Which Of The Following Exhibits Dipole Dipole Attraction Between Molecules
Ah, the humble molecule! While it might sound like something you’d encounter in a stuffy chemistry lab, the way these tiny building blocks interact is actually at the heart of so many things we enjoy and rely on every single day. Think about the satisfying fizz of your favorite soda, the way your skin feels after applying a luxurious lotion, or even the way the colors in a vibrant painting seem to pop. These everyday wonders often owe their existence to the subtle, yet powerful, forces between molecules. Today, we're going to dive into one of these fascinating forces: dipole-dipole attraction. Don't worry, we’ll keep the jargon to a minimum and focus on why this invisible dance matters to you!
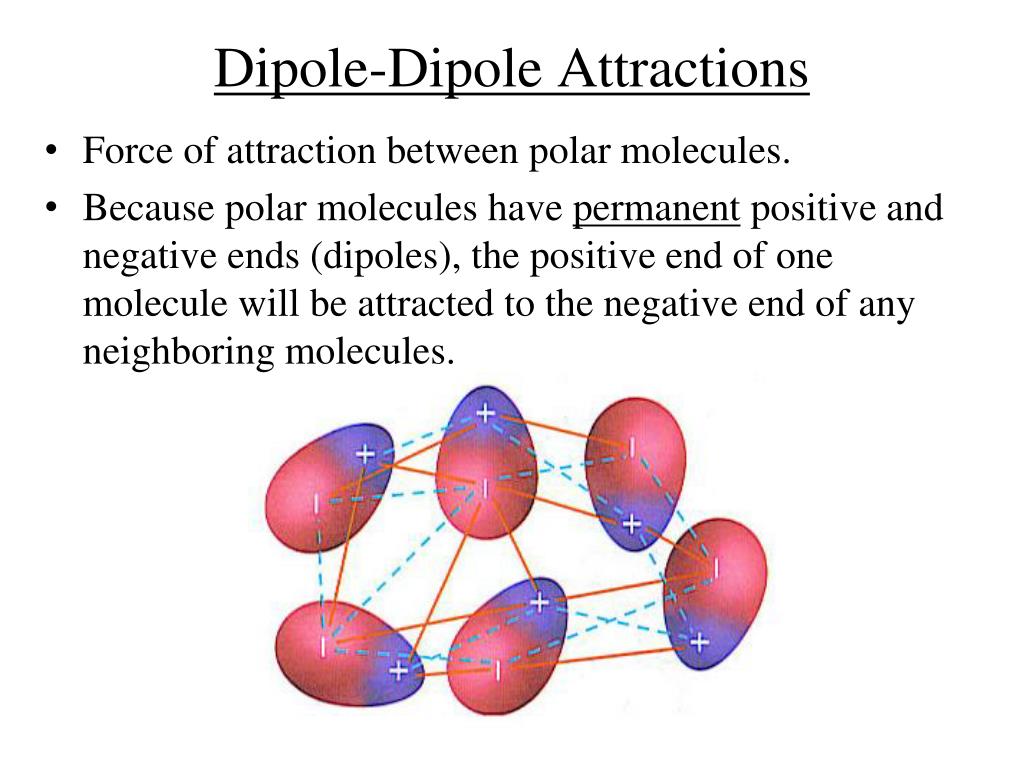
So, what exactly is dipole-dipole attraction, and why should we care? Imagine molecules like tiny magnets, but instead of north and south poles, they have slightly positive and slightly negative ends. This uneven distribution of electrical charge is called a dipole. When molecules with these dipoles get close, the positive end of one molecule is attracted to the negative end of another. It's like a gentle hug between molecules, holding them together. This attraction is crucial for many everyday phenomena. It influences how substances dissolve, how they change from liquid to gas, and even how they stick to other surfaces. Without dipole-dipole forces, many of the materials and processes we take for granted simply wouldn't work!
You encounter dipole-dipole attraction more often than you might think. Take water, for example. Its bent shape means the oxygen atom has a partial negative charge, and the hydrogen atoms have partial positive charges. This makes water a highly polar molecule, and the dipole-dipole attractions between water molecules are responsible for its ability to dissolve so many substances – it’s the universal solvent for a reason! Think about perfumes. The pleasant aromas we enjoy are often due to molecules with dipoles interacting with each other and with our olfactory receptors. Even the way paint sticks to a wall or how adhesives work involves these subtle molecular attractions. It's the silent architect behind many of our sensory experiences and practical solutions.
Now, how can you better appreciate or even enhance your experience with things that rely on dipole-dipole attraction? For starters, when you're choosing products, consider their composition. For instance, if you're looking for a lotion that spreads smoothly and feels nourishing, you might be drawn to products with ingredients that exhibit strong dipole-dipole interactions, leading to a pleasant texture. When you're enjoying a cup of coffee or tea, take a moment to appreciate how the water's polarity allows it to extract the delicious flavors from the coffee grounds or tea leaves. If you're a painter or enjoy crafts, understanding how different solvents interact with pigments can lead to better results. Perhaps try experimenting with different types of glues or paints and observe how their application and drying times differ, which can be a subtle indication of molecular forces at play. It’s about paying attention to the small details that make the big things happen in our world!
