Which Of The Following Best Describes The Term Thermal Energy
Hey there, fellow coffee enthusiast! So, you're wondering about thermal energy, huh? It’s one of those words that pops up everywhere, isn't it? Like, "Wow, this coffee is hot thermal energy!" Or maybe, "Brr, my hands are cold, not enough thermal energy here!" It's like a secret code for how much oomph stuff has when it comes to heat.
Let's break it down, shall we? Think of it like this: everything around us, even that comfy chair you're probably sinking into right now, is made of tiny little bits. We're talking atoms and molecules, the real VIPs of the universe. And guess what? These little guys are never, ever still. Nope. They’re constantly jiggling, wiggling, and dancing. It's like a never-ending disco in every single thing!
Now, the speed of that jiggle-wiggle dance? That’s where thermal energy comes in. The faster these tiny particles are moving, the more thermal energy something has. Pretty straightforward, right? It's like when you're really excited and you can't stop fidgeting – you've got a lot of energy, a lot of oomph. Same principle, but on a super-tiny, invisible level.
So, when your coffee is piping hot, it’s because the water molecules in it are going absolutely bonkers. They're zooming around at a million miles an hour (okay, maybe not a million, but you get the picture!). They're bumping into each other, doing the cha-cha, the tango, the whole shebang. That frenetic movement is what we feel as heat. It’s that energetic buzz that warms you up.
On the flip side, when something is cold? Think of that ice cream cone you're trying to finish before it melts into a sugary puddle. The particles in the ice cream are moving much, much slower. They’re doing a gentle sway, a slow waltz. Less jiggling, less wiggling, means less thermal energy. It's like they’re all politely waiting for their turn on the dance floor.
Now, here’s a fun little twist for you. Thermal energy is technically the total kinetic energy of all the particles in a substance. So, it's not just one particle’s speedy dance, but the grand sum of all their movements. Imagine a huge ballroom full of dancers. The thermal energy is the energy of the entire crowd, not just that one person doing the splits in the corner (though they contribute!).

We often use the word "heat" interchangeably with thermal energy, and that's not entirely wrong, but it's a bit like saying "car" when you mean "transportation." Heat is actually the transfer of thermal energy. It’s the movement of that energetic dance from one place to another. Like when your warm mug of coffee transfers its thermal energy to your chilly hands. Your hands are like the receptive audience, soaking up that delightful energy!
So, if you're ever asked to describe thermal energy, think of it as the internal energy of a substance due to the random motion of its atoms and molecules. That’s the fancy scientific way of saying it. But in our casual coffee chat? It's the energy of movement of all those tiny little doodads that make up everything.
Let's ponder some scenarios, shall we? Imagine a metal spoon sitting in that hot cup of coffee. The spoon's molecules are already wiggling, but when they touch the hot coffee molecules, they get a serious energy boost. They start wiggling even faster, and this energetic transfer zips all the way up the spoon. That’s why your spoon gets hot too! It's like a chain reaction of happy, energetic particles.
What about something like a block of ice? The water molecules in the ice are pretty sluggish, just doing their slow-motion dance. But if you leave it out in a warm room, the warmer air molecules will bump into the ice molecules. This transfer of energy makes the ice molecules jiggle a bit faster. Eventually, they jiggle so much that they break free from their frozen structure and become liquid water. Voila! Melting. All thanks to a little bit of thermal energy transfer.
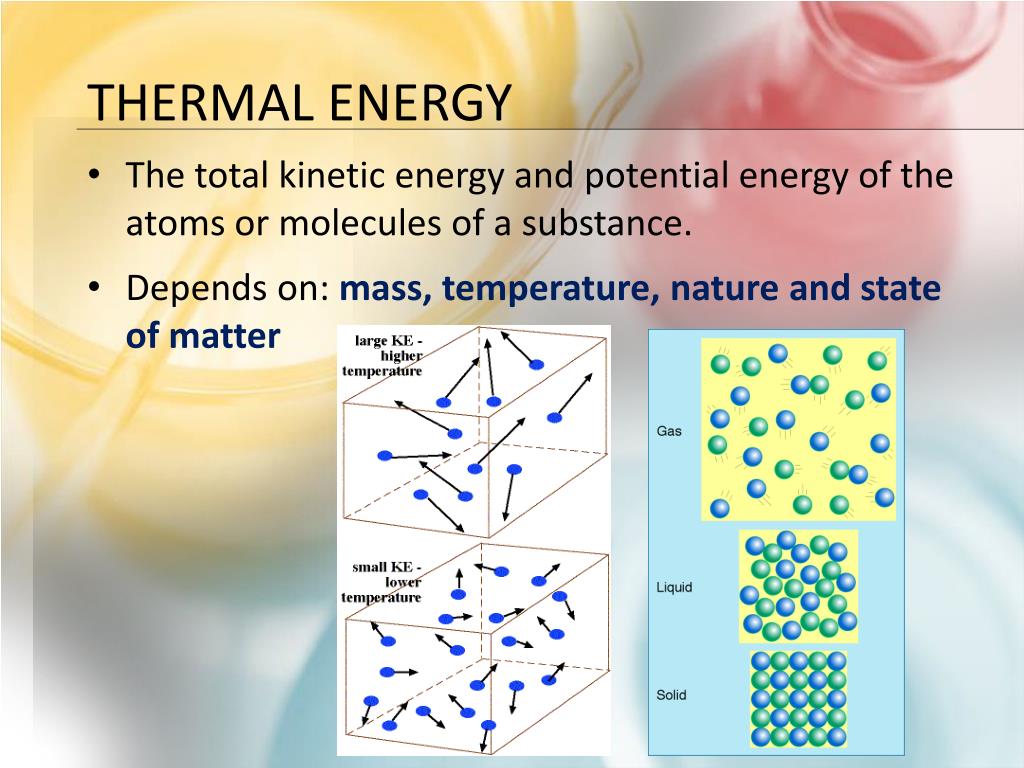
Sometimes, we talk about temperature. Is temperature the same as thermal energy? Almost, but not quite. Temperature is more like the average kinetic energy of those particles. It’s like looking at the overall tempo of the dance floor. If the average dancer is moving really fast, the temperature is high. If they're moving slowly, the temperature is low.
So, you could have two things with the same temperature, but different amounts of thermal energy. Imagine two cups of water, both at 50 degrees Celsius. One is a tiny espresso cup, and the other is a giant mug. The tiny cup has fewer water molecules, so even though they're dancing at the same speed, there are fewer of them to contribute to the total energy. The giant mug, with way more water molecules doing their dance, will have more thermal energy. It's like comparing a small, energetic puppy to a big, lazy St. Bernard – they might have similar levels of "excitement" (temperature), but the St. Bernard has way more overall "enthusiasm" (thermal energy) just by virtue of being bigger!
This concept is super important in so many areas. Think about cooking! When you bake a cake, you're transferring thermal energy from the oven to the cake batter. That energy makes the molecules in the batter move faster, causing chemical reactions that turn goo into deliciousness. It’s like a science experiment with a yummy outcome.
Or what about the weather? The sun sends tons of thermal energy our way, warming up the Earth's surface, oceans, and atmosphere. This energy drives weather patterns, creating winds, rain, and all those other atmospheric shenanigans. It’s the ultimate source of warmth for our planet.

Even in your own body, thermal energy is playing a starring role. Your metabolism is constantly generating thermal energy as it breaks down food for fuel. This internal heat helps maintain your body temperature, keeping you warm and alive. It’s like having a tiny furnace inside you, working 24/7. Pretty neat, huh?
So, if we were to distill it down to the absolute essence, which of the following best describes thermal energy? Let’s think about some options you might encounter:
Option A: The amount of heat that can be transferred from one object to another.
Hmm, this is close, but not quite it. Remember how we said heat is the transfer? This option is talking about the potential for transfer, which is related, but it’s not the energy itself. It’s like saying a battery is the amount of electricity that can be used, rather than the stored electrical energy.
Option B: The measure of how hot or cold something is.
Nope, that's temperature! We already chatted about that. Temperature is the average energy. This option describes the average, not the total. So, while related, it’s not the full picture of thermal energy.

Option C: The total kinetic energy of the particles within an object.
Aha! Now we’re talking! This one hits the nail on the head. It’s that sum of all the wiggles and jiggles of all those tiny atoms and molecules. It’s the complete energetic dance party happening inside anything and everything. This is the one!
Option D: The energy released during a chemical reaction.
While chemical reactions can produce or consume thermal energy, this isn't the definition of thermal energy itself. It’s like saying "fire" is the definition of "burning." Burning involves fire, but fire is a phenomenon, not the entire definition of the process. Thermal energy exists even when no reaction is happening!
So, there you have it. When you hear "thermal energy," just think of all those little particles inside things doing their energetic jig. The more they jig, the more thermal energy it’s got. It's the energy of movement at the atomic and molecular level. Simple as that, really. Now, where did I put my coffee? I think my hands could use a little more thermal energy transfer!
It’s fascinating how much is going on at a level we can’t even see, isn’t it? That invisible dance is what makes the world go ‘round, literally and figuratively. From the warmth of your morning brew to the vastness of the sun, thermal energy is the unsung hero. So next time you feel warm, you can mentally thank all those busy, energetic particles doing their thing. They’re the real MVPs of the universe!
