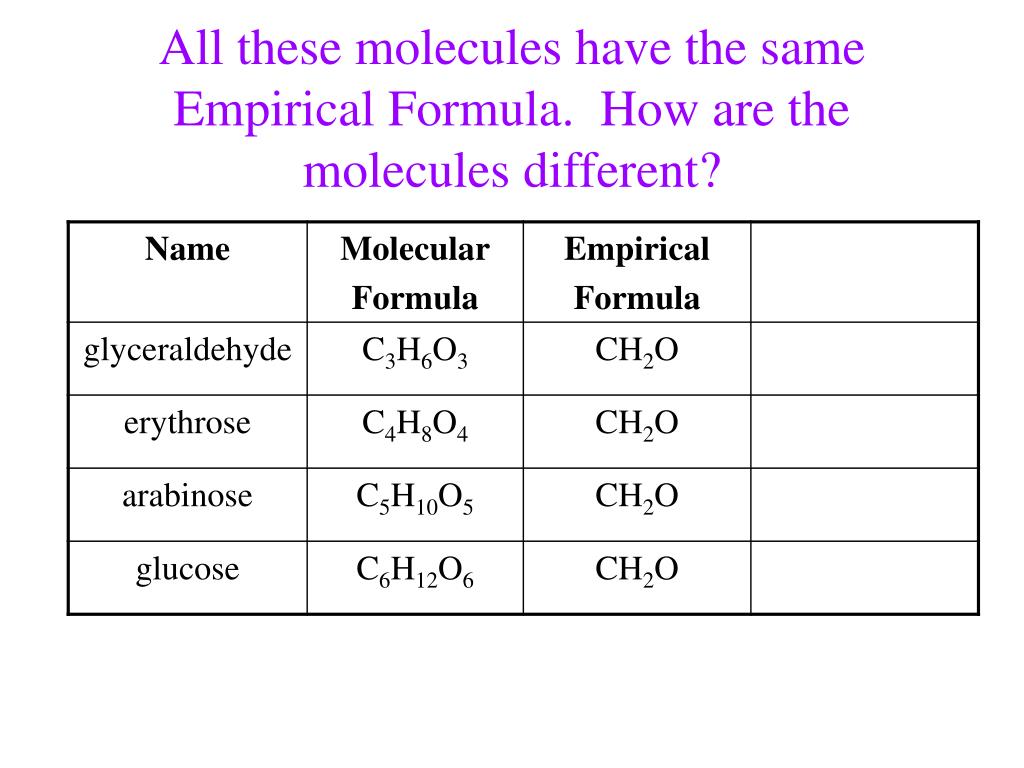
Which Of The Following Compounds Have The Same Empirical Formula
Alright, gather 'round, you lovely bunch of humans! Let's talk about chemistry, but before you start picturing bubbling beakers and lab coats that mysteriously turn yellow, I promise this is going to be more like a treasure hunt. We're on a mission, a quest to find out which of these compounds are secretly related, like estranged cousins at a family reunion. They might look different, they might act different, but deep down, they share a common ancestor. And that ancestor, my friends, is called the empirical formula.
Now, what in the name of all things molecular is an empirical formula? Think of it as the simplest possible ratio of atoms in a compound. It's like getting the recipe down to its absolute bare-bones ingredients, stripped of all the fancy extras. For example, if you have a giant, decadent chocolate cake (let's call it C12H22O11 – fancy name, right?), its empirical formula might just be C1H2O1. We just found the smallest whole number that divides all those numbers. It's the "chocolate, flour, sugar" of the molecule, not the "artisanal cocoa powder from a single origin, sustainably farmed almond flour, and hand-harvested unrefined cane sugar." You get the drift.
So, the big question is: Which of the following compounds have the same empirical formula? Let's say we’re staring down a lineup of chemical suspects. We’ve got:
- Glucose: C6H12O6 (This is the stuff that fuels your brain, or that you crave after a particularly brutal spin class.)
- Sucrose: C12H22O11 (Ah, the sweet, sweet sugar that makes your coffee sing.)
- Water: H2O (The ultimate beverage. Hydration nation!)
- Hydrogen Peroxide: H2O2 (The stuff that cleans cuts and sometimes makes your hair a bit lighter, if you’re brave.)
- Ethane: C2H6 (A simple hydrocarbon, building block for bigger things.)
- Propane: C3H8 (You know, for your barbecue. Makes burgers taste like victory.)
Our mission, should we choose to accept it, is to simplify these bad boys down to their most basic ratios. Let's put on our chemical detective hats.
Operation: Empirical Formula Extraction!
First up, we have Glucose. We're looking at C6H12O6. Now, we need to find the greatest common divisor (GCD) for the numbers 6, 12, and 6. Think of it like finding the biggest number that can evenly divide all of them. In this case, it's a super straightforward 6. So, we divide each subscript by 6:
C6/6H12/6O6/6 = C1H2O1. Or, more simply, CH2O. This is our empirical formula for glucose. It’s like saying, "Yep, this molecule is basically one carbon, two hydrogens, and one oxygen, repeated."

Next, let's tango with Sucrose. Its formula is C12H22O11. Now, let's try to find a common divisor for 12, 22, and 11. Uh oh. The only number that divides all three perfectly is 1. Eleven is a prime number, folks! You can't divide 22 by 11 and get a whole number and do the same for 12 and 11. So, sucrose, in its full glory, is already at its simplest form. Its empirical formula is C12H22O11. It's like finding a perfectly symmetrical snowflake – no need to simplify further.
Moving on to the life-giving nectar, Water! H2O. The subscripts are 2 and 1. The greatest common divisor here is 1. So, the empirical formula for water is H2O. Yep, it's already as simple as it gets. Sometimes, the simplest things are the most profound, right? Like a good cup of tea.
Now for Hydrogen Peroxide. Its formula is H2O2. We have subscripts 2 and 2. The GCD is a lovely, round 2. Divide by 2:

H2/2O2/2 = H1O1. Or, more commonly written as HO. So, hydrogen peroxide's empirical formula is HO. Interestingly, water and hydrogen peroxide have different empirical formulas. Water is H2O, hydrogen peroxide is HO. They're like siblings: same basic elements, but different personalities and slightly different (and in hydrogen peroxide's case, more explosive!) natures.
Let’s get our hands dirty with Ethane. C2H6. The subscripts are 2 and 6. The GCD is 2. Divide by 2:
C2/2H6/2 = C1H3. Or, CH3. So, ethane’s empirical formula is CH3. It’s a humble beginning, this CH3. Like a single Lego brick.

Finally, the king of the barbecue, Propane! C3H8. The subscripts are 3 and 8. Now, these two numbers are coprime. That means their only common divisor is 1. So, propane, like sucrose, is already in its simplest form. Its empirical formula is C3H8. No simplification needed. It's already as lean and mean as a perfectly grilled steak.
The Grand Reveal: Who Shares a Secret Identity?
So, let's line up our empirical formulas:
- Glucose: CH2O
- Sucrose: C12H22O11
- Water: H2O
- Hydrogen Peroxide: HO
- Ethane: CH3
- Propane: C3H8
Now, the moment of truth! Which of these have the same empirical formula? Look closely. Do you see it?

The answer is... Glucose! Its empirical formula is CH2O. And guess what? It shares this secret identity with some other molecules that aren't on our list right now, but are super important. Things like formaldehyde (CH2O) and acetic acid (C2H4O2 – if you divide that by 2, you get CH2O!). They all boil down to the same simplest ratio of atoms. It’s like finding out that your unassuming neighbor is secretly a world-renowned chef, or that the quiet librarian is actually a karate master. Mind. Blown.
So, to recap: we’ve cracked the code, we’ve simplified the molecular jargon, and we've discovered that while compounds might have different molecular formulas (that's the full, fancy version), they can totally share the same empirical formula. It's a bit like how many different types of bread are made from flour, water, and yeast. Different shapes, different textures, but the same fundamental ingredients.
And there you have it! Chemistry, served with a side of fun facts and a dash of mild astonishment. Now, who’s up for a coffee? I think I’ve earned a sweet treat, and I’m pretty sure sucrose and I are going to have a long talk about our shared elemental destiny.
