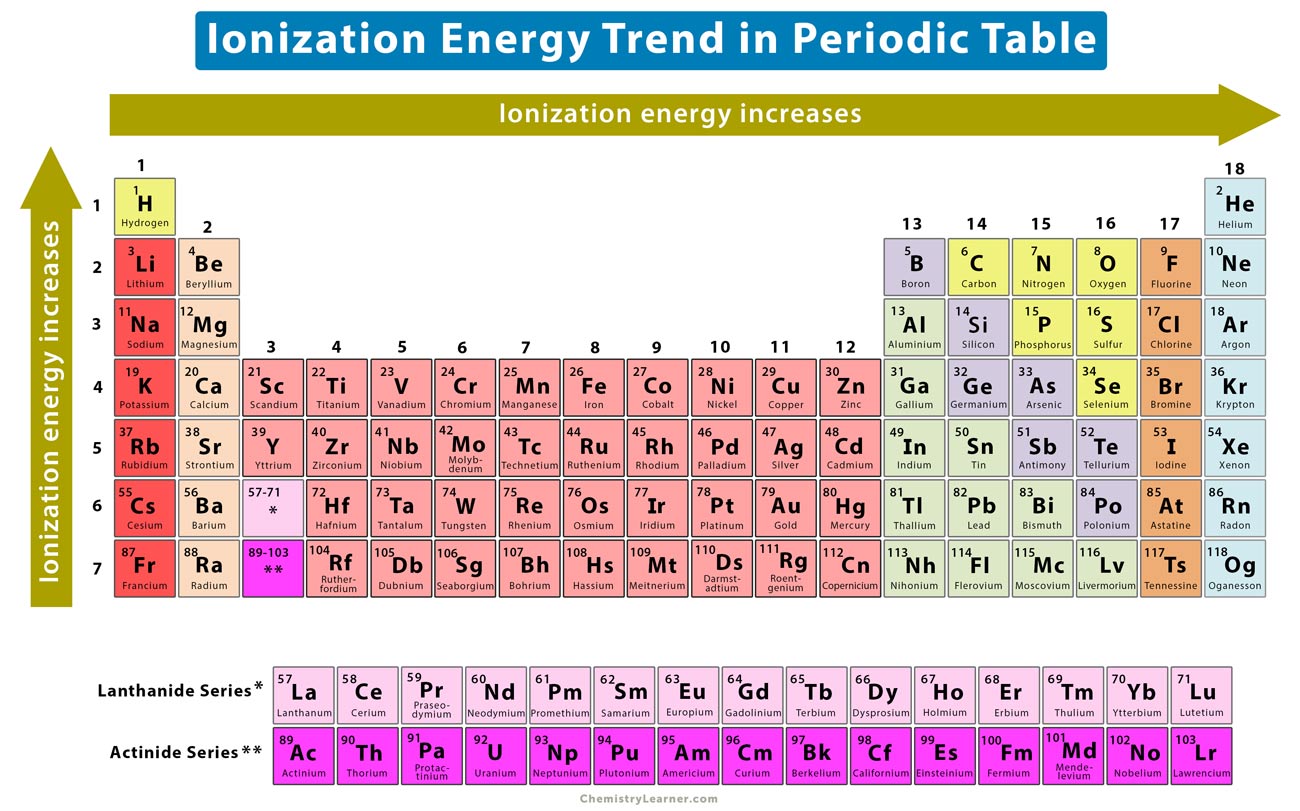
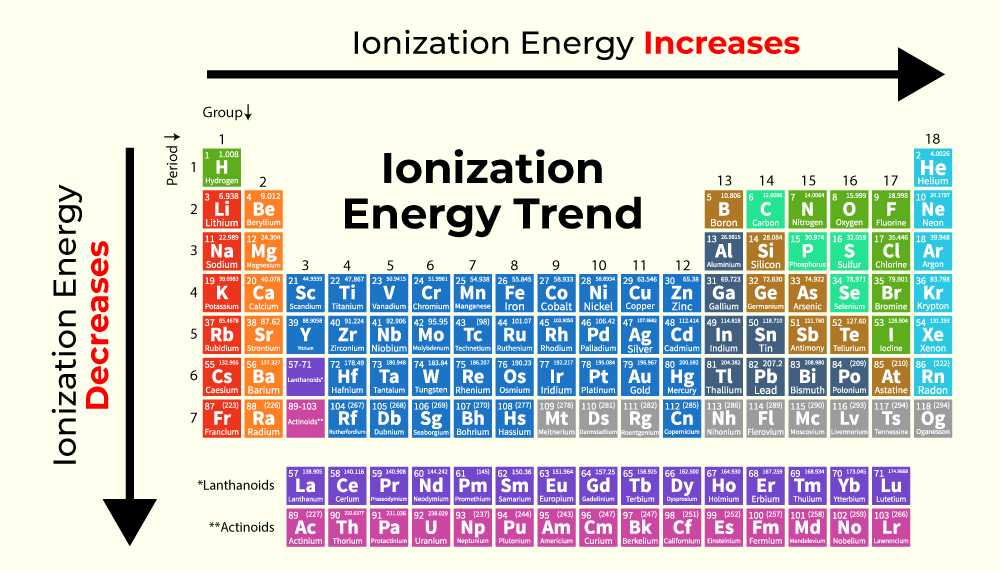
So, to recap:
- Ionization energy generally increases as you move across a period (left to right).
- Ionization energy generally decreases as you move down a group (top to bottom).
Think of the periodic table like a cozy neighborhood. Elements on the left are generally more relaxed and happy to share their electrons (low ionization energy – think alkali metals like Sodium). Elements on the right are a bit more guarded, holding onto their electrons tightly (high ionization energy – think halogens like Fluorine). The noble gases, way over on the far right, are the ultimate introverts – their electron shells are full, making them extremely stable and incredibly difficult to ionize. They have very high ionization energies.
The Contenders: A Hypothetical Lineup
Let’s imagine a common scenario where you might be asked: "Which of the following has the highest first ionization energy?" and given a list like:
- Lithium (Li)
- Sodium (Na)
- Potassium (K)
- Fluorine (F)
Let's analyze these contenders using our newfound knowledge.
First, let’s locate them on the periodic table. Lithium, Sodium, and Potassium are all in Group 1 (alkali metals). Lithium is in Period 2, Sodium in Period 3, and Potassium in Period 4. As we learned, ionization energy decreases as you go down a group. This means Potassium will have the lowest ionization energy among these three, followed by Sodium, and then Lithium will have the highest within this subgroup. They are all eager to give up an electron, but Lithium, being the smallest and with its valence electron closest to the nucleus, requires the most effort among the three to do so.

Now, let’s bring in Fluorine (F). Fluorine is in Group 17 (halogens) and is in Period 2. This is a crucial position! Fluorine is in the same period as Lithium. As we move across a period, ionization energy generally increases. Fluorine is much further to the right than Lithium. Moreover, Fluorine has a significantly higher effective nuclear charge attracting its valence electron.
Comparing Lithium (Period 2, Group 1) and Fluorine (Period 2, Group 17), Fluorine’s valence electrons are much closer to a nucleus with a greater positive charge and less shielding from inner electrons. This makes Fluorine's outermost electron held much, much more tightly.
Therefore, between Lithium, Sodium, Potassium, and Fluorine, Fluorine (F) will have the highest first ionization energy. It takes a considerable amount of energy to persuade Fluorine to part with even one of its precious electrons. It’s like trying to convince a cat to willingly jump into a bath – it’s going to take a lot of convincing and perhaps some creative coaxing!
Beyond the Big Players: Fun Facts and Nuances
It’s not always a straightforward march. There are sometimes little quirks. For instance, within a period, there can be slight dips and rises. For example, the ionization energy of Boron is slightly lower than that of Beryllium. This is because Beryllium has a filled 2s subshell, which is a stable configuration. Removing an electron from Boron involves disrupting a subshell that has unpaired electrons, making it slightly easier to remove compared to Beryllium's more stable filled shell situation. These are the atomic equivalent of those surprising plot twists in your favorite TV show – keeps things interesting!
And what about those noble gases? Helium (He), Neon (Ne), Argon (Ar), and so on, are the kings and queens of high ionization energy. Their outermost electron shells are completely full, making them incredibly stable and reluctant to participate in chemical reactions. It takes a monumental amount of energy to even think about removing an electron from them. They are the ultimate hermits of the chemical world, perfectly content in their electron-rich solitude. So, if Helium or Neon ever pops up on your list, they're usually the winners for the highest ionization energy.
Interestingly, the concept of ionization energy is not just confined to chemistry textbooks. It has real-world applications. For example, in spectroscopy, ionization energies help identify elements. In materials science, it can influence how materials interact with light and electricity. Even in astrophysics, understanding ionization energies is crucial for studying stars and nebulae. It’s a fundamental property that underpins so much of our physical world.
Think about it: the way elements bond, the colors of fireworks (which often involve exciting electrons and then observing the energy they release as they return to their ground state), the very conductivity of metals – all these phenomena are intricately linked to how easily electrons can be removed or added. So, that seemingly abstract concept of ionization energy is actually woven into the fabric of the universe around us.
And the ultimate element, the one that takes the most oomph to ionize? That would be Helium (He). With only two electrons in its first shell, tightly bound to a nucleus with two protons, and no inner shielding electrons, Helium has an astronomically high first ionization energy. It’s the ultimate loner, the rock star who demands the highest security and the biggest rider. It’s a testament to the elegance and power of atomic structure.
A Daily Dose of Ionization Energy
So, the next time you look at a table of elements, or even just glance at the periodic table poster in your local café (you know, the one next to the fair-trade quinoa bowls), remember the hidden drama of ionization energy. It’s a dance of attraction and repulsion, a testament to the forces that govern the very building blocks of our reality.
Reflecting on ionization energy, it's a bit like understanding ourselves and our relationships. We all have our "outermost electrons" – our vulnerabilities, our boundaries, the parts of ourselves that are most accessible. Some of us are more open and readily share our energy and thoughts (low ionization energy), while others are more guarded, requiring significant effort and trust to reveal their inner selves (high ionization energy). Recognizing these differences, both in atoms and in people, fosters a greater understanding and appreciation for the unique ways in which things interact with the world. It’s a reminder that even in the seemingly rigid laws of science, there’s a relatable rhythm, a fundamental truth about attraction, resistance, and the energy it takes to connect.
