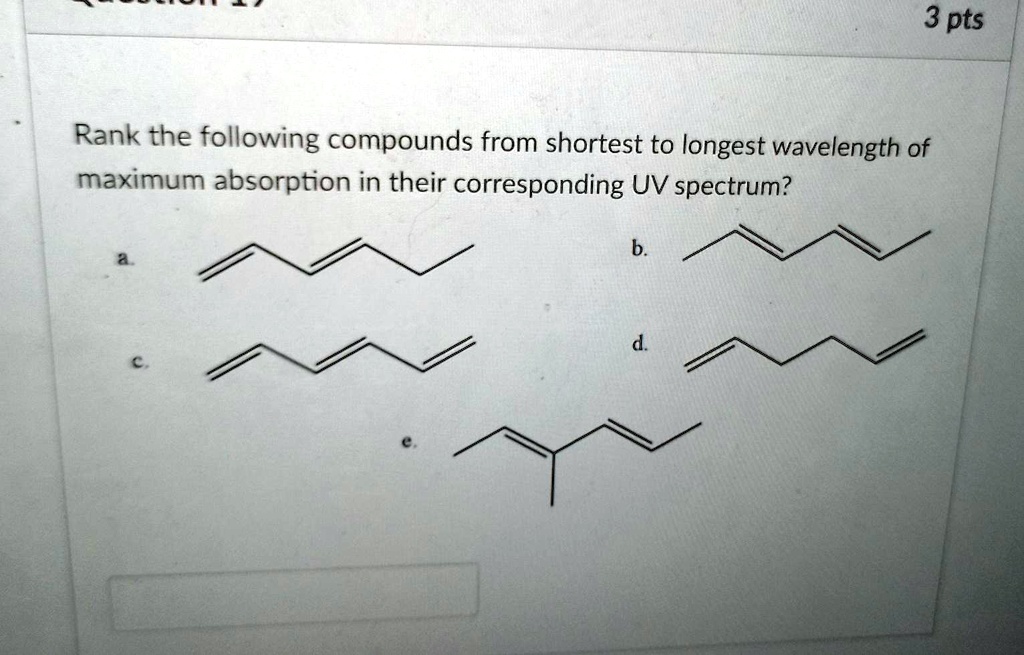
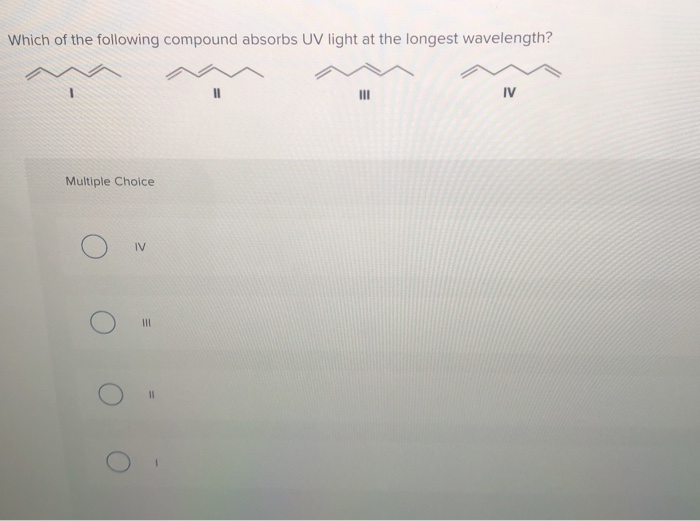
Which Of The Following Compounds Absorbs At The Longest Wavelength

Hey there, fellow science enthusiasts! Ever stare at a rainbow and wonder why some colors seem to stretch out longer than others? Or maybe you've been fiddling with some cool chemical experiments and noticed how different stuff can glow or change color under different lights? Well, today we're diving into the wonderfully weird world of how molecules interact with light, specifically, which of our mystery compounds is soaking up light like a sponge at the longest wavelengths. Think of it as a light-catching contest, and we're here to figure out who the champion is!
Now, I know what you might be thinking. "Wavelengths? Longest? Sounds a bit… science-y." But stick with me, because it's actually pretty neat and not nearly as complicated as it sounds. Imagine light as a bunch of waves, like ripples on a pond. Some waves are tiny and choppy, while others are long and lazy. These different sizes are what we call wavelengths. And different wavelengths of light have different amounts of energy. The longer the wave, the less energy it packs. Think of a gentle, rolling wave at the beach compared to a tiny, frantic splash. The big, lazy wave is like our long wavelength light. Get it?
So, when we talk about a compound "absorbing" light, it’s like that compound is grabbing onto a specific type of light wave and using its energy. It's like the compound is saying, "Ooh, a nice, long, chill wave! I'll take that one!" And when a compound absorbs certain wavelengths of light, the light that isn't absorbed is what bounces back to our eyes, which is how we see color! Pretty cool, right? It’s like the compound is wearing a specific outfit of wavelengths and showing off the ones it didn't choose. Mind. Blown.
Our quest today is to figure out which compound is the ultimate chill wave collector. Generally speaking, the more complex a molecule is, and the more it can spread out its electrons (like little happy dancers doing a synchronized routine), the more likely it is to absorb longer wavelengths of light. It's like a bigger dance floor means more room for elaborate moves, which can then interact with longer, slower music (our light waves). Smaller, simpler molecules tend to be a bit more energetic and prefer those shorter, punchier wavelengths. They're like the folks at the club who want to jump around to the fast beats!
Let's imagine we have a few hypothetical compounds on our table, ready for their close-up. For the sake of our fun little investigation, let's give them some fun, quirky names. We've got:
Compound A: The Tiny Sparkler
This little guy is probably something pretty simple. Think something like water (H₂O), or maybe a simple alkane like methane (CH₄). These molecules are small, tightly packed, and don't have a lot of wiggle room for their electrons. They're like a tiny, tightly wound spring. They tend to absorb light in the ultraviolet (UV) range. UV light has really short wavelengths and is quite energetic. You know how UV rays from the sun can give you a sunburn? That's because they're energetic enough to mess with our skin cells. So, our Tiny Sparkler is definitely not the long-wavelength absorber we're looking for. It's too busy absorbing energetic, short-wavelength stuff!
Compound B: The Steady Glow Stick
Now, this one's a bit more interesting. Let's say Compound B is something like an alkene with a few double bonds, or maybe a small aromatic ring like benzene (C₆H₆). These molecules have a bit more structure. They have these things called pi electrons that are a bit more delocalized, meaning they can spread out a little more than in Compound A. Think of it as our molecule having a slightly bigger stage for its electron dancers. Because these electrons can move around more freely, they can absorb light with a slightly longer wavelength than the UV range. They might be dipping into the visible light spectrum, perhaps absorbing in the blue or violet region. When they absorb blue light, what do we see? We see the complementary color, which is yellow or orange! So, Compound B is starting to get warmer, but it's still not the champion of long-wavelength absorption. It's a good contender, though!
Compound C: The Rainbow Weaver
Here's where things get exciting! Compound C is our complex beast. Imagine something with lots of conjugated double bonds – alternating single and double bonds, like a molecular caterpillar. Think of pigments like beta-carotene (the stuff that makes carrots orange) or even larger organic molecules found in dyes. In these molecules, the pi electrons are highly delocalized. They can spread out over a huge portion of the molecule, almost like a giant, interconnected network of electron dancers all moving to the same slow, groovy beat. This extensive electron delocalization means they have very low energy gaps between their electron states. And what does a low energy gap mean? It means they can absorb light with very long wavelengths and low energy. They'll be soaking up visible light, maybe even into the red or infrared regions of the spectrum. When they absorb these longer wavelengths, they reflect the shorter, higher-energy wavelengths, which is why they appear in vibrant colors to us. Our Rainbow Weaver is looking like a strong contender, possibly the winner!
Compound D: The Ghostly Whisperer
Let's invent one more for good measure, just to make sure we’re really thinking about this. Compound D could be something like an ionic compound, or a simple inorganic salt. Think something like sodium chloride (NaCl), table salt. These compounds are made of charged ions held together by strong electrostatic forces. Their electrons are generally tightly bound to individual ions. They don't have large, delocalized pi systems like our organic compounds. Because their electrons are so localized and the energy gaps are so large, they typically don't absorb visible light at all. They might absorb in the very short wavelength UV region, or perhaps not even there. They're basically invisible to our light-catching contest in the visible spectrum. So, definitely not our winner!
So, let's put on our detective hats. We're looking for the compound that absorbs at the longest wavelength. This means we're looking for the compound with the lowest energy absorption, because longer wavelengths have less energy. And what gives a molecule the ability to absorb lower energy light? It's that magical thing called electron delocalization! The more spread out the electrons are, the easier it is for them to absorb lower-energy photons (those light particles).
Compound A, our Tiny Sparkler, is small and has localized electrons. It absorbs high-energy UV light. Nope.

Compound B, the Steady Glow Stick, has some delocalization, so it's absorbing a bit longer wavelengths, but still likely in the shorter end of visible or UV. Getting closer, but not there yet.
Compound D, the Ghostly Whisperer, is likely not absorbing much visible light at all. Definitely out.
And then there's Compound C, the Rainbow Weaver, with its extensive system of conjugated double bonds. Its electrons are practically doing a marathon across the molecule. This means it has the smallest energy gap between its ground state and excited state. And a smaller energy gap means it can be excited by photons with less energy. Less energy per photon means longer wavelengths! Bingo!
So, when you're asked "Which of the following compounds absorbs at the longest wavelength?" you're essentially being asked: "Which compound has the most delocalized electrons and therefore the smallest electronic energy gap?" In our fun little lineup, that's our magnificent Compound C, the Rainbow Weaver! It's the champion of soaking up those long, lazy light waves.
It's a beautiful dance, isn't it? The way molecules and light interact. It's what gives us all the colors we see, from the deepest blues of the ocean to the fiery reds of a sunset. Every time you see a vibrant color, remember the complex interplay of electrons and light waves happening at the molecular level. It’s a constant, silent performance, and Compound C is clearly stealing the show with its long-wavelength absorption!
So, next time you're marveling at a colorful flower, a vibrant dye, or even a perfectly ripe piece of fruit, give a little nod to the molecules that are masters of light absorption. They're not just sitting there; they're actively participating in the visual symphony of our world. And that, my friends, is something pretty amazing to smile about!
