Which Of The Following Are Correct For Zero Order Reactions
Let's dive into the wild and wacky world of chemical reactions! Specifically, we're talking about the ones that are so chill, so laid-back, they barely break a sweat. We're talking about Zero Order Reactions. Now, some people might find this topic a bit dry. They might yawn and think, "Chemical kinetics? Snooze." But I'm here to tell you, it's actually a hoot! Think of it like this: some reactions are like a toddler on a sugar rush, going a mile a minute. Others are like my grandpa on a Sunday afternoon – moving at their own pace, and you can't rush them. Zero order reactions are definitely in the grandpa category.
So, what makes a reaction a zero-order reaction? It’s a bit of a rebel, really. You see, most reactions are all about proportions. Like, if you double the ingredients, you get double the product, right? Makes sense. But zero-order reactions? They just don't care. They're like, "Nope. Whatever you do to the amounts of stuff you're mixing, I'm going to react at the same speed. Period." It’s almost like they have a mind of their own, a very stubborn, unyielding mind. And honestly, sometimes I feel like that too. Especially on a Monday morning.
Let's get a little more specific, though. We’re playing a fun game today: "Which of the following are correct for zero-order reactions?" It’s like a pop quiz, but way more fun because there are no wrong answers, just answers that fit the zero-order vibe. Imagine you're at a party, and there are different types of dancers. Some are doing the electric slide, all synchronized and predictable. Then you have the zero-order dancers. They're just doing their own thing, maybe a slow sway, and whether there are ten people on the dance floor or just two, they keep swaying at the exact same rhythm.
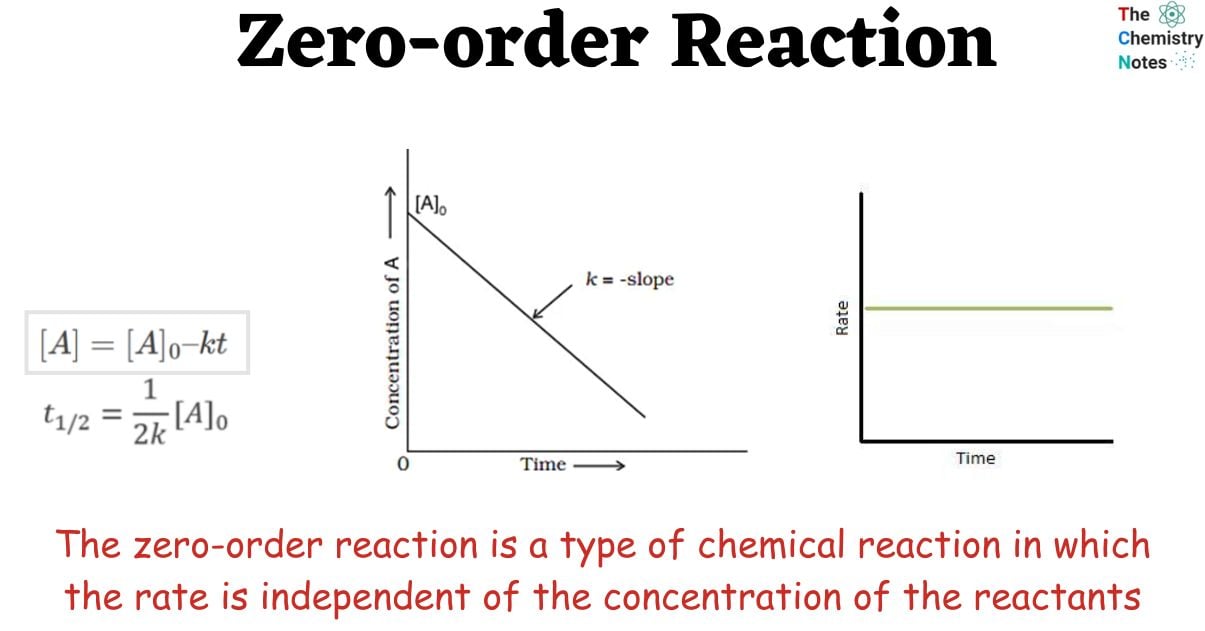
So, what are some of the things that are totally, unequivocally, correct for these groovy zero-order reactions? Well, for starters, their reaction rate is just… constant. It’s like a steady heartbeat. You can throw more reactants into the mix, but the speed doesn't change. It's like trying to speed up a turtle by giving it more lettuce. It'll still eat at its own pace. This is a big one, folks. The rate is independent of the concentration of the reactants. It’s the ultimate “I’ll get to it when I get to it” attitude in chemistry.
Another super important point, and this is where things get really interesting, is the integrated rate law. Don't let the fancy name scare you. It's basically the equation that tells you how much stuff is left after a certain amount of time. For zero-order reactions, this integrated rate law looks like a beautiful, simple straight line. Yep, a straight line! If you plot the concentration of the reactant against time, you get a perfectly straight line going downwards. It's so predictable, so… linear. It’s like drawing a line on a piece of paper. No curves, no wiggles, just pure, unadulterated linearity. It's almost too perfect, isn't it? Makes you wonder if it's hiding something. But nope, it's just that straightforward.

Think about it this way: if you’re baking a cake, and you decide to double the flour, you expect a bigger cake, right? And maybe it takes a bit longer to bake if it’s a huge cake. That’s not zero-order. Zero-order is like saying, "I’m baking this cake. It takes 30 minutes. Whether I use one egg or ten eggs, it will still take 30 minutes. The number of eggs is just… there." It’s a bit absurd, but that’s the charm! It defies our everyday logic, and that’s what makes it fun to ponder.
Now, let's talk about the half-life. This is the time it takes for half of your reactant to disappear. For zero-order reactions, the half-life is not constant. And this is where it gets really interesting and maybe a little counter-intuitive. The half-life actually depends on the initial concentration. So, if you start with a lot of reactant, the first half will disappear relatively quickly. But then, the remaining half will take longer to disappear. It's like saying, "Okay, I'll eat half my pizza. Then I'll take a break. Then I'll slowly, meticulously eat the rest." It's not a consistent munching pace. It’s a bit like how some people approach dessert – the first piece is gone in a flash, the second lingers.
So, to recap our little adventure:
The rate is independent of reactant concentration. This is the superstar rule.
It’s like the reaction has its own personal clock and it’s not checking anyone else’s watch.

The integrated rate law results in a linear plot of concentration versus time. Remember that beautiful straight line? That's the signature move.
It’s the chemical equivalent of a perfectly made bed.
The half-life is dependent on the initial concentration. It's not a fixed amount of time.
This means the reaction can be a bit of a procrastinator when there's less stuff left. It’s the tortoise and the hare, but the tortoise is getting slower as it goes.
Are these the only things that are correct for zero-order reactions? For our little game today, yes! These are the main hallmarks. These are the quirky traits that make zero-order reactions stand out in the crowd. They’re not like the others, and that’s perfectly fine. In fact, it's better than fine. It’s delightfully different. So next time you hear about a zero-order reaction, don't just nod and pretend you understand. Smile, think of the laid-back grandpa or the slow-swaying dancer, and appreciate its wonderfully uncomplicated, yet intriguing, nature. It’s a reaction that marches to the beat of its own drum, and honestly, who doesn't admire that a little? I know I do. It’s like finding a hidden gem in the vast ocean of chemical reactions, and it’s always a treat to discover its simple, elegant truths.
