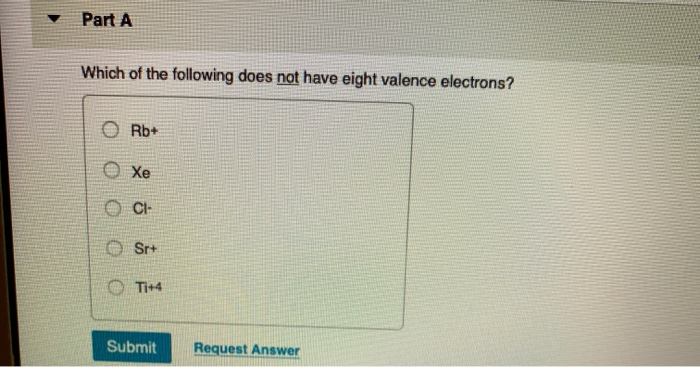
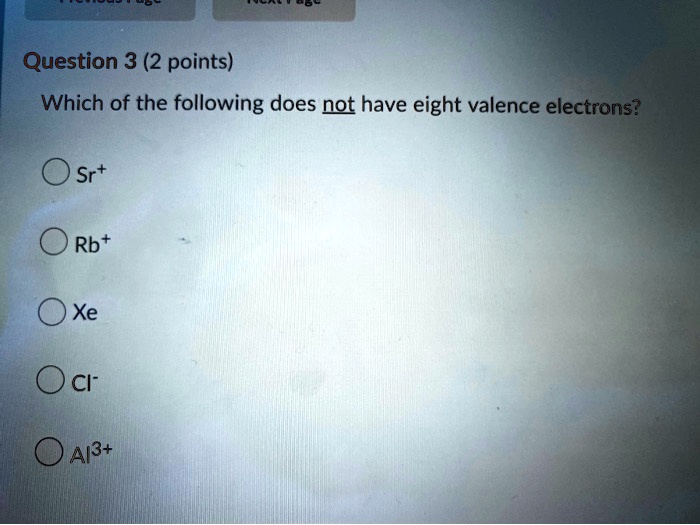
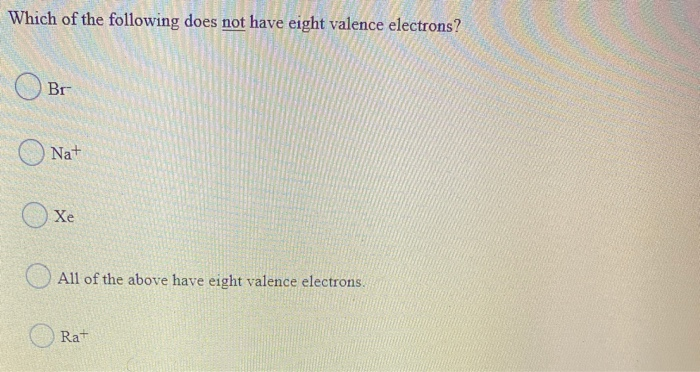
Which Of The Following Does Not Have Eight Valence Electrons

Alright, gather ‘round, you lovely bunch of curious cats and chemistry dabblers! So, imagine this: we’re all hanging out at our favorite cozy café, the aroma of freshly brewed coffee wafting through the air, and someone – let’s call them Barry, because Barry always has the most obscure trivia – throws out a question that’s supposed to be a bit of a brain-tickler. Barry, bless his cotton socks, loves these “which of the following…” quizzes. Today, he’s got one that’s all about the microscopic party animals of the universe: atoms! Specifically, he’s asking, “Which of the following does NOT have eight valence electrons?”
Now, before your eyes glaze over like a perfectly baked donut, let’s break this down. Think of atoms as tiny LEGO bricks that build everything. Seriously, everything! Your phone, your latte, that questionable fashion choice you made in the 90s – it’s all atoms. And these atoms have these little buzzing things around them called electrons. These electrons are like the social butterflies of the atomic world, flitting around the nucleus. But some electrons are more… well, let’s say strategically placed than others. We’re talking about the valence electrons. These are the outermost electrons, the ones that get to mingle with other atoms and decide whether to form a bond, steal a friend’s electron, or just do their own thing.
And here’s the juicy gossip: most atoms, when they’re feeling particularly stable and, dare I say, happy, like to have eight valence electrons. It’s like they’ve reached their personal comfort zone, their atomic happy place. It’s called the octet rule, and it’s the reason why so many molecules stick together like superglue. Imagine a bunch of kids at a party, and everyone wants to hold hands with exactly eight friends. It’s a crowded dance floor, let me tell you!
So, Barry’s question is basically asking: which atom is the odd one out at this eight-electron party? Who’s the rebel? Who’s the introvert who can only handle three friends? Who’s the show-off with a ridiculously large entourage?
Let the Atom-Identifying Games Begin!
Barry, in his usual dramatic fashion, probably scribbled down some options on a napkin, looking all smug. Let’s invent some suspects for our little mystery. We’ve got:
- The Noble Gases (the ultimate introverts of the periodic table)
- The Alkali Metals (the overly generous electron givers)
- The Halogens (the electron hoarders)
- And a couple of other characters who might be playing by different rules.
Now, we need to figure out who’s not rocking that eight-electron vibe. Let’s dive in, shall we? Think of the periodic table as a really organized, slightly nerdy dating app. Each element has a profile, and we’re looking at their "electron count" section.
Suspect Number One: The Noble Gases (Helium, Neon, Argon, etc.)
Ah, the Noble Gases. These guys are the ultimate wallflowers. They are so content with their electron situation that they rarely interact with anyone. Why? Because most of them already have eight valence electrons! Neon has 8, Argon has 8, Krypton has 8. They’re the epitome of "I’m good, thanks." However, there’s one exception, a mischievous little imp named Helium. Helium is like the toddler at the party who insists on sitting in the corner with just one best friend. It only has two valence electrons, and for Helium, that’s its happy place. So, if Helium were an option, it would be a strong contender for our “not eight” club. It’s the exception that proves the rule, the lone wolf in a pack of social butterflies.
Suspect Number Two: The Alkali Metals (Lithium, Sodium, Potassium, etc.)
These guys are the opposite of the Noble Gases. They’re like the people who always offer to pay for the first round of drinks. They have one valence electron in their outermost shell. They’re practically begging someone to take it off their hands. So, an alkali metal is definitely not going to have eight valence electrons. They’re desperately trying to get rid of their one, not accumulate eight! Imagine Sodium (Na) trying to get to eight valence electrons. It would have to steal seven! That’s exhausting, and frankly, a bit desperate. So, it’s much easier for Sodium to just give away its one lonely electron and feel all stable and complete with a full inner shell. It’s like decluttering your life, but with electrons.
Suspect Number Three: The Halogens (Fluorine, Chlorine, Bromine, etc.)
These are the clingy ones. The ones who are always looking to borrow something. Halogens have seven valence electrons. They’re so close to that magical number eight. They’re like someone standing at the door of the party, peering in, really wanting to join in. They’ll happily snatch one electron from another atom to achieve that coveted octet. So, a halogen itself doesn’t have eight valence electrons, but it’s constantly striving for it. Think of Chlorine (Cl). It’s got seven, and it’s always eyeing up a nice, lone electron from a neighbor. It’s the ultimate electron borrower.
So, Who’s the Non-Eight-Electron Culprit?
Barry’s question, “Which of the following does NOT have eight valence electrons?”, could have a few answers depending on the list he provided. However, the most common and fundamentally important examples are:
- Helium: As we discussed, it’s perfectly happy with its two valence electrons. It’s the OG chill atom.
- Alkali Metals (like Sodium, Lithium): These guys only have one valence electron and are looking to ditch it, not hoard eight.
- Alkaline Earth Metals (like Beryllium, Magnesium): These fellas have two valence electrons. They’re a bit more laid back than the alkali metals but still not hitting the octet goal on their own. They’re like the group who brought a two-person board game to a party of eight.
If Barry’s options included any of these, then that’s your answer! It’s the atom that doesn’t fit the popular "eight is great" club. It’s the lone wolf, the solo artist, the one who prefers a smaller circle. It’s the atom that reminds us that in the grand, chaotic, and utterly fascinating universe of chemistry, not everyone plays by the same rules. And isn’t that just the most wonderfully, hilariously, atomically true thing?
So next time Barry whips out his trivia, you’ll be armed with the knowledge of electron-sharing, octet-seeking, and the peculiar charm of atoms who march to the beat of their own electron drum. Now, who wants another coffee? I’m feeling an urge to bond with a pastry.
