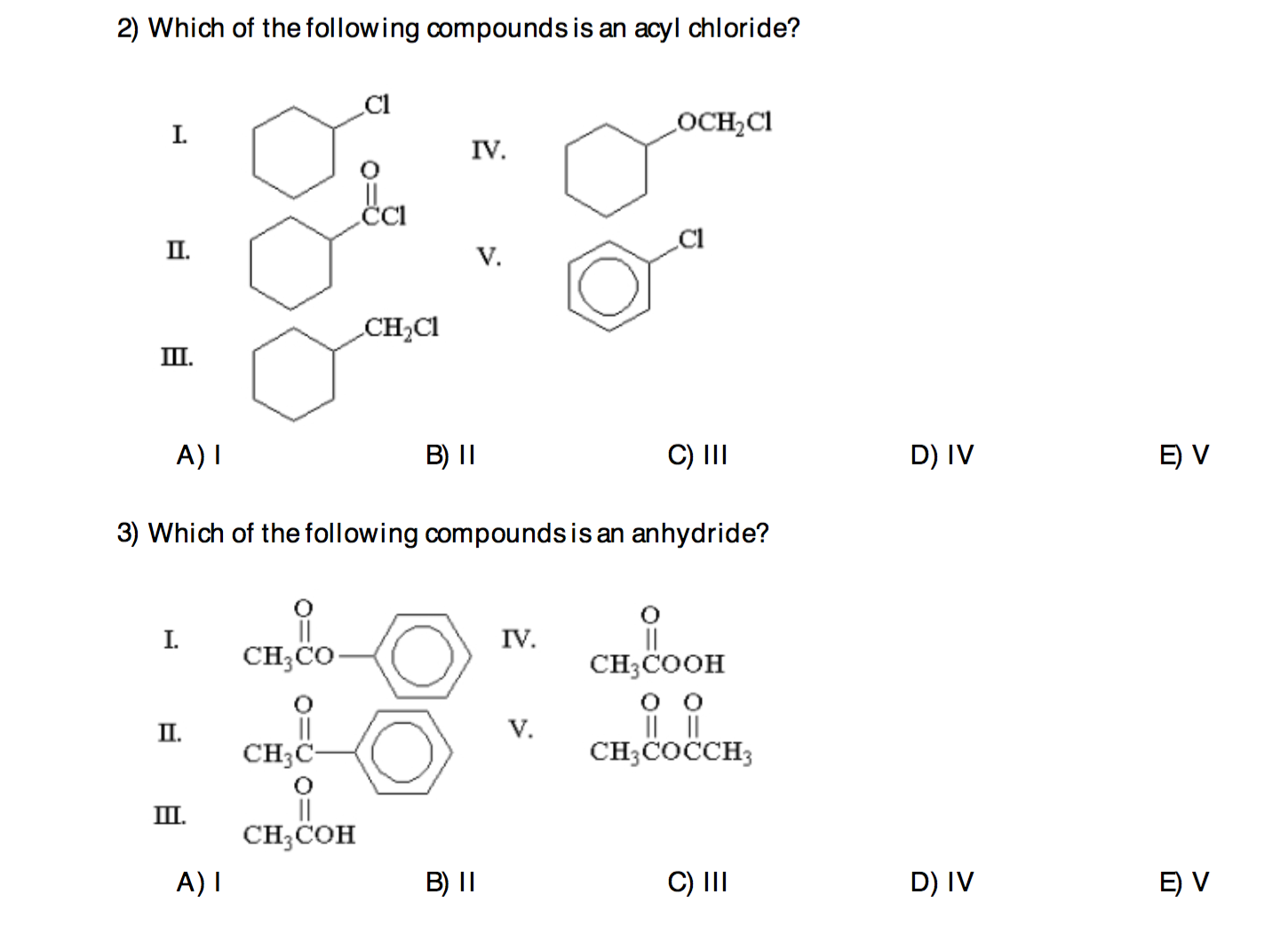
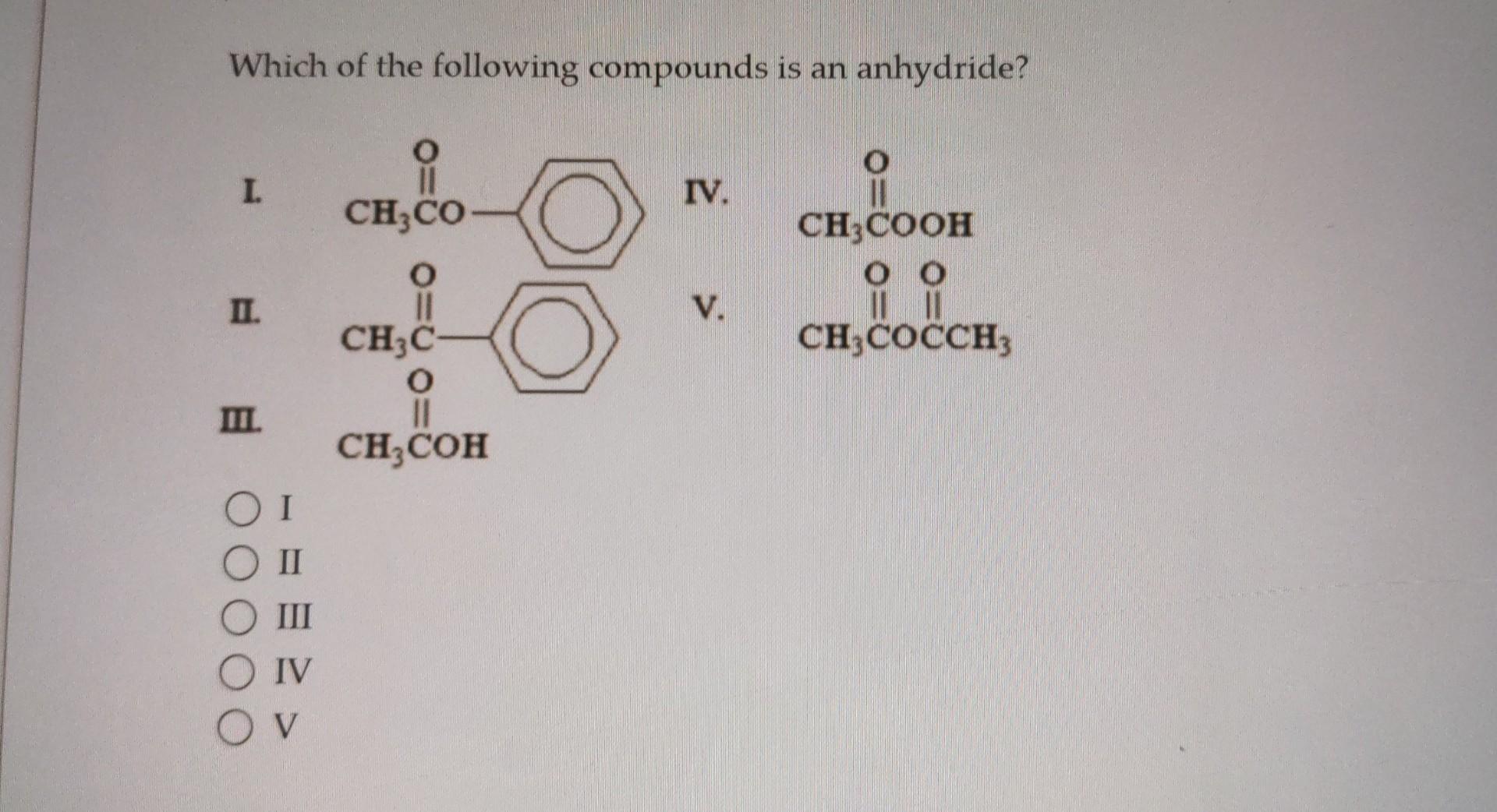
Which Of The Following Compounds Is An Anhydride
Hey there, curious minds! Ever stared at a chemistry textbook, or maybe even a quirky science documentary, and felt a little… well, bewildered by all the fancy chemical names? Don't worry, you're not alone! Today, we're diving into a topic that might sound a tad technical at first, but trust me, it’s a little secret that can make understanding the world around you just a little bit more sparkly. We're talking about anhydrides! Yes, I know, it sounds like something from a sci-fi movie, but stick with me, because this is actually pretty neat.
So, the big question we're tackling, with a smile and a metaphorical beaker of enthusiasm, is: Which of the following compounds is an anhydride? Now, I'm not going to bombard you with a list of confusing chemical formulas right off the bat. That would be downright rude, wouldn't it? Instead, let's break down what an anhydride even is in the first place. Think of it like this: the word "anhydride" comes from Greek words meaning "without water." Pretty straightforward, right? These are compounds that have essentially lost a molecule of water during their formation.
Unlocking the Mystery: What's So "Anhydrous" About Them?
Imagine you have a parent compound, say, an acid. And then, poof! Through some clever chemical magic, it sheds a water molecule. What's left behind? You guessed it: an anhydride! They're like the leftover, super-concentrated versions of their watery cousins. This "loss of water" is a key concept, and it’s the fundamental characteristic that defines an anhydride.
Why does this even matter? Well, these little water-shedders have some pretty cool properties. They're often highly reactive, which means they can be super useful in making all sorts of other, more complex molecules. Think of them as the essential building blocks for so many things we use and see every day. From the plastics in your phone to the medicines that keep you healthy, there's a good chance an anhydride played a starring role somewhere in their creation!
A Little Chemical Detective Work
So, when we’re faced with a question like "Which of the following compounds is an anhydride?", we're essentially becoming chemical detectives. Our mission, should we choose to accept it (and you should, it's fun!), is to identify the compound that fits the "water-shedding" description. This usually involves looking at the chemical formula and seeing if it structurally resembles a molecule that could have been formed by removing H₂O from a related acid.

It's a bit like a puzzle. You're given a few pieces, and you have to figure out which one is the missing link. Is it the one that clearly looks like it's "missing" a water molecule compared to its acidic counterpart? That's your prime suspect!
Spotting the "Water-Haters"
Let's get a little more concrete, but still keeping it light. Take a look at common examples. You might see things like acetic anhydride. If you know about acetic acid (the stuff that gives vinegar its tang!), you can see how acetic anhydride is essentially two molecules of acetic acid that have hooked up and kicked out a water molecule between them. Pretty clever, right? It’s like they decided to travel light!
Or consider sulfuric anhydride (also known as sulfur trioxide). This one is formed when sulfuric acid loses a water molecule. The resulting compound, SO₃, is a powerful substance that’s crucial in the production of… well, more sulfuric acid, and a whole bunch of other industrial chemicals. It’s a bit of a chemical ouroboros, isn't it?

The key is to recognize the pattern: an anhydride often looks like a central atom (like carbon or sulfur) bonded to oxygen atoms, and then to organic groups or other atoms in a way that suggests a water molecule has been removed from a parent acid. It’s a bit like seeing two puzzle pieces that clearly should fit together, with a tiny gap where a third piece (water) used to be.
Why This Stuff Makes Life More Fun (Seriously!)
Okay, I can hear you thinking, "How does identifying an anhydride make my life more fun?" Well, think about it! The more you understand how things are made, the more you appreciate the incredible complexity and ingenuity of the world around you. It’s like suddenly seeing the hidden gears and levers that make everything work!

When you can look at a chemical name or formula and go, "Ah, I see! That's an anhydride! It's missing water and probably means it's going to be a real workhorse in making something useful!" – that's a moment of genuine intellectual satisfaction. It’s like unlocking a secret code!
And it’s not just about textbooks. This knowledge can actually pop up in unexpected places. Maybe you're reading about new materials being developed, or learning about how certain foods are processed, or even understanding the chemistry behind why certain cleaning products work. Having this little bit of chemical insight can make those discoveries so much more engaging and, dare I say, exciting!
Plus, it’s a fantastic conversation starter. Imagine casually dropping into a chat, "Oh, you know, the reason those polymers are so strong is often thanks to the reactivity of anhydrides!" You’ll sound like a bona fide science whiz, and who doesn't love a little bit of that?
The Joy of Discovery Awaits!
So, the next time you encounter a question about anhydrides, don't let it intimidate you. See it as an opportunity to be a curious explorer. You’re not just answering a question; you’re peeking behind the curtain of how our world is put together.
The world of chemistry is vast and wonderful, filled with countless fascinating compounds and reactions. And understanding concepts like anhydrides is just the first step in a journey of continuous discovery. Each new chemical term you learn, each new reaction you grasp, is like adding another vibrant color to your understanding of the universe.
Don't be afraid to ask questions, to seek out more information, and to marvel at the intricate dance of atoms and molecules. You've got the curiosity, and the universe has all the answers waiting to be uncovered. So, go forth, embrace the learning, and let your scientific spirit soar! Who knows what amazing connections you'll make next?
