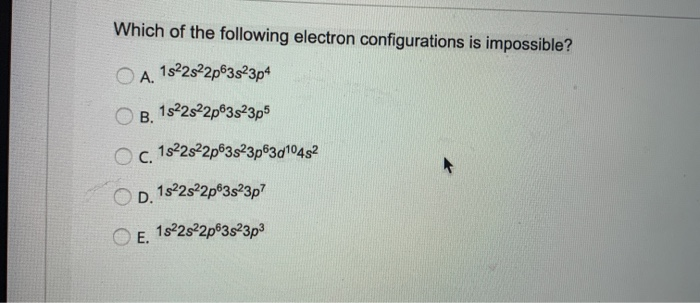
Which Of The Following Electron Configurations Is Impossible

Alright, let's dive into the wonderfully weird world of electron configurations. Now, I know what you're thinking. "Electron configurations? Sounds like homework I failed." But hear me out! It's actually kind of like picking out outfits for tiny little particles. And just like with my own wardrobe, sometimes you pick something that just… doesn't work.
We're going to play a little game. Imagine we have a few different ways these electrons, these teeny-tiny specks that orbit the nucleus of an atom, decide to arrange themselves. Think of them as deciding where to sit in a very special stadium. Each seat has a specific label, and there are rules about who can sit where.
Now, the universe, bless its chaotic heart, has some pretty strict rules about this. It's not like you can just cram a million electrons into one tiny, comfy chair. Nope. There's an order to things. There are shells, subshells, and orbitals. It's like a cosmic apartment building with very specific floor plans.
So, let's look at some of these proposed arrangements. Imagine we're looking at a lineup of potential electron configurations, like nominees for an award. Some of them are perfectly plausible. They follow all the cosmic etiquette. They’re the supermodels of the atomic world, effortlessly fitting into their designated spots.
But then, there are the others. The ones that make you scratch your head. The ones that just… don't make sense. These are the electron configurations that are, shall we say, a little out there. They're the fashion faux pas of the subatomic realm.
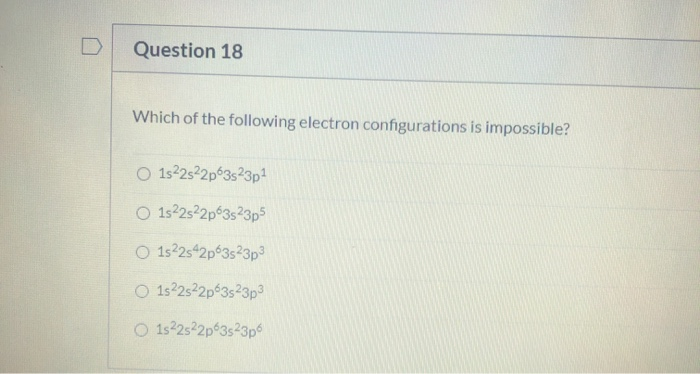
Let's take a peek at a few of these hypothetical scenarios. Picture this:

1s²2s²2p⁶3s¹
This one? Totally fine. Imagine the first floor (the '1s') is full, with two electrons. The second floor ('2s' and '2p') is also completely occupied. And then, on the third floor ('3s'), we have one lonely electron. It’s like a perfectly organized bookshelf. Everything in its place.
How about this one?
1s²2s²2p⁴
Solved Which of the following electron configurations is | Chegg.com
Also, perfectly acceptable. The first floor is packed. The second floor is getting there, with two electrons in the '2s' orbital and four in the '2p' orbitals. Think of it as a cozy apartment where everyone has a designated spot, even if some rooms are a little more crowded than others.
Now, here’s where things start to get… interesting. Let’s consider this possibility:
1s²2p⁶3s²
Wait a minute. Did we just skip the entire '2s' floor? That's like saying you're going to live on the third floor of a building but completely ignore the second floor. It’s just… not how the elevator works. The electrons have to fill up the energy levels in a specific order. They can't just hop over a whole section.
It’s like trying to drive your car directly to the third lane on the highway without ever going through the second lane. The road simply isn't built that way. The electrons are bound by these fundamental rules. They have to ascend through the energy levels sequentially. They can't just skip a grade, so to speak.
This is where the magic of impossibility comes in. We're looking for that one configuration that breaks the cosmic traffic laws. It's the one that says, "I'm going to wear socks with sandals to a formal ball." It's an egregious violation of established norms.
Let's imagine another oddball:
1s²3s¹
Solved 38. Which of the following electron configurations | Chegg.com
Again, what happened to the '2s' and '2p' levels? It’s like trying to build a LEGO tower by stacking the bottom brick and then immediately putting the fifth brick on top. You're missing crucial support structures! These lower energy levels are essential for the electrons to occupy before they can even think about venturing to higher energy levels.
It's not just about having a certain number of electrons. It's about where they decide to hang out. The universe has a preference for order, a cosmic tidiness that cannot be ignored. Think of it as a strict librarian who insists you check out books in the correct order from the Dewey Decimal System. You can't just pull a book from the very back without browsing the shelves in between.
So, when we present you with a list of potential electron configurations, your mission, should you choose to accept it, is to be the atomic fashion police. You're looking for the outfit that's just plain wrong. The arrangement that defies the fundamental principles of how electrons behave. It's the one that whispers, "This is not going to fly, darling."
The impossible configuration is the one that tries to cheat the system. It's the one that suggests a shortcut where no shortcut exists. It’s the one that simply violates the basic rules of energy level filling. It’s the anomaly, the outlier, the one that makes even the most seasoned chemist raise an eyebrow and mutter, "Well, that's just not how it's done." And that, my friends, is a beautiful thing in its own peculiar way.