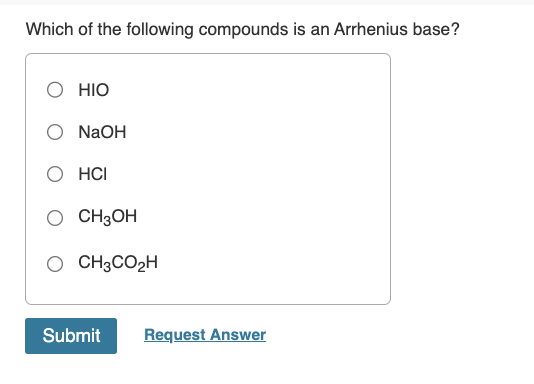
Which Of The Following Compounds Is An Arrhenius Base

Alright, let's chat about bases, specifically those good ol' Arrhenius bases. Now, I know what you're thinking: "Chemistry? Bases? Isn't that for lab coats and Bunsen burners?" And yeah, a little bit. But trust me, understanding what makes something an Arrhenius base is actually pretty darn close to stuff we deal with every single day. Think of it like figuring out why your favorite cookies are suddenly so chewy or why that stubborn coffee stain just won't budge. It's all about the tiny, invisible helpers doing their thing.
So, what in the world is an Arrhenius base? Basically, it's a compound that, when you toss it into water – our universal solvent, like the superhero of beverages – decides to go a little wild. It breaks apart and releases these things called hydroxide ions. You know, the chemical equivalent of that friend who always brings the extra napkins to a picnic? They're there to help soak things up and make the situation a bit more... well, basic.
Imagine you're making lemonade. You add lemon juice (which is acidic, the opposite of a base, like a sour patch kid) and sugar. Now, what if you wanted to make it a bit less tart? You might reach for something to balance it out. That's kind of what bases do. They bring that sweet, smooth vibe to things.
The "Arrhenius" part is just a fancy name for the dude who figured this out. Svante Arrhenius, a Swedish scientist who probably had a really neat beard, noticed that certain compounds acted in a specific way in water. He was like, "Aha! When these guys go into water, they churn out these OH- things (that's the hydroxide ion for you science nerds) and that's what makes them bases!" It’s like discovering your neighbor’s weird humming is actually a new, catchy tune. Revolutionary!
So, when we ask, "Which of the following compounds is an Arrhenius base?" we're essentially playing a game of chemical "spot the hydroxide producer." We're looking for a compound that has that specific knack for releasing OH- ions when dissolved in water. It’s not about them being "mean" or "grumpy" like some acids can be. Bases are generally more about being smooth operators, making things feel a bit more mellow.
Let's think about some real-world examples. You know those antacids you might grab when your stomach feels like a mosh pit after a spicy meal? Many of those are bases! They work by neutralizing that excess stomach acid. It's like a little peace treaty happening inside you. The acid is all aggressive and jiggly, and the base comes in and says, "Whoa there, buddy, let's calm down." And poof, relief!
Or consider soap. Ever wonder why soap cleans so well? A big part of it is that soap molecules often have basic properties. They can break down grease and grime. Think of it as the soap's way of saying, "This dirt? Not today, Satan!" It’s like a tiny, bubbly bouncer escorting the gunk out of your life. Without bases, our dishes would be perpetually greasy, and our clothes would look like they survived a paintball match.
Now, the question often comes with a list of compounds. Let's pretend we have a few options. Maybe we see something like HCl. That's hydrochloric acid. Sounds fancy, right? But it's a classic acid. It gives away a hydrogen ion (H+) when dissolved. It's the opposite of what we're looking for. It's like the one friend who takes your last fry instead of offering you one.
Then we might see NaCl. That's table salt. It's a pretty neutral character. It dissolves, sure, but it doesn't really make a big fuss about releasing H+ or OH-. It's like the dependable friend who just hangs out and doesn't cause any drama. Good for flavoring your popcorn, though!

But then, boom! We might spot something like NaOH. That's sodium hydroxide. This guy is practically screaming, "I'm a base!" When you put NaOH in water, it splits up into a sodium ion (Na+) and, ding ding ding, a hydroxide ion (OH-). Bingo! This is our guy. This is the Arrhenius base we're hunting for. It’s like finding a twenty-dollar bill in an old coat pocket.
Another common Arrhenius base you might encounter is KOH, potassium hydroxide. It's very similar to NaOH. It's like the slightly fancier, more exotic cousin. Toss it in water, and it’ll happily churn out potassium ions (K+) and those all-important hydroxide ions (OH-). These guys are the workhorses of the base world, used in everything from making batteries to refining oil. They're the unsung heroes, quietly making modern life possible.
Think about baking soda, too. That fluffy white powder you use to make cakes rise? That's sodium bicarbonate ($NaHCO_3$). While it's a bit more complex than a simple Arrhenius base, in water, it can indirectly lead to the formation of hydroxide ions. It’s the little helper that makes your muffins fluffy and your bread light. It’s like the secret ingredient to a good mood and a good bake.
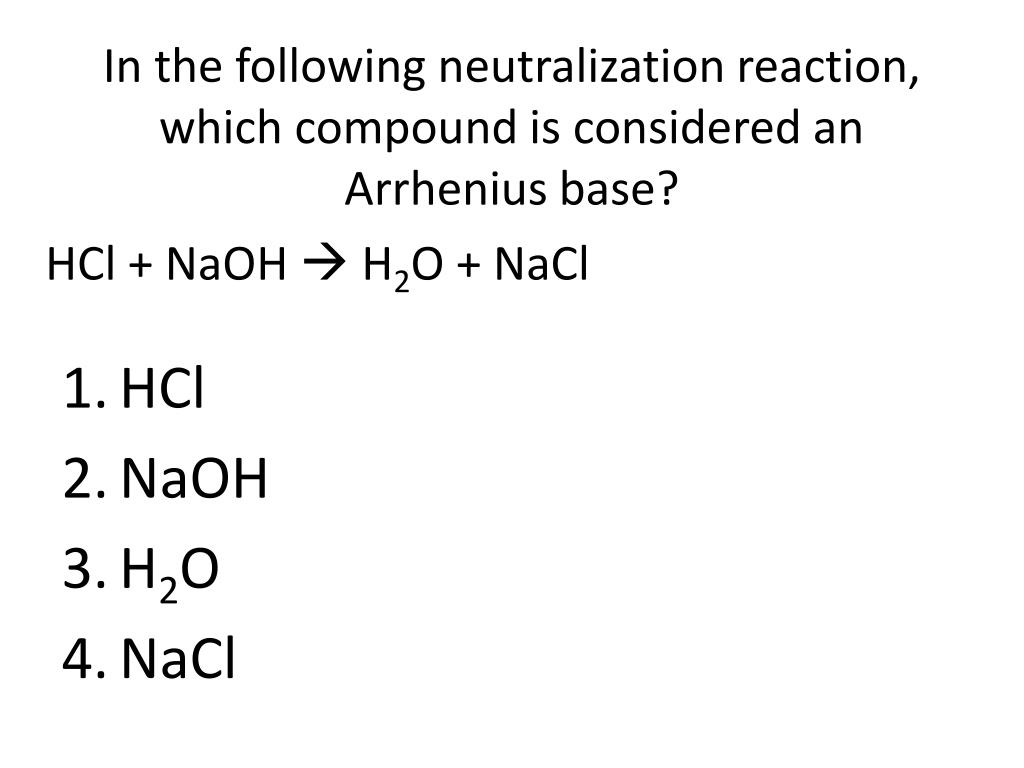
So, how do we identify these Arrhenius bases in a lineup? We're primarily looking for compounds that contain a metal cation (like sodium, potassium, or calcium) bonded to a hydroxide group ($OH^-$). When these ionic compounds hit the water, the bond between the metal and the hydroxide breaks, and voilà – free-floating hydroxide ions!
Consider Ca(OH)₂, calcium hydroxide. This one is a bit of a power player. When it dissolves, it actually releases two hydroxide ions for every calcium ion ($Ca^{2+}$). This makes it a pretty strong base. Think of it like a double-fisted high-five of basic-ness. It's used in things like cement and is even found in some over-the-counter remedies for heartburn. It’s got the muscle to tackle tough jobs, both in industry and in your digestive tract.
Now, it's important to remember that not all compounds containing OH are Arrhenius bases. Water itself has OH groups, but it's sort of a special case. It can act as both an acid and a base (a phenomenon called amphoterism, which is a whole other kettle of fish). Also, organic compounds like alcohols ($R-OH$) have OH groups, but they behave very differently and don't typically release hydroxide ions in the Arrhenius sense. They’re more like cousins who show up to the family reunion but don’t quite fit the mold. They have the right letters, but the wrong personality for the job.

The key takeaway is the release of hydroxide ions in water. This is the defining characteristic of an Arrhenius base. It's like recognizing a duck by its quack. If it quacks like a duck and swims like a duck, it's probably a duck. If it dissolves in water and spits out hydroxide ions, it's probably an Arrhenius base.
So, when you're faced with a question like "Which of the following compounds is an Arrhenius base?", don't panic. Just channel your inner chemist detective. Look for those compounds that have that specific talent for generating hydroxide ions in water. Think about NaOH, KOH, Ca(OH)₂, and similar metal hydroxides. These are your suspects, the ones most likely to be the Arrhenius base.
It’s a simple concept, really, once you strip away the jargon. It's about understanding what makes things smooth, what helps neutralize sharp edges, and what’s behind that squeaky-clean feeling. So next time you’re using soap, taking an antacid, or even just admiring a well-risen cake, give a little nod to the Arrhenius bases. They’re the quiet, often unsung, heroes of our everyday chemical world, making things a little bit easier, a little bit cleaner, and a whole lot more palatable. They’re the backstage crew that makes the show run smoothly, and for that, we can all be grateful.
