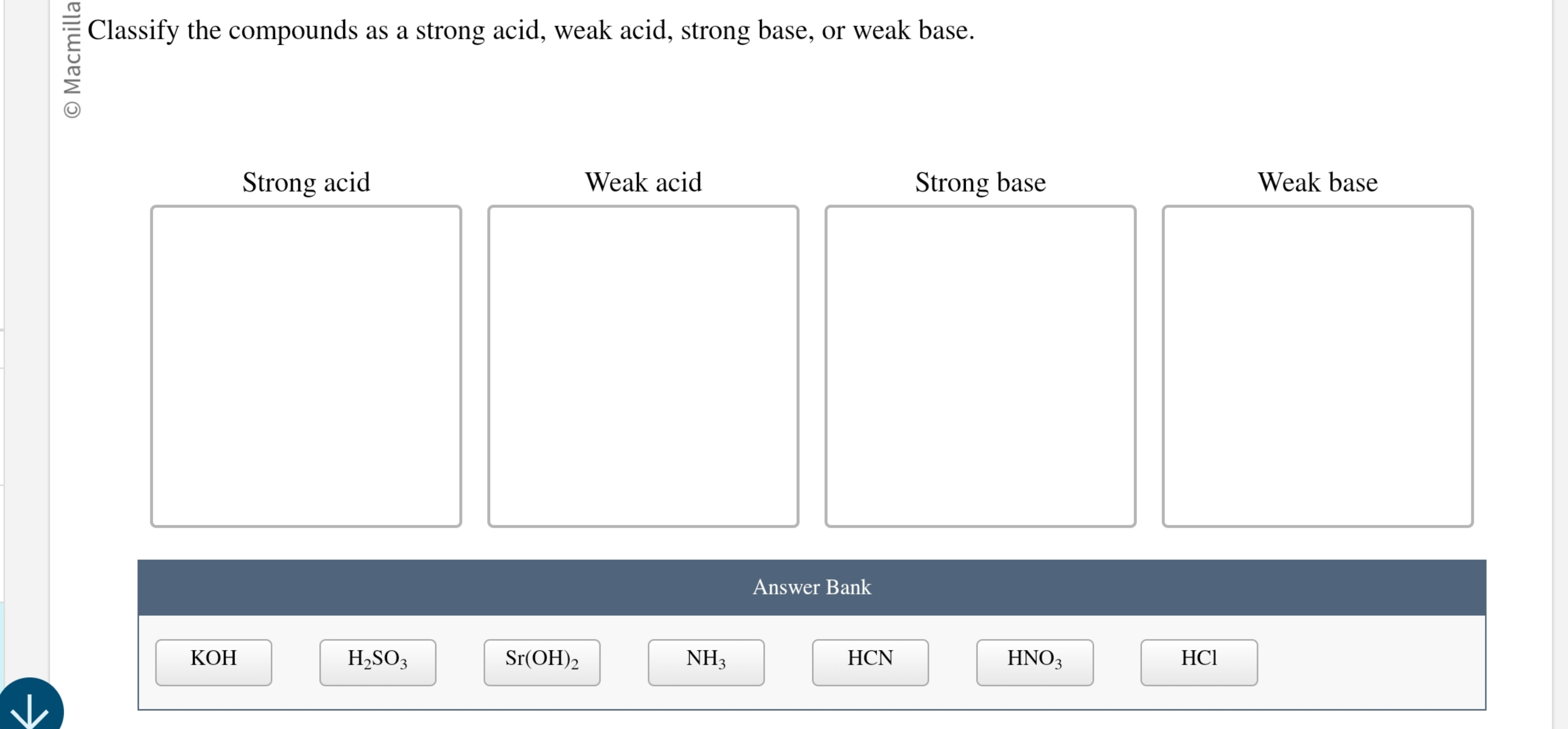
Which Of The Following Compounds Is A Weak Acid
Hey there, science curious friends! Ever wondered why some things make your mouth pucker up like a prune, while others are just… chill? Today, we’re diving into the wonderfully mild world of weak acids. No need to grab your lab coat or remember any complicated formulas. Think of this as a friendly chat over a cup of tea, exploring why these "gentle giants" of the chemical world are actually super important in our everyday lives.
So, what’s the big deal with acids, anyway? Well, the word "acid" can sound a bit scary, right? Like something that’ll eat through your clothes or make your eyes water uncontrollably. And some acids are like that – we call them strong acids. Imagine a really, really sour lemon. That’s a strong acid at play! It’s got a real oomph when it comes to donating its special "hydrogen ions" (don't worry too much about the fancy name, just think of them as little tiny guys that make things acidic).
But then, there are the weak acids. These are the chill cousins. They’re still acids, they still have those little hydrogen ion guys, but they’re not in a huge hurry to let them go. They’re more like, "Okay, I'll share a few, but I'm keeping most of them close." This makes them much kinder and less reactive.
Let's think about a common scenario. You're making a vinaigrette for your salad, and you add some vinegar. That tang? That’s the acetic acid in the vinegar doing its thing. Is it going to melt your spoon? Nope! It’s a weak acid. It gives your salad that delightful zing without causing any chemical chaos. Imagine if you used a strong acid to dress your lettuce! You'd be left with… well, not much of a salad. Acetic acid is a perfect example of a weak acid being our friend in the kitchen.
Another way to think about it is like a shy toddler. A strong acid is like a toddler who immediately runs up to everyone and demands hugs. A weak acid is like a toddler who peeks out from behind their parent’s legs and only offers a hesitant wave. They’re still interacting, but on their own terms and with less intensity. This gentle nature is precisely why they’re so valuable.

Why should we even care about these mild-mannered molecules? Because they are everywhere! They’re in the food we eat, the drinks we enjoy, and even in our own bodies. Understanding them helps us appreciate the science behind so many things we take for granted. It's like knowing how your favorite recipe works – it makes the final dish even more satisfying!
Let’s talk about another familiar weak acid: citric acid. You find this little gem in all sorts of fruits, especially citrus ones like oranges, lemons, and grapefruits. That refreshing tartness you get from a glass of orange juice? Yep, citric acid again! It’s what gives those fruits their bright, zesty flavor profile. Without it, our fruits would be a lot more… bland. It’s like a flavor enhancer, a natural one at that!
Think about it: if citric acid were a strong acid, your morning OJ would be a dangerous concoction! But because it’s a weak acid, it’s perfectly safe and delicious to consume. It’s responsible for that pleasant tingle on your tongue, not a burning sensation.

So, when we ask, "Which of the following compounds is a weak acid?", we're not just playing a guessing game. We're trying to identify those substances that are mildly acidic, that don't fully break apart into their components when dissolved in water. They’re like a team that mostly sticks together, only sending out a few players at a time.
Consider the context of biological systems. Our bodies are incredibly sophisticated chemical factories, and weak acids play crucial roles. For instance, lactic acid is produced when we exercise. It’s what can make our muscles feel a bit sore afterward. While it sounds like a negative thing, lactic acid is a normal byproduct of energy production and is managed by our bodies. If it were a strong acid, our muscles would be in serious trouble!
The ability of weak acids to exist in a balance, to donate and accept hydrogen ions as needed, is what makes them so useful in buffering systems. Buffering is like having a shock absorber for pH levels. Imagine you’re driving on a bumpy road. A buffer system, often involving weak acids and their related bases, helps to smooth out the ride, keeping the pH stable. This is incredibly important in our blood, for example, where maintaining a very specific pH is vital for life.

Let’s imagine another scenario. You’ve spilled something on your favorite shirt, and you’re trying to clean it. Some cleaning products use weak acids to help break down stains. They have enough acidity to tackle the mess but are gentle enough not to damage the fabric. It’s like a delicate operation where you need precision, not brute force.
So, when you encounter a question about identifying a weak acid, think about these everyday examples. Is the substance known for its mild tang? Does it contribute to pleasant flavors in food? Is it found in common household items without being overly corrosive? These are all clues pointing towards a weak acid.
It’s also worth noting that many compounds that are weak acids are also found naturally. This is a testament to their role in nature. From the acids in fruits that help preserve them to the acids involved in biological processes, the natural world relies heavily on the gentle power of weak acids.

The opposite, strong acids, are things like hydrochloric acid, which is used in digestion (but in a very controlled way in our stomach!) or sulfuric acid, which is a powerful industrial chemical. These have a much greater tendency to release their hydrogen ions, making them far more reactive and potentially dangerous if not handled with extreme care.
Understanding the difference between weak and strong acids isn’t just for chemists. It helps us make informed choices, appreciate the science behind our food and drinks, and understand how our own bodies function. It’s about recognizing the subtle but significant players in the chemical world that make our lives easier, tastier, and healthier.
So, the next time you’re enjoying a citrusy drink, a tangy salad dressing, or even just thinking about how your body works, give a little nod to the weak acids. They’re the unsung heroes, the gentle forces that contribute so much to our everyday experiences. They prove that you don't need to be loud and aggressive to be incredibly important!
