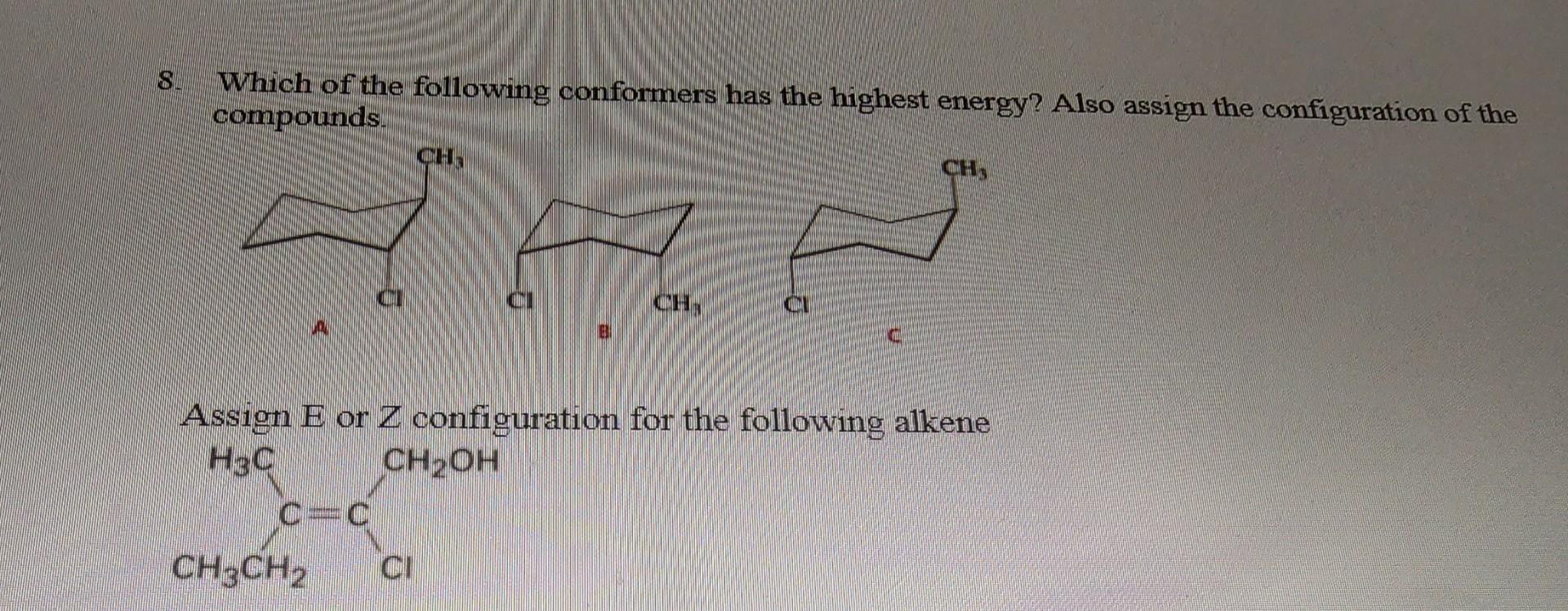
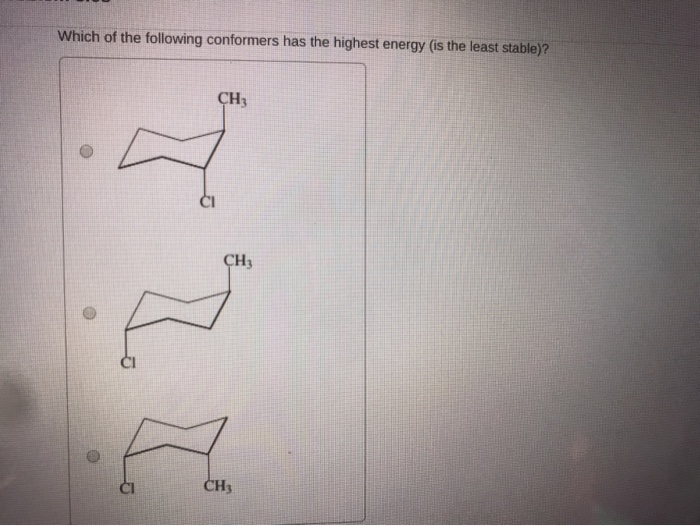
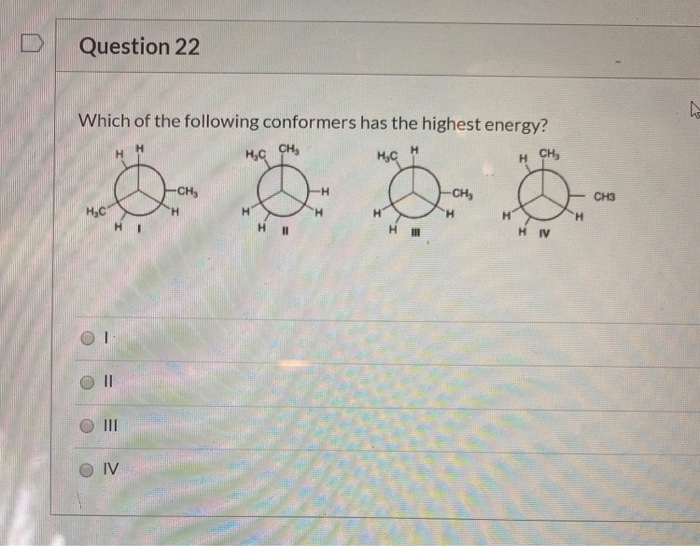
Which Of The Following Conformers Has The Highest Energy

Hey there, science curious peeps! Ever looked at a molecule and thought, "Man, that thing's gotta be doing some serious gymnastics"? Well, you're not wrong! Today, we're diving into the wild world of molecular shapes, specifically, the super fun, slightly bonkers question: Which of the following conformers has the highest energy?
Now, before you run for the hills imagining complex equations, let's keep it light and breezy. Think of molecules like little Lego creations. They can twist and turn and bend. These different shapes a molecule can take without breaking any bonds are called conformers. It's like a dancer doing different poses. Some poses are chill, and some are, well, a bit more… strained.
And that's where the energy comes in! Just like a stretched rubber band has more stored energy than a loose one, some molecular poses are just plain more energetic. They're less stable. They want to relax!
So, why is this a thing?
Because molecules aren't static blobs. They're constantly jiggling and wiggling. And depending on how they're arranged, they can bump into themselves, or their electron clouds can get all squished together. This squishing and bumping is what we call steric strain, and it's a major energy hog.
Imagine trying to shove two really big people into a tiny closet. It's gonna get crowded, right? Things will get awkward. That's steric strain in a nutshell, but for atoms! They don't like being too close for comfort, especially if they have bulky bits.
Another energy offender is torsional strain. This is like twisting something that doesn't want to be twisted. Think of a squeaky door hinge. You're putting in effort to turn it, and it's resisting. Certain bonds in molecules can get this kind of resistance too, especially when they're lined up in specific ways.
Let's Get Specific (But Not Too Specific!)
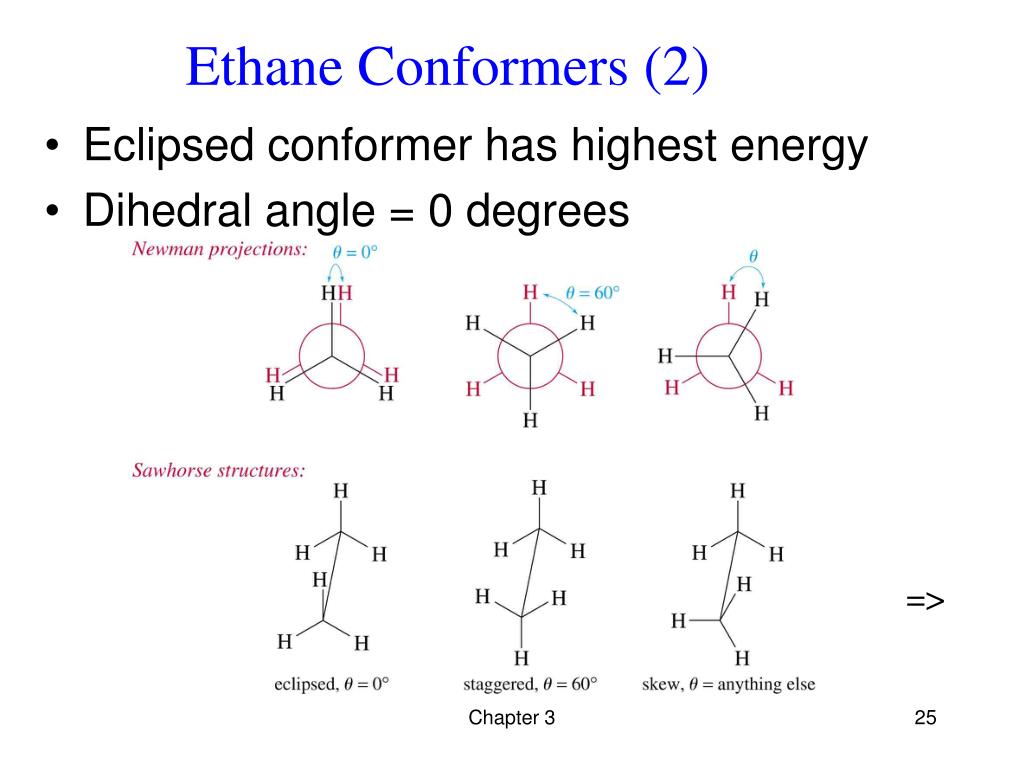
The classic example we usually talk about is a simple molecule like ethane. It's basically two carbon atoms with some hydrogens hanging off. You can imagine them spinning relative to each other. There are two main ways they can "sit":
1. Eclipsed Conformer: Picture this. You're looking down the bond connecting the two carbons. In the eclipsed form, the hydrogens on the front carbon are directly lined up with the hydrogens on the back carbon. It's like a perfectly organized, but super crowded, line-up. Everything's in the way!
2. Staggered Conformer: Now, imagine you twist those carbons a bit. The hydrogens on the front carbon are now between the hydrogens on the back carbon. It’s much more spread out. Plenty of personal space for everyone!
And the winner for highest energy is… the STAINED chair!
Okay, kidding! But seriously, in the eclipsed conformer of ethane, those hydrogens are practically giving each other nose-bleeds. They're bumping into each other's electron clouds, causing a whole lot of repulsion. This repulsion means higher energy. It's like trying to sleep in a crowded bunk bed where the person above you keeps kicking you in the face. Not ideal for a good night's rest (or molecular stability!).
The staggered conformer, on the other hand, is the chill one. The hydrogens are as far apart as they can get. Less bumping, less repulsion, less stress. That means lower energy. It's like having your own comfy bed with plenty of legroom. Ah, bliss!
But wait, there's more! The cyclohexane caper!
Now, if we're talking about rings, things get even more interesting. Cyclohexane is a six-membered ring, and it's famously flexible. It can twist into a bunch of shapes, but the two most important are:
1. Chair Conformer: This is the superstar of cyclohexane. It’s super stable, like a well-built armchair. All the angles are pretty happy, and the hydrogens have plenty of room. Low energy, folks!
2. Boat Conformer: Now, imagine you've bent the chair into a boat shape. It's not as comfy. There are these two hydrogens at the "bow" and "stern" that are getting a bit too close for comfort. They're called flagpole hydrogens, and they give each other the side-eye. This close proximity causes steric strain, making the boat conformer much higher in energy than the chair.
So, if you were given a choice between a chair and a boat for your molecular seating, you’d definitely pick the chair for maximum chill and minimum energy cost!
Why is this fun to talk about?
Because it’s like a secret language of the universe! We're peeking behind the curtain at how matter behaves. It’s the tiny details that make big things happen. Plus, thinking about molecules getting "strained" or having "awkward close encounters" is just inherently kind of funny, right?
It reminds us that even the most fundamental building blocks of reality have their preferences. They like their personal space. They don't like being twisted in ways they find uncomfortable.
The takeaway? It's all about STRESS!
When we talk about which conformer has the highest energy, we're basically asking: Which one is the most stressed out? Which one has the most things bumping into each other, the most uncomfortable angles, the most "I just want to relax!" vibe?
So, next time you’re looking at a molecule, imagine its little atoms doing a dance. Some are gracefully pirouetting, and others are tripping over themselves. The ones tripping? They're the high-energy champs, eager to find a more comfortable pose.
It's a little peek into the invisible world, a reminder that even the smallest things have their own drama. And honestly, who doesn't love a bit of molecular drama?
