Which Of The Following Compounds Is Are Chiral
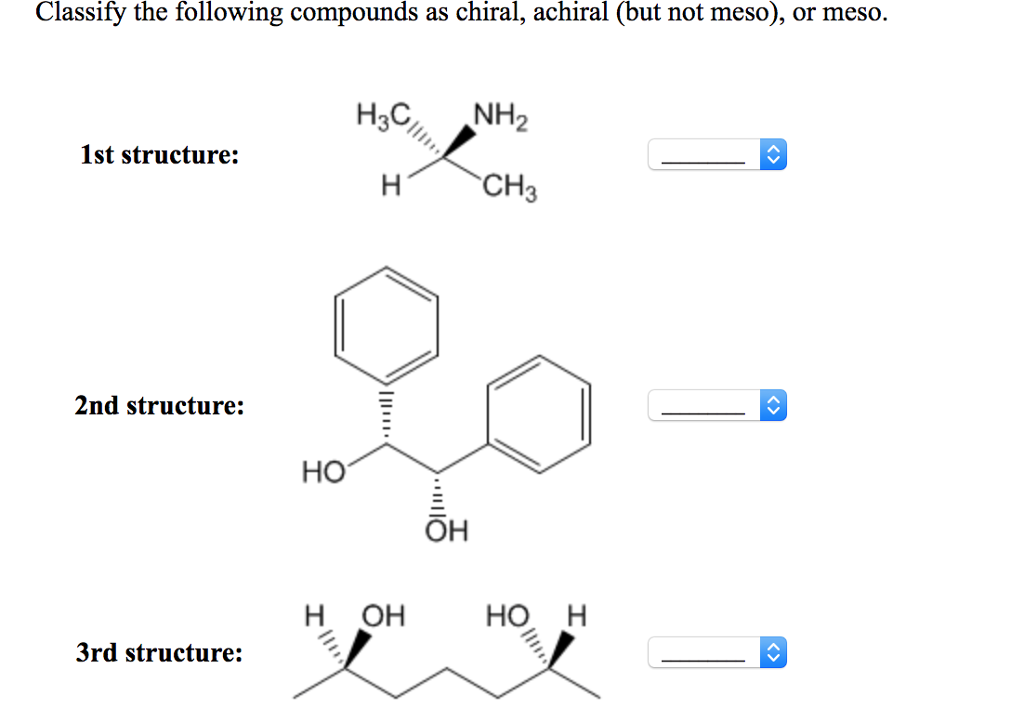
Hey there, science explorers and curious minds! Ever found yourself staring at a molecule and wondering, "Is this thing playing a trick on me?" Well, get ready to dive into the wonderfully weird world of chirality, because today we're tackling a question that might sound a little intimidating, but trust me, it's actually a blast! We're going to figure out: Which of the following compounds is/are chiral?
Now, before you start picturing tiny, spinning tops (though that's not entirely wrong, in a way!), let's break down what "chiral" even means. Think of your hands, right? You've got a left hand and a right hand. They look pretty darn similar, don't they? But no matter how you twist and turn them, you can't perfectly superimpose your left hand onto your right hand. One is always a mirror image of the other. That, my friends, is the essence of chirality! In the molecular world, a chiral molecule is one that is non-superimposable on its mirror image. It's like a left-handed glove and a right-handed glove – they're both gloves, but they're definitely not the same for your hand!
Why should you care about this? Oh, buckle up, because this is where things get really fun! Chirality is everywhere. It's in the air you breathe, the food you eat, and most importantly, it's in your very own body. Your DNA? Chiral. The amino acids that build your proteins? Chiral. Even the way your brain processes information can be influenced by chiral molecules. Isn't that mind-blowing? It means that a tiny difference in the "handedness" of a molecule can have huge implications.
Imagine a drug. If a drug is chiral, it can exist as two different mirror images, called enantiomers. Sometimes, one enantiomer is a life-saving medicine, while its mirror image is completely inactive, or worse, harmful! It's like having the right key to unlock a door, and then trying the exact opposite, mirrored key – it just won't work, right? This is why understanding chirality is absolutely crucial in fields like medicine, pharmaceuticals, and even food science.
So, how do we actually spot a chiral compound? The most common culprit, the easiest way to spot a potential chiral center, is to look for a carbon atom that is bonded to four different groups. Yep, that's it! Four different things attached to a single carbon. This is often called a "stereogenic center" or, more commonly, a "chiral center." If a molecule has one of these, there's a very high chance it's chiral. It's like a little molecular handshake: if that central carbon is holding hands with four different buddies, it's probably going to be a chiral molecule!
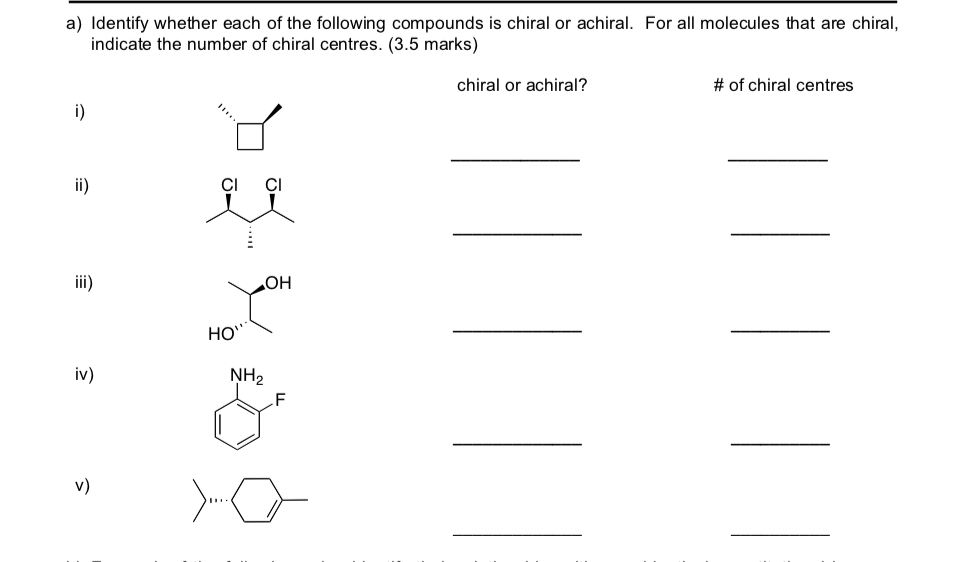
Let's get a little more specific. Imagine we have a hypothetical list of compounds (since the actual list wasn't provided, we'll create some fun examples!). Let's say we're looking at things like:
Compound A: Methane (CH4)
Methane. Simple, right? One carbon atom in the middle. What's it bonded to? Four hydrogen atoms. Are they different? Nope! They're all the same. So, methane is like a perfectly symmetrical little ball. No mirror image trickery here. Methane is achiral.

Compound B: Ethanol (CH3CH2OH)
Ethanol, the stuff in… well, let's just say some beverages. Let's look at the carbon atoms. The first carbon is bonded to three hydrogens and another carbon. Not four different groups. The second carbon is bonded to two hydrogens, an oxygen, and the first carbon. Still not four different groups. So, ethanol, as a whole, is looking pretty symmetrical too. Ethanol is achiral.
Compound C: 2-Chlorobutane (CH3CHClCH2CH3)
Alright, now we're getting somewhere! Let's zero in on that second carbon atom (the one with the chlorine attached). What's it bonded to?
- A methyl group (CH3)
- A chlorine atom (Cl)
- A hydrogen atom (H)
- An ethyl group (CH2CH3)
Compound D: Propane (CH3CH2CH3)
Propane, another simple alkane. Let's check the carbons. The end carbons are bonded to three hydrogens and a carbon. Not four different groups. The middle carbon is bonded to two hydrogens and two identical ethyl groups. Nope, still no four different groups. Propane is achiral.
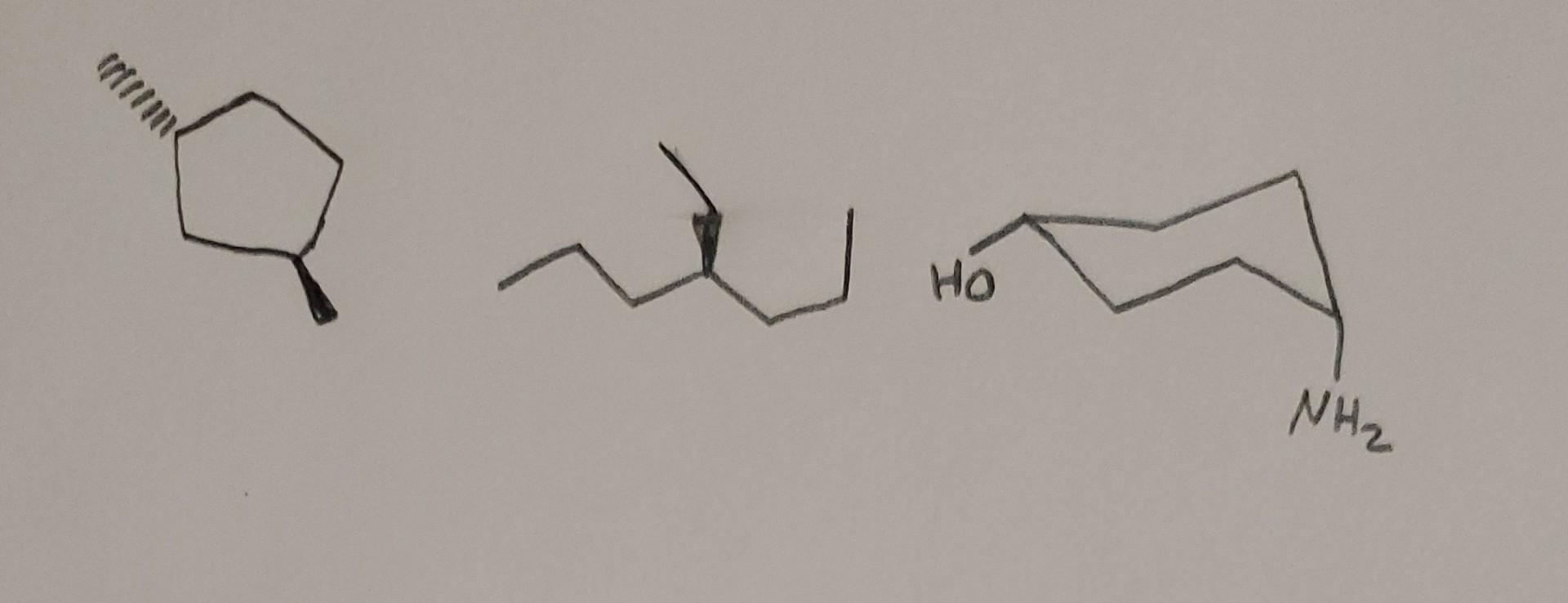
Compound E: Lactic Acid (CH3CH(OH)COOH)
Lactic acid! This one is super cool because it's found in your muscles after a good workout (ouch!) and also in yogurt. Let's look at the middle carbon. What's attached to it?
- A methyl group (CH3)
- A hydroxyl group (OH)
- A carboxyl group (COOH)
- A hydrogen atom (H)
So, to recap our little experiment, if we were given those compounds, the chiral ones would be the ones with a carbon atom bonded to four distinct groups. It’s a fantastic way to start seeing the hidden complexity and elegance in the molecular world.

Learning about chirality isn't just about memorizing rules; it's about developing a new way of seeing. It's like gaining molecular superpowers! You start noticing the subtle differences, the potential for distinct properties, and the incredible precision that nature uses. This understanding opens up a whole new dimension of appreciation for chemistry, biology, and the world around us.
Don't let the terminology scare you off. Every scientist started somewhere, and asking these kinds of questions is the first step to becoming a fantastic discoverer. The next time you encounter a chemical structure, remember our little trick: look for that carbon with four different friends. It's a fun challenge, and who knows what other molecular mysteries you'll unlock!
So, go forth and be curious! Dive deeper into the world of stereochemistry. It’s a journey filled with fascinating insights and a healthy dose of molecular magic. You’ve got this!
