Which Of The Following Elements Has The Lowest Ionization Energy
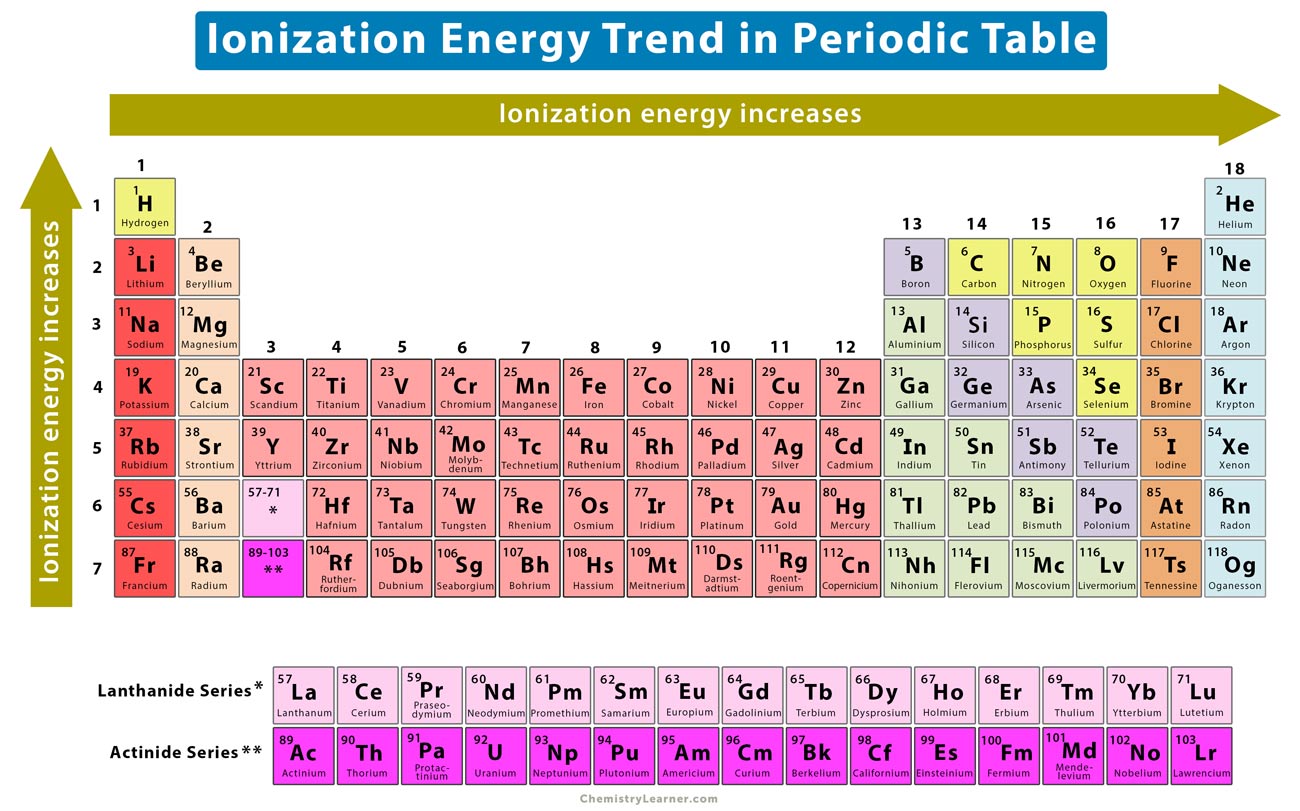
Ever feel like some things just want to let go? Like that sock that mysteriously vanishes in the wash, or your willpower around a freshly baked batch of cookies? Well, in the wild, wacky world of atoms, there are certain elements that are total rebels. They're practically itching to ditch an electron, to fling it out into the universe like a kid tossing a toy. And today, we're talking about which of these atomic free spirits has the lowest ionization energy.
Now, before you start Googling "ionization energy" and wondering if it's a new kind of energy drink, let's just say it's basically the amount of oomph it takes to convince an atom to give up one of its little electron buddies. Think of it as the "letting go" factor for atoms. Some hold on tight, like a toddler with a precious crayon. Others are like, "Here, you can have it! See ya!"
So, the big question, the one that keeps chemistry nerds up at night (or maybe just gives them something fun to chat about at parties), is: which element is the ultimate electron- McNugget yeeter? We've got a lineup of contenders, each with their own unique brand of atomic nonchalance.
First up, we have the noble gases. These guys are like the introverts of the periodic table. They're perfectly happy in their own little atomic bubbles. They’ve got their electron shells all snug and full, and they’re not about to mess with that. Trying to snatch an electron from a noble gas? Good luck! It’s like trying to take a cookie from a grumpy badger guarding its den. They’ve got high ionization energy, meaning they put up a serious fight. They’re the opposite of what we're looking for here.
Then we have the halogens. These are the drama queens. They’re one electron short of a full house and they really want to complete their set. They're pretty eager to grab an electron from someone else, which means they don't like giving their own up easily. They're like the kid who desperately wants to join a game but is also super possessive of their own toys. So, nope, not them either. They have fairly high ionization energies, too.
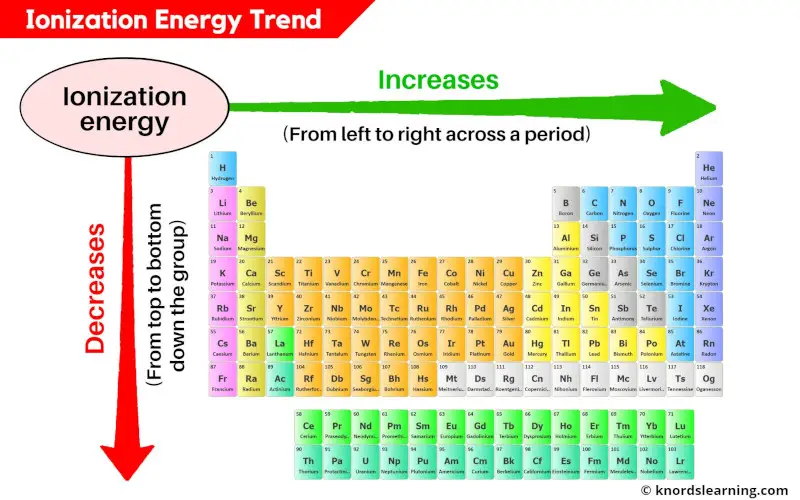
Now, let's venture into the heart of the periodic table, where things get a little more… shall we say, generous. We’re talking about the metals. And not just any metals, but the alkali metals. These guys are the poster children for "give it away." They’ve got one lone electron hanging out in their outermost shell, practically waving hello and saying, "Take me! I'm bored!"
Imagine your atomic element is a small business. The noble gases are the ones with a perfectly balanced ledger and no need for outside investment. The halogens are the ones desperately seeking a loan to expand. But the alkali metals? They're the ones with a surplus of cash just sitting there, practically begging someone to borrow it. They’re practically giving their electrons away.

So, who is the absolute champion of this electron-flinging fiesta? Drumroll, please! It's none other than Francium. Yes, Francium! This element is so laid-back about its electrons, it’s practically horizontal. It’s the ultimate chill atom. If atoms had personalities, Francium would be the one lounging on a beach with a tiny umbrella in its drink, utterly unbothered by the need to hold onto anything.
Think about it. Francium is right down at the bottom of the alkali metal group. As you go down that group, the atoms get bigger. Their outer electron is further away from the positively charged nucleus. It's like trying to hold onto a balloon with a really long string – easier for the balloon to float away, right? The nucleus has less of a grip. So, the further down you go in the alkali metals, the easier it is to pry that electron loose.
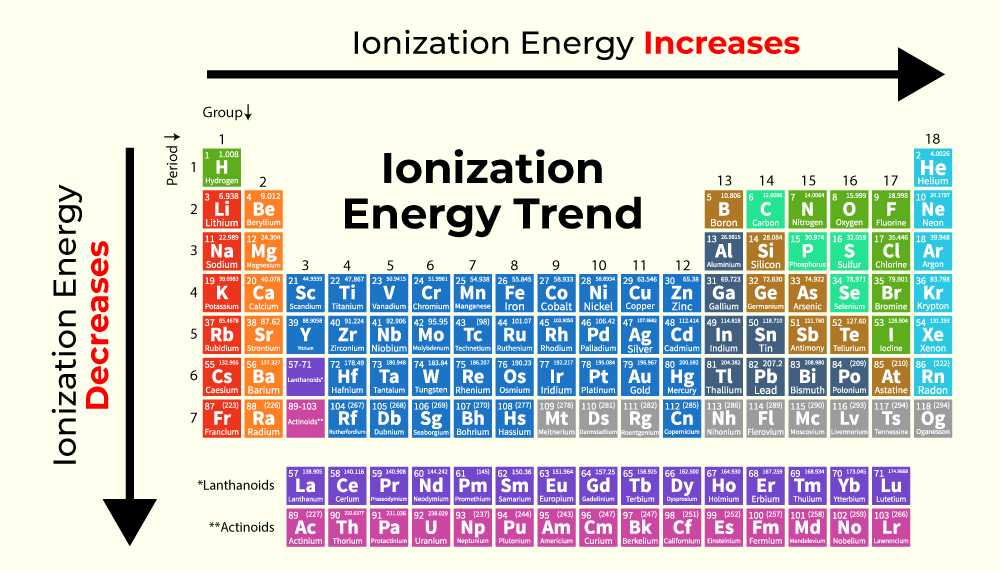
And Francium? It's the king of this low-energy, electron-shedding castle. It has the lowest ionization energy of all the naturally occurring elements. It’s like, "Electron? What electron? Oh, that one? Yeah, it’s gone. Didn’t even notice." It’s so eager to lose an electron, it almost feels rude to ask it to keep it.
Of course, Francium is also super rare and quite radioactive, so you're not going to find it chilling in your kitchen drawer. But in the grand scheme of atomic tendencies, it’s the undisputed champ of letting go. It’s the element that understands that sometimes, the best way to feel lighter is to just ditch what’s weighing you down. In this case, that’s an electron. A very, very easily ditchable electron.
So, the next time you hear about ionization energy, you can chuckle and think of Francium, the element that’s basically the human equivalent of hitting the snooze button one too many times. It just can’t be bothered to hold on. And in its own atomic way, that’s kind of admirable. Or at least, it’s a fun little science fact to impress your friends with. You can tell them, with a knowing smile, about the element that has the lowest ionization energy and why it’s so… well, loose.
