Which Of The Following Best Describes Cis-trans Isomers

Hey there, curious minds! Ever stumbled upon a science term and thought, "Whoa, what’s that all about?" Well, today we're diving into a little something called cis-trans isomers. Now, don't let the fancy name scare you! Think of it less like a daunting chemistry lecture and more like a fun little puzzle, a bit like finding hidden treasures in everyday life. And trust me, understanding this can actually make life a whole lot more interesting.
So, what exactly are these elusive cis-trans isomers? Imagine you have two identical Lego bricks, but you can snap them together in slightly different ways. That's kind of the vibe we're going for here, but with molecules! Basically, cis-trans isomers are molecules that have the same atoms and the same chemical bonds, but they are arranged differently in three-dimensional space. It's all about the spatial arrangement, the way things are positioned relative to each other.
The "Same, But Different" Superpower
Think about it this way: you’ve got two identical twins. They look almost exactly the same, right? But if you get to know them, you’ll notice subtle differences in their personalities, their quirks, and how they hold their coffee mug. Cis-trans isomers are like those twins. They’re made of the same building blocks, connected in the same order, but their 3D orientation is different. And that, my friends, is where the magic happens!
This little difference in orientation can have some pretty big consequences. It's like the difference between a handshake and a high-five – same basic concept of greeting, but a totally different feel and implication. In chemistry, these subtle spatial tweaks can change how molecules interact with each other, how they taste, how they smell, and even how they work in our bodies.
Let's Get Visual: The Double Bond Dance Party
To really grasp this, we need to talk about a special kind of bond: the double bond. Imagine two atoms holding hands, but instead of just one hand each, they’re linking up with two hands each! This double bond is pretty rigid; it doesn't allow for easy rotation. Think of it like a tightly fastened screw – you can’t just spin it freely.

Because of this rigid double bond, the atoms or groups attached to the atoms involved in the double bond are kind of stuck in place. They can't freely twirl around each other. This is the crucial factor that allows for cis-trans isomerism. If there were no double bond, or if there was free rotation around a single bond, everything would be able to spin around, and you wouldn't get these distinct spatial arrangements.
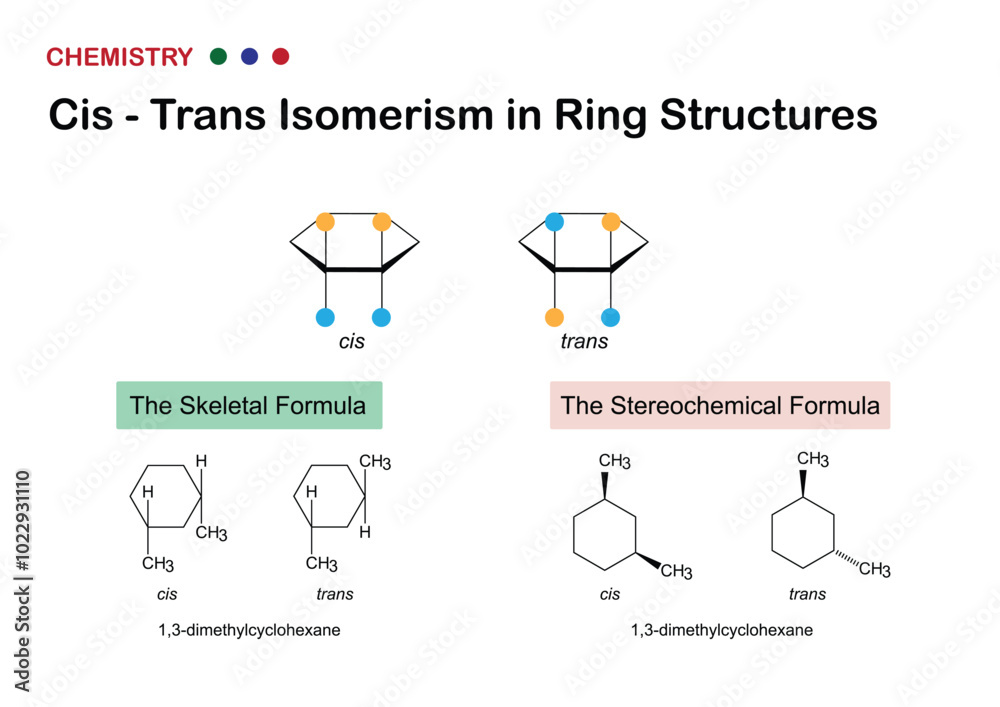
So, picture this: you have a molecule with a double bond in the middle. On either side of this double bond, you have some groups attached. Now, here's the fun part. If you have two identical groups on the same side of the double bond, we call that the cis isomer. Think "cis" sounding like "same" – two similar things on the same side. It's like two friends standing next to each other on the same side of a street.
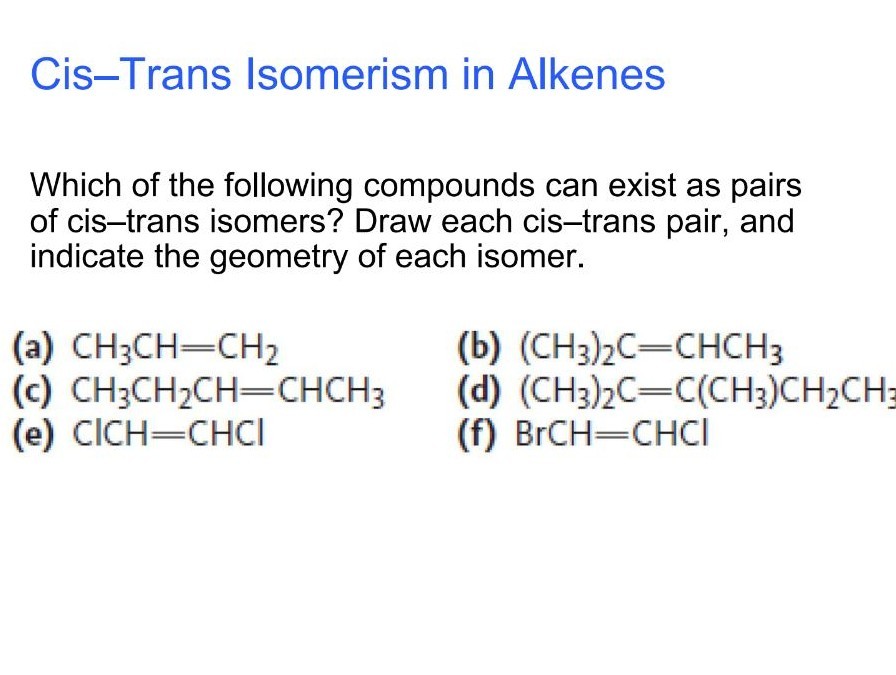
On the flip side, if those two identical groups are on opposite sides of the double bond, we've got ourselves the trans isomer. "Trans" sounds a bit like "across" or "transfer" – the groups have crossed over to the other side. Imagine those same two friends, but now one is on one side of the street and the other is on the opposite sidewalk. They’re still friends, but their positions are distinctly different.
Why Should You Care? Because It's Everywhere!
You might be thinking, "Okay, that's neat, but how does this relate to my life?" Oh, buckle up, because it’s more involved than you think! Think about your favorite foods. The way a molecule is arranged can drastically affect its flavor and aroma. Take, for example, molecules that contribute to the smell of fruits. A cis isomer might smell like a ripe strawberry, while its trans counterpart could have a completely different, perhaps less appealing, scent. So, next time you're enjoying a delicious fruit, you can ponder the fascinating world of cis-trans isomers at play!
And what about your health? Our bodies are intricate chemical factories, and the way molecules are shaped is super important for how they interact with our cells and enzymes. Certain vitamins, like Vitamin A, exist as both cis and trans isomers. Your body is designed to use specific forms of these molecules for various functions. So, the correct isomer matters for everything from vision to cell growth!
Let’s not forget about the pharmaceutical industry. The effectiveness and even the side effects of many medications can depend on whether they are in the cis or trans form. It's a critical consideration for chemists designing drugs to make sure they're getting the right shape to do the job they're intended to do. Imagine a key fitting into a lock – the shape has to be just right, and that's where cis-trans isomerism comes into play in drug design.
The Takeaway: It's All About Perspective!
So, to circle back to our initial question, which of the following best describes cis-trans isomers? It’s the molecules that have the same atoms and connectivity, but differ in the spatial arrangement of their atoms around a rigid structure, most commonly a double bond. It's not about having different ingredients; it's about arranging those ingredients in a slightly different, yet significant, way.

This concept is a beautiful illustration of how small differences can lead to significant outcomes. It's a reminder that in the vast and intricate world of chemistry, the subtle nuances of structure can have profound impacts. It’s like looking at a painting from different angles – you see the same colors and shapes, but the overall impression can shift.
Isn't that inspiring? The universe is full of these little molecular "twists" and "turns" that create the incredible diversity and functionality we see all around us. From the food we eat to the medicines that heal us, cis-trans isomers are quietly playing their part.
So, the next time you encounter a scientific concept, don't shy away! Dive in, ask questions, and explore. You might just discover that the world of molecules is a whole lot more fun and fascinating than you ever imagined. Keep that curiosity alive, and who knows what other molecular marvels you'll uncover!
