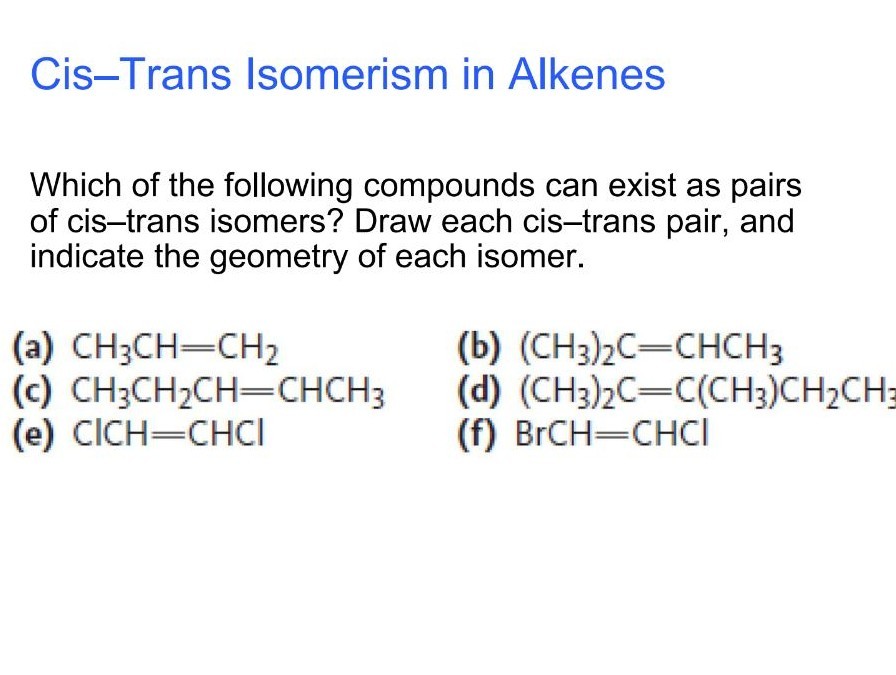
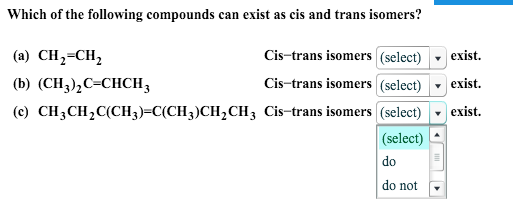
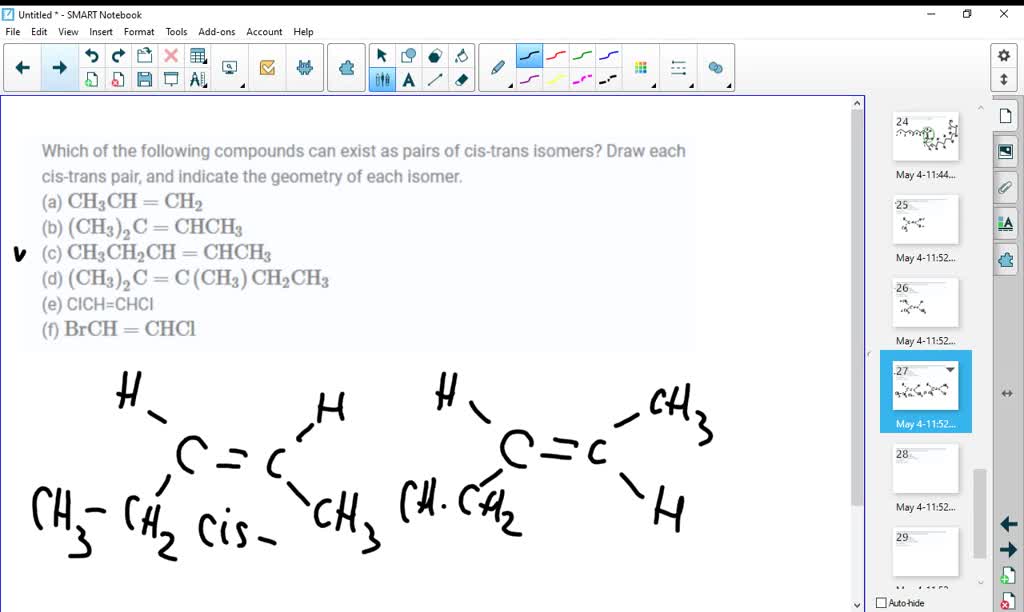
Which Of The Following Compounds Can Exist As Cis-trans Isomers

Hey there, curious minds! Ever feel like chemistry is all about confusing formulas and scary-sounding words? Well, buckle up, buttercup, because we're about to dive into something super cool that can actually make your everyday life a little more… interesting! We're talking about cis-trans isomers. Don't let the fancy name fool you; it's all about how things are arranged, and trust me, it can be a real game-changer!
So, what's the big deal? Imagine you've got a molecule, right? Think of it like a tiny Lego construction. Now, sometimes, those Lego bricks can be put together in different ways, even if you use the exact same pieces. That's kind of what isomerism is all about. And cis-trans isomerism is a particularly neat flavor of this phenomenon.
The Magical World of Molecular Jiggery-Pokery
Alright, let's get down to the nitty-gritty. For a compound to be able to pull off this cis-trans trick, it needs a couple of key features. Think of it as needing the right "stage" and the right "actors" for the show to go on!
The Non-Negotiable: A Double Bond (or a Ring!)
First things first, our molecule needs a bit of rigidity. It can't be flopping around all willy-nilly. The most common culprit for this necessary stiffness is a double bond, usually between two carbon atoms. You know, like this: C=C. This double bond is like a super-strong connection that doesn't allow the atoms attached to it to spin freely. They're kinda locked in place!
Alternatively, a ring structure can also create this rigidity. Imagine a bicycle wheel. The atoms forming the ring are stuck in a circular arrangement, limiting their movement. So, either a double bond or a ring is your first requirement for the cis-trans party to begin.
The Crucial: Different Groups on Each Side
Now, here's the really important part. For cis-trans isomerism to even be possible, each of the atoms involved in the double bond (or part of the ring) needs to have two different groups attached to it. Let's break this down. If you have a C=C double bond, look at one of the carbon atoms. If it has, say, two identical hydrogens attached (like CH2=), then nope, no cis-trans action here. It's like trying to have two different people wear the exact same hat – it just doesn't work for differentiation!
So, for our C=C to play the cis-trans game, each carbon needs to be sporting a unique pair of "accessories." For example, one carbon might have a hydrogen and a methyl group (CH3), and the other carbon might have a hydrogen and a chlorine atom (Cl). See? Different groups on both sides!
The Big Reveal: Cis vs. Trans!
Once we've got our molecule with its rigid double bond (or ring) and different groups on each carbon, then the magic happens. We get two distinct arrangements, and these are our cis and trans isomers!
The "Same Side" Sensation: Cis
The cis isomer is like two best friends who decide to stand on the same side of the street. In molecular terms, this means that two similar groups (or the same group on each carbon) are on the same side of the double bond (or across the ring). Think "cis" sounding a bit like "this side."
For instance, if we have two methyl groups attached to our double bond, in the cis isomer, both methyl groups would be pointing up (or both down) relative to the double bond. It's a neat, tidy arrangement!

The "Opposite Side" Odyssey: Trans
Now, the trans isomer is like those same friends deciding to hang out on opposite sides of the street. Here, two similar groups are on opposite sides of the double bond (or across the ring). Think "trans" like "transported" to the other side!
Using our methyl group example again, in the trans isomer, one methyl group would be pointing up, and the other would be pointing down. They're across from each other, maintaining a bit more "personal space," if you will.
Why Does This Even Matter? It's More Than Just Pretty Pictures!
You might be thinking, "Okay, so they look different. Big whoop!" But here's where it gets really fun. These different arrangements aren't just for show; they can have a massive impact on the molecule's properties!
Think about how different shapes can fit into things. A key that's slightly bent won't fit into a lock designed for a straight key, right? Molecules are similar! The cis and trans isomers of a compound can have different boiling points, melting points, solubilities, and even different biological activities.
Real-World Wonders!
This isn't just theoretical stuff, oh no! These isomers pop up all over the place. Take fats, for example. The fats we eat are made of molecules with long chains, and whether they have cis or trans double bonds makes a huge difference. Trans fats, which have those opposite-side arrangements, were notoriously bad for our health. Our bodies just aren't built to process them efficiently. On the other hand, cis fats are generally considered healthier. See? A subtle change in arrangement can have a profound impact on your well-being!
Or consider vision! The molecule responsible for detecting light in our eyes, rhodopsin, works because of a molecule called retinal. Retinal exists in both cis and trans forms. When light hits it, the cis form temporarily changes to the trans form, triggering a chain reaction that sends a signal to your brain. Pretty neat, huh? Your ability to see the world depends on these molecular shape-shifters!
Even things like flavors and fragrances can be influenced by cis-trans isomerism. Different isomers can smell or taste completely different. One might be a delightful perfume, while its isomer is, well, less than pleasant. So, the next time you smell a beautiful flower or enjoy a delicious meal, give a little nod to the amazing world of isomers!
Could This Compound Be a Cis-Trans Star?
So, how do we figure out if a compound can even do the cis-trans dance? It's like a quick checklist:
- Does it have a double bond or a ring structure?
- If it has a double bond, are the two carbons in that bond each attached to two different groups?
- If it has a ring, are there at least two atoms in the ring where substituents (attached groups) can be on the same side or opposite sides?
If the answer to these questions is a resounding "YES!" then congratulations, you've found a potential cis-trans isomer! It's like finding a hidden treasure in the world of molecules.
It's amazing to think that such small differences in arrangement can lead to such big differences in how things behave. It reminds us that sometimes, the most profound changes come from subtle shifts. It encourages us to look closer, to appreciate the details, and to understand that even the smallest structures have their own unique stories to tell.
So, the next time you encounter a chemical name, take a moment to wonder: could this molecule be a secret shapeshifter? Could it be a cis-trans marvel? This journey into the world of molecular arrangements is just the beginning. Keep exploring, keep questioning, and you'll discover that chemistry isn't just a subject; it's a vibrant, fascinating universe waiting to be understood!
