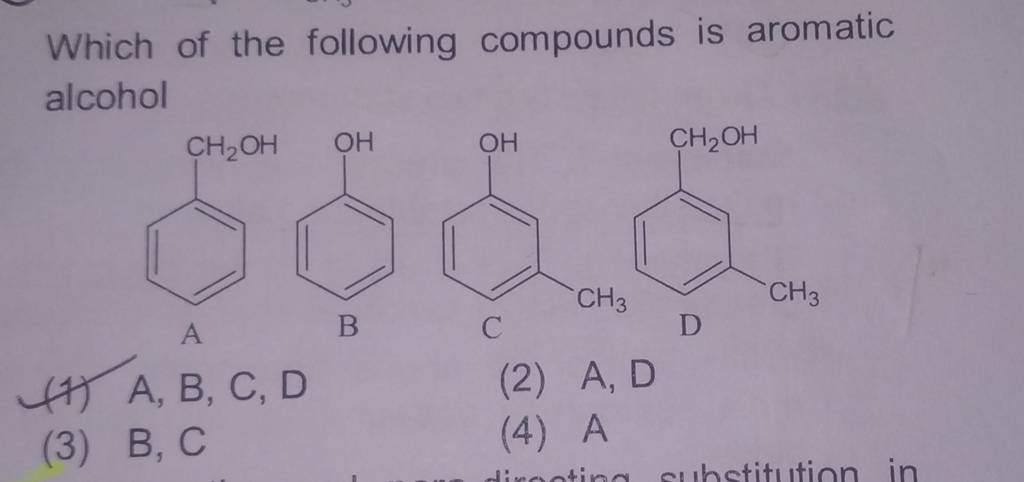
Which Of The Following Compounds Is An Alcohol

Ever found yourself staring at a grocery list, a recipe, or even just a weird-looking bottle at a friend's house, and a little voice in your head asks, "Wait a minute, is that an alcohol?" Yeah, me too. It's like a tiny chemistry pop quiz that pops up when you least expect it, usually right when you're about to whip up that amazing apple crumble or, you know, just trying to figure out what’s in that fancy cleaning spray your aunt swears by.
We hear the word "alcohol" and our brains immediately conjure up images. Maybe it's a celebratory toast with champagne, the soothing warmth of a whisky on a chilly evening, or perhaps the unmistakable scent of rubbing alcohol when you've had a little run-in with a rogue rose bush. But the truth is, "alcohol" in chemistry class is a much, much bigger club than just the stuff you'd find at the liquor store.
Think of it like this: "alcohol" is the family name, and the stuff we drink is just one very popular, very famous branch of that family tree. There are tons of other relatives, some you know and love, and some you probably encounter every single day without even realizing they're part of the "alcohol" crew. It’s like finding out your quiet, bookish cousin you only see at Thanksgiving is secretly a rock star in their free time. Mind. Blown.
So, how do we figure out who's who in this chemical family reunion? It all comes down to a little bit of molecular structure. Don't worry, we're not going to get bogged down in a lecture hall with chalk dust flying. We're going to keep it light, breezy, and relatable. We're talking about the magic ingredient, the signature move that screams "I'm an alcohol!"
The key player in the alcohol club is a little functional group called the hydroxyl group. Fancy name, right? But all it really means is an oxygen atom (O) linked to a hydrogen atom (H). Think of it like a tiny, eager handshake happening between an oxygen and a hydrogen. This O-H little duo then tethers itself to the rest of the molecule, like a little flag waving from a flagpole. And that, my friends, is the hallmark of an alcohol.
So, when you see an organic compound – that’s just chemistry talk for a carbon-based molecule, the building blocks of life and pretty much everything else – and it’s got this O-H group hanging off it, you can pretty much give it a nod and say, "Yep, you're an alcohol." It’s like spotting a specific type of hat that everyone in a particular club wears. You see the hat, you know the club.

Let's dive into some examples, because that's where the fun really starts. Imagine you’re looking at a compound. What’s your first move? You scan it for that tell-tale hydroxyl group. It's like being a detective, and the O-H is your smoking gun.
Consider ethanol. This is the big cheese of the drinking world. You find it in wine, beer, spirits – the whole shebang. Its chemical formula is often written as C2H5OH. See that "OH" at the end? That’s our O-H handshake, our little hydroxyl flag. Ethanol is a classic alcohol, no doubt about it. It’s the life of the party, the one everyone recognizes.
But what about other things we use every day? Take rubbing alcohol. You know, the stuff you dab on a scraped knee or use to disinfect surfaces. The most common type is isopropanol, or isopropyl alcohol. Its formula is C3H7OH. Again, there’s that familiar "OH" group, firmly attached. So, rubbing alcohol? Definitely an alcohol. It's the sensible, hardworking cousin of ethanol, always ready to clean up a mess.
Now, things get a bit more interesting. Let’s talk about glycerol, also known as glycerin. You find this in lotions, soaps, and even some foods as a sweetener or humectant. Its formula is C3H8O3, which can be written as C3H5(OH)3. Notice the three "OH" groups in there? This makes glycerol a polyol, meaning it has multiple hydroxyl groups. It's like the aunt who’s always invited to every family gathering because she’s so versatile and brings a bit of everything. Glycerol is an alcohol, a very useful one at that, and it's not typically something you'd get tipsy on.
What about something like sugar? You might be thinking, "Sugar? Is that even on the radar?" Well, some sugars, like glucose, are indeed classified as carbohydrates, but they also contain hydroxyl groups. Glucose has the formula C6H12O6, and it has several -OH groups attached to its carbon chain. So, technically, in a broad sense, it’s got alcohol characteristics. It's like finding out your favorite candy bar has a secret ingredient that’s also in your disinfectant. Weird, right? But that's chemistry for you – it's all about the building blocks.
Here's a little trick: if you see a compound ending in "-ol" in its name, there's a pretty good chance it's an alcohol. Ethanol, methanol, propanol, isopropanol, butanol – the "-ol" is a classic sign. It’s like seeing a surname that’s instantly recognizable. Of course, there are exceptions, because the universe loves to keep us on our toes, but it's a great starting point.
Methanol is another alcohol, C H3OH. This one is also known as wood alcohol. It’s quite toxic if ingested, so definitely not for sipping. Think of it as the distant, slightly dangerous relative you hear about but never actually meet. It’s an alcohol, but not one you’d invite to your dinner party.
So, the next time you’re trying to decipher a chemical name or just curious about what’s what, remember the hydroxyl group. That little O-H handshake is your VIP pass to identifying an alcohol. It’s the secret handshake of the molecular world.
Let's consider a few things that are not alcohols, just to make sure we’ve got the concept locked down. Take acetic acid, the main component that gives vinegar its zing. Its formula is C2H4O2, or CH3COOH. See that "COOH" at the end? That's a carboxyl group, not a hydroxyl group. It’s like seeing a different type of hat, one that signifies a different club altogether. Acetic acid is an acid, and while it’s related to alcohols, it’s not one itself. It's the grumpy neighbor who lives next door to the cheerful alcohol family.
Or how about ethers? Compounds like diethyl ether, which used to be used as an anesthetic. Its formula is C4H10O, and it's structured like R-O-R (where R represents carbon chains). There's an oxygen atom, but it's sandwiched between two carbon atoms, not attached to a hydrogen. It's like a different kind of handshake, where the oxygen is holding hands with two different carbon friends instead of one hydrogen. Ethers are their own distinct group, not alcohols.
It's fascinating how a tiny difference in arrangement can completely change a compound's properties and its family affiliation. It's like one misplaced comma in a recipe that turns your delicious cake into a savory bread. The ingredients are similar, but the outcome is entirely different.

So, if you’re presented with a list of compounds and asked to identify the alcohol, your strategy is simple: look for the -OH. Does the molecule have a carbon atom bonded to an oxygen atom, which is then bonded to a hydrogen atom? If the answer is a resounding "yes," then congratulations, you've found yourself an alcohol! It’s like a treasure hunt, and the O-H is your X marks the spot.
It’s not just about the stuff we drink. Alcohols are everywhere. They’re in solvents, fuels, plastics, pharmaceuticals, and even the building blocks of our DNA. They are incredibly versatile and essential to modern life. Think of them as the Swiss Army knives of the chemical world – you can do so much with them.
The next time you’re looking at a bottle of something, whether it’s your hand sanitizer, a bottle of vanilla extract (which contains ethanol!), or even just reading an ingredient list, take a moment to look for that tell-tale hydroxyl group. You might be surprised at how many of your everyday items have a connection to the fascinating world of alcohols. It’s a little secret of the universe, revealed by a simple O-H bond. And honestly, isn’t that just the coolest?
So, to recap, when you're trying to spot an alcohol in the wild, remember the hydroxyl group. It's the key. It's the signature. It's the little O-H handshake that makes an alcohol an alcohol. And with this knowledge, you can navigate the world of chemistry with a little more confidence and a lot more smiles. Happy identifying!
