Which Of The Following Compounds Is A Meso Compound
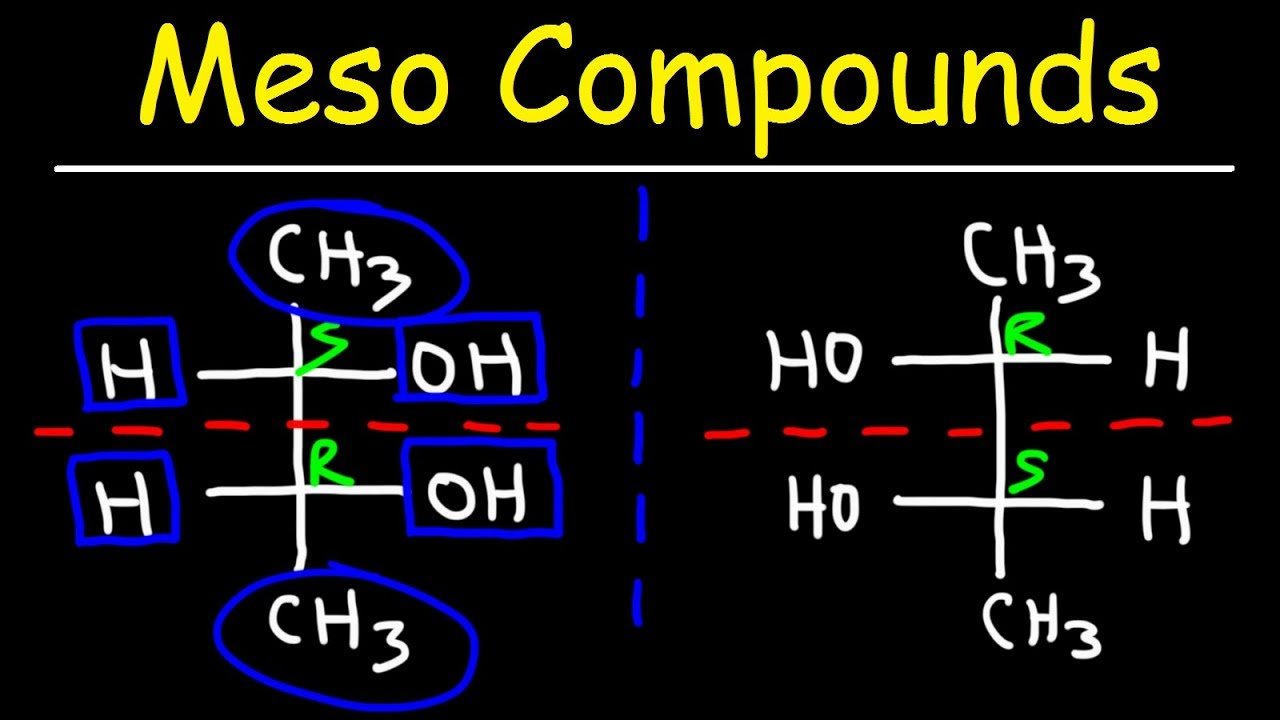
We’ve all been there, right? Staring at a list of chemical compounds, wondering, "Which one of you is the imposter? The one that’s a bit of a… trickster?" If you’re nodding along, you’re probably familiar with the delightful, albeit sometimes mind-bending, world of stereochemistry and, more specifically, the quest to identify a meso compound. It’s like a chemical scavenger hunt, and for many, cracking these molecular puzzles is incredibly satisfying. It’s a game of symmetry, a dance of molecules, and understanding it unlocks a deeper appreciation for the intricate structures that make up our world.
So, why do we bother with these seemingly esoteric concepts? Beyond the academic intrigue, understanding chirality and the nuances of molecular structure, like identifying meso compounds, has profound implications in our everyday lives. Think about it: the effectiveness of many pharmaceuticals hinges on their specific 3D shape. A drug molecule that’s a mirror image of its intended form might be completely inactive or, worse, harmful. Identifying meso compounds helps chemists understand and predict the behavior of molecules, leading to safer and more effective medicines, from pain relievers to complex cancer treatments. It also plays a role in understanding the flavors and fragrances of the foods we eat and the perfumes we wear. Sometimes, it's the specific 3D arrangement, not just the atoms themselves, that dictates whether something smells like a rose or, well, something else entirely!
The concept of a meso compound often pops up when discussing molecules with multiple chiral centers. A chiral center is essentially a carbon atom bonded to four different groups, giving it a "handedness," much like your left and right hands. Most molecules with chiral centers exist as enantiomers – non-superimposable mirror images. However, a meso compound is a fascinating exception. It possesses chiral centers but is achiral overall due to an internal plane of symmetry. This means that despite having those handed parts, the molecule as a whole is identical to its mirror image. Common examples you might encounter in textbooks include tartaric acid and certain sugars like meso-inositol. These compounds, despite having multiple chiral carbons, behave as if they don't have any stereoisomers other than themselves and their reflection, which are actually the same molecule!
Ready to dive deeper and become a meso compound spotting pro? Here are a few practical tips. Firstly, visualize the molecule. Don't just look at the 2D structure on paper; try to imagine its 3D arrangement. Drawing it out using wedge-and-dash notation can be incredibly helpful. Secondly, look for internal symmetry. Can you draw a plane through the molecule that divides it into two halves that are mirror images of each other? If you can, and the molecule has chiral centers, it's a strong indicator of a meso compound. Thirdly, practice makes perfect. Work through examples, and don't be afraid to consult resources. Understanding the rules and then applying them to various molecular structures will solidify your ability to identify these unique molecules. The satisfaction of correctly identifying a meso compound is truly a rewarding part of understanding the beautiful complexity of chemistry.
