Which Of The Following Compounds Is An Aldehyde
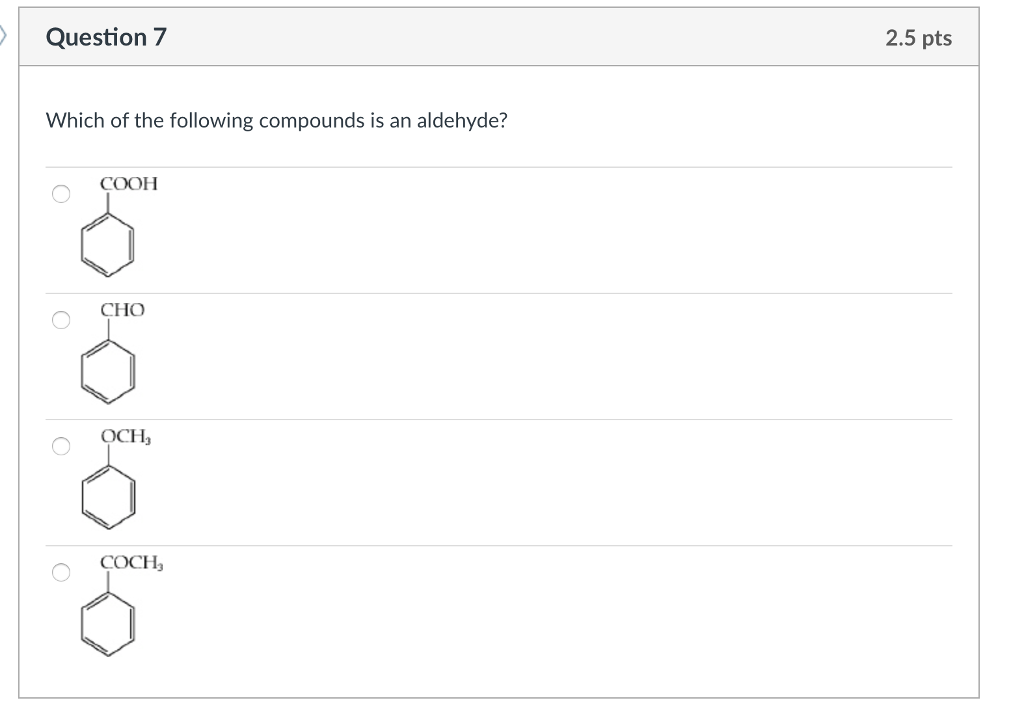
Okay, so imagine we’re just chilling, right? Coffee's brewed, maybe there’s a sneaky biscuit involved… don’t judge! And we’re diving into the wild, wonderful world of chemistry. Specifically, we’re playing a little game of "Which one's the aldehyde?" It’s like a fun little quiz, but instead of pop stars, we're looking at molecules. Exciting stuff, I know.
So, the question is, out of a bunch of options (which we’ll get to, don’t worry!), which one is an aldehyde? Sounds simple enough, doesn't it? But you know how it is with chemistry – the devil is in the details, or in this case, the functional group. That’s the special bit of the molecule that gives it its personality, its quirks, its entire vibe.
Think of it like this: you’ve got a bunch of people at a party. Some are super loud, some are quiet readers, some are just there for the free snacks. The functional group is like their defining characteristic. For aldehydes, this characteristic is pretty darn neat.
What is an aldehyde, then? It’s a molecule that has a specific structure. It's got a carbon atom that's double-bonded to an oxygen atom, and also single-bonded to a hydrogen atom. And that whole party is happening at the end of a chain. Like, right at the very tip-top. It’s a pretty distinctive look.
So, you’ve got this C=O group, right? That’s called a carbonyl group. It’s pretty common, found in lots of different types of molecules. But for an aldehyde, this carbonyl group has to be hanging off the end. And to one side of that carbonyl carbon, you have to have a hydrogen. That’s the key. The other side? It can be another hydrogen (which makes it the simplest aldehyde, formaldehyde – yikes, right?), or it can be a carbon chain.
Why is this so important? Because it dictates how the molecule behaves. Aldehydes are super reactive. They’re like the energetic toddler of the organic chemistry world, always ready to react with something. They love to get involved in all sorts of chemical shindigs.
Let’s break down what makes an aldehyde an aldehyde. You absolutely need that carbonyl group (C=O). No ifs, ands, or buts. But here’s the kicker, the bit that separates the aldehydes from their cousins, the ketones. For a molecule to be an aldehyde, that carbonyl carbon must be attached to at least one hydrogen atom. And where is this carbonyl carbon? It’s always at the end of a carbon chain.

So, picture a string of beads, representing carbons. At one end of that string, you’ve got a special little bead – the carbonyl carbon. And that special bead is holding hands with an oxygen (a double high-five, really) and also giving a little wave to a hydrogen. The other side of the carbonyl carbon is then attached to either another hydrogen or the rest of the bead string.
This is what makes them so special. They're the "terminal" reactive groups. Think of it as the VIP section of the molecule. That hydrogen on the carbonyl carbon is super important for their reactivity. It's like the "get out of jail free" card that allows them to participate in so many cool reactions.
Now, let’s contrast this with a closely related group: ketones. Ketones also have that carbonyl group (C=O). They’re like the aldehydes’ cooler, more laid-back cousins. But here's the crucial difference: in a ketone, the carbonyl carbon is sandwiched between two other carbon atoms. No hydrogen atoms directly attached to that carbonyl carbon. None. Zero. Nada.
So, if you see a molecule with a C=O, but the carbon in that C=O is not at the end of the chain, and it's surrounded by other carbons, poof! That’s a ketone. Not an aldehyde. Close, but no cigar. It’s like mistaking a celebrity look-alike for the real deal. They might look similar, but they’re not the same.

Why does this even matter in the grand scheme of things? Well, aldehydes and ketones have different properties and react in different ways. Aldehydes, because of that handy little hydrogen, are excellent reducing agents. That means they can donate electrons in a reaction, which is pretty darn useful. They’re also often found in nature, giving things their smells. Think of the lovely scent of vanilla – that’s vanillin, an aldehyde! Or the almondy smell of marzipan – benzaldehyde! See? They’re not just boring molecules; they’re the reason your world smells good.
So, when we look at our mystery compounds, we're basically playing detective. We’re going to scrutinize each one, looking for that tell-tale sign: a carbonyl group at the end of a chain, with a hydrogen attached. No exceptions.
Let’s imagine some suspects.
Suspect Number One: Ethanol
Okay, first up, we’ve got ethanol. You might know it as the stuff in your drink. It’s an alcohol. Its structure is CH3-CH2-OH. See that -OH group? That’s a hydroxyl group. It’s the signature of an alcohol. No carbonyl group here, folks. So, ethanol is a definite no-go for our aldehyde hunt. It's not even in the same league.
Suspect Number Two: Acetic Acid
Next, we have acetic acid. You know, the stuff that makes vinegar sting. Its structure is CH3-COOH. Now, this one’s a bit trickier. See that -COOH? That looks a little bit like a carbonyl. And it is! It has a carbonyl group, yes. But it also has a hydroxyl group attached to the same carbon. This combination is called a carboxyl group, and it’s the defining feature of a carboxylic acid. So, acetic acid is also out. It’s like mistaking a fancy hat for a crown. Close, but definitely not the same regal status.
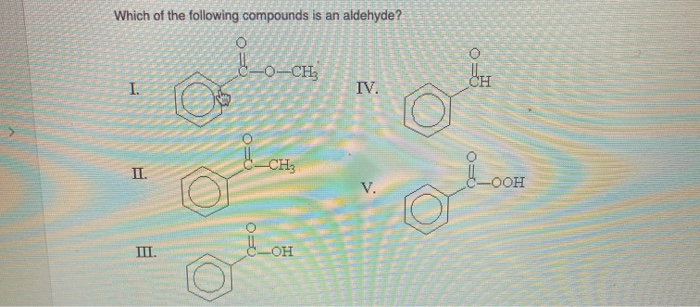
Suspect Number Three: Acetone
Alright, our third suspect is acetone. This is a common solvent, and if you’ve ever used nail polish remover, you’ve met acetone. Its structure is CH3-CO-CH3. Now, this one has a C=O group, doesn't it? Yes, it does! But where is that carbonyl carbon? Is it at the end of a chain? Nope. It’s in the middle, sandwiched between two methyl groups (CH3). This, my friends, is the hallmark of a ketone. So, acetone is not an aldehyde. It’s a ketone, its stylish cousin, but not our target.
Suspect Number Four: Formaldehyde
And then there’s formaldehyde. Its structure is HCHO. Or, if we want to be super clear about the bonding, H-CO-H. Look at that! We’ve got a carbonyl group (C=O). And what's attached to the carbonyl carbon? Two hydrogen atoms! And that carbonyl carbon is at the end of the… well, the shortest possible chain. This is the absolute simplest aldehyde. It fits all the criteria. Formaldehyde is an aldehyde. Bingo!
Suspect Number Five: Propanal
Let’s throw in another one, just for kicks. How about propanal? Its structure is CH3-CH2-CHO. Let’s break that down. We've got a CHO group at the end. What does that mean? It means a carbonyl carbon (C=O) with a hydrogen attached. And what’s on the other side of that carbonyl carbon? A -CH2-CH3 group, which is a carbon chain. So, we have a carbonyl group at the end of a chain, with a hydrogen attached. Propanal is definitely an aldehyde. Absolutely!
Suspect Number Six: Butanone
One more for good measure. Butanone. Its structure is CH3-CO-CH2-CH3. Again, we see that C=O group. But is it at the end? No! It’s smack-dab in the middle, between a methyl group and an ethyl group. Yep, you guessed it – butanone is a ketone. So, not an aldehyde.
So, when you’re faced with a question like "Which of the following compounds is an aldehyde?", your mission, should you choose to accept it, is to look for that specific arrangement. That lonely carbonyl carbon at the end of a carbon chain, cozied up with a hydrogen atom. It’s the ultimate identifier.
It’s not just about memorizing formulas; it’s about understanding the architecture of these molecules. The functional groups are like the building blocks, and knowing what they look like and where they hang out is the key to unlocking all sorts of chemical mysteries.
Think of it like being a birdwatcher. You don't just see a bird; you see its beak shape, its feather pattern, its call. In chemistry, you don't just see a molecule; you see its functional groups, its bond types, its structure. And that’s how you identify it.
So, to sum it up, you’re on the hunt for: 1. A carbonyl group (C=O). 2. That carbonyl carbon being attached to at least one hydrogen atom. 3. That carbonyl carbon being located at the end of a carbon chain.
If all those boxes are ticked, congratulations! You've found yourself an aldehyde. It’s like finding a rare gem. And trust me, in the world of chemistry, finding an aldehyde is a pretty satisfying discovery. They’re the workhorses, the smell-makers, the reactive powerhouses. So, next time you’re looking at a chemical structure, you’ll know exactly what to search for. Happy hunting!
