Which Of The Following Alkenes Is The Most Stable
Okay, so imagine you're at a party. You've got a bunch of cool people milling around, right? Some are super popular, always the center of attention. Others are a little more reserved. Well, alkenes are kind of like that! They're organic molecules with a special double bond between two carbon atoms. Think of it as their little secret handshake. And just like people, some alkenes are more stable, more… chill, than others. We're gonna find out which one's the ultimate party animal in the world of alkenes!
So, what makes an alkene "stable"? It's all about how much energy it takes to break that double bond. The more energy you need, the more stable it is. It's like trying to get a really popular person to leave a party – it takes a lot of convincing! Low energy means it's happy where it is. High energy means it's just waiting for something to happen, like a bouncy ball ready to pop.
Let's get down to the nitty-gritty. We're talking about a few contenders here. Imagine them lined up, ready for a popularity contest. We've got the simple ones, the ones with a bit more going on, and then some real show-stoppers. It’s not just about how many carbons are hanging around; it’s about how they're hanging around that double bond. It’s a bit like a dance floor – the more people dancing, the more energy, but also, maybe the more organized the whole scene can become?
First up, let's consider something super basic. Think of ethene. That's just two carbons with a double bond and some hydrogens chilling. It's the wallflower of our alkene party. It's got the double bond, sure, but not much else to stabilize it. It’s the baseline, the starting point. It’s like the plain ol' water at a fancy cocktail bar. It serves its purpose, but it's not exactly turning heads.
Now, let's spice things up a bit. What happens when we add some substituents? These are like little decorations or friends that join the party. For alkenes, these substituents are usually alkyl groups – basically, chains of carbon and hydrogen. Think of them as adding more arms and legs to our dancing alkenes. The more alkyl groups attached to the carbons of the double bond, the more stable the alkene tends to be. It's like a group hug for the double bond!
Why does this happen? It's all about something called hyperconjugation. Don't let the fancy word scare you! It's basically like the adjacent alkyl groups are lending a hand to the double bond, spreading out its electron density. Imagine the double bond is like a really bright light. Hyperconjugation is like putting frosted glass around it – it diffuses the light, making it less intense and therefore more stable. It’s a subtle but super important effect!
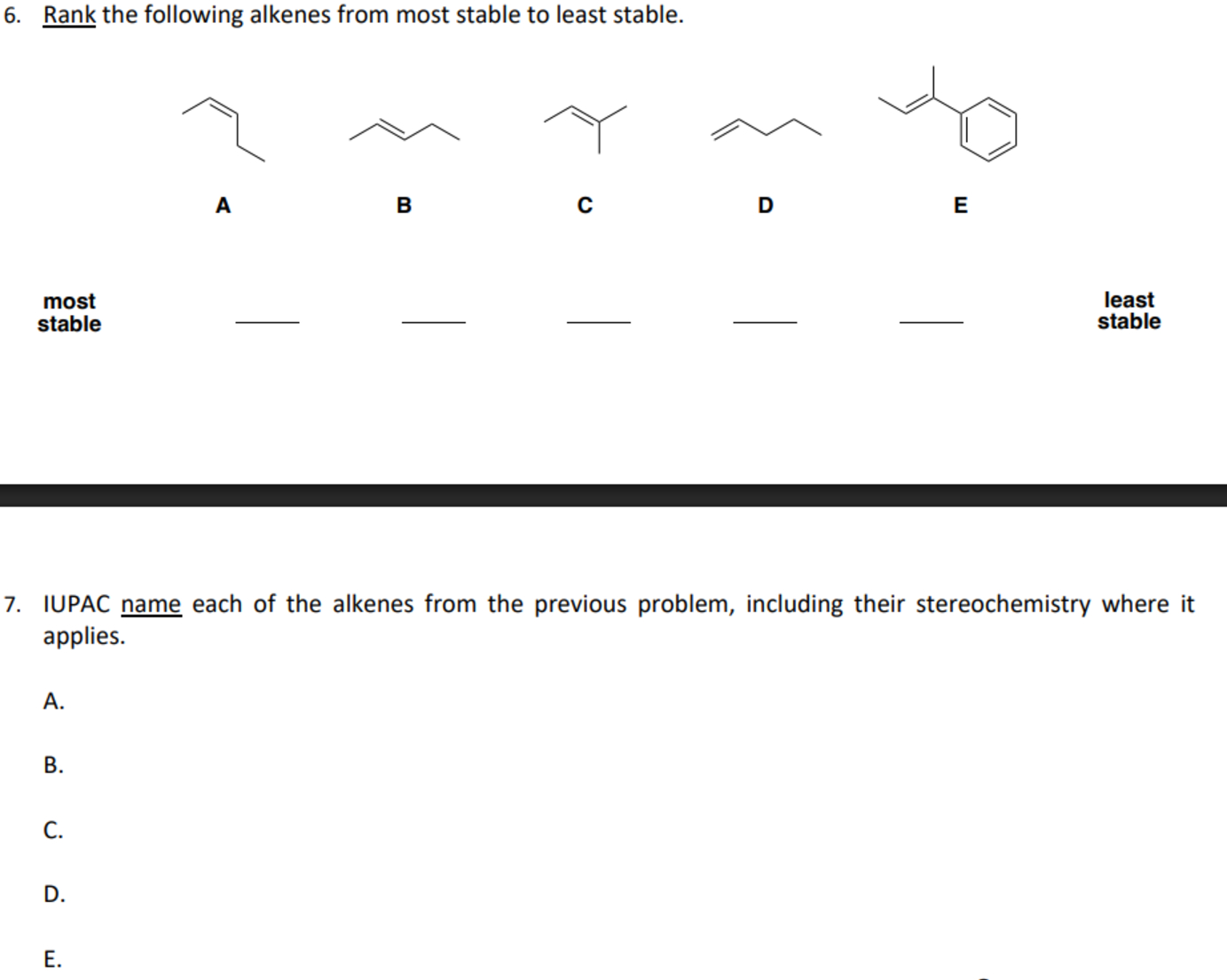
So, if we have one alkyl group attached to the double bond, it's more stable than ethene. That's a monosubstituted alkene. If we have two, that's a disubstituted alkene, and it's even more stable. Three? Trisubstituted. Four? Tetrasubstituted. It's a clear trend, a predictable pattern. It’s like a game of "more is more" for stability.
But here's where it gets really interesting. We're not just talking about any old alkyl groups. The position of these groups matters. And sometimes, the way the double bond is arranged in space matters even more. We're talking about cis and trans isomers. This is where things get a little mind-bendy, and it's super fun.
Imagine our double bond as a rigid structure, like a fence. The groups attached to the carbons are like flags on either side. In a cis isomer, the two identical (or similar) groups are on the same side of the double bond. Think of two friends standing on the same side of the fence, waving. In a trans isomer, they are on opposite sides. Like two friends on opposite sides of the fence, maybe giving each other a thumbs-up across the gap. It’s like a little molecular choreography!
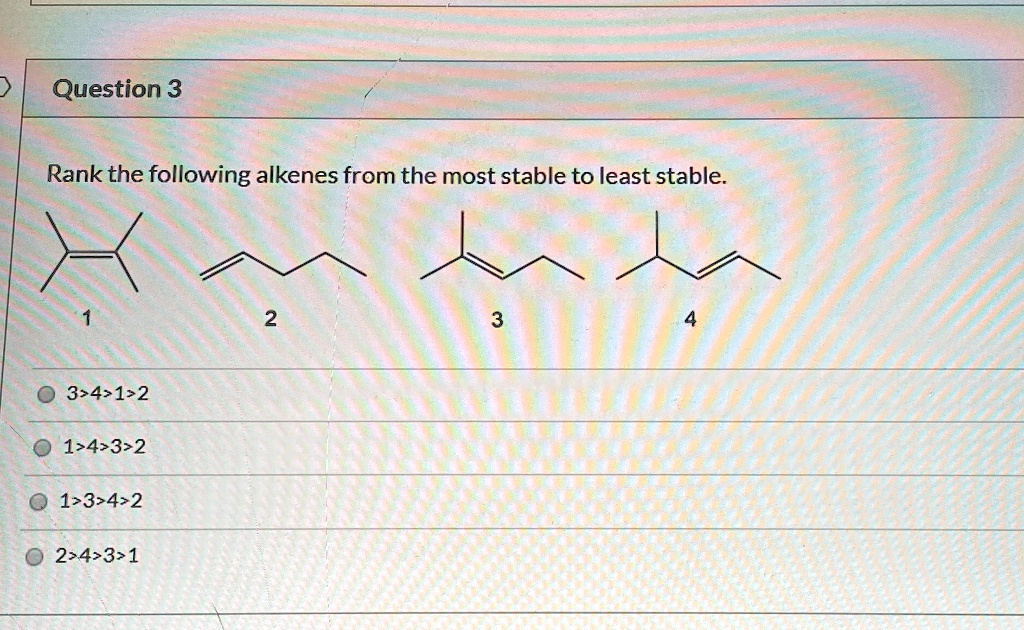
Now, here’s the quirky fact: trans isomers are generally more stable than cis isomers. Why? Because in the cis isomer, those bulky groups can bump into each other. Imagine trying to fit two big suitcases right next to each other on a tiny shelf. They're gonna be a bit crowded and uncomfortable, right? That's steric hindrance! The trans isomer avoids this awkwardness. The groups are further apart, giving each other plenty of personal space. Ah, the sweet relief of personal space!
So, if we're comparing a cis disubstituted alkene to a trans disubstituted alkene, the trans one wins the stability crown. It's just more comfortable being in that spread-out position.
Now, let's put it all together. Imagine we have a few options laid out. Let's say we're choosing between:
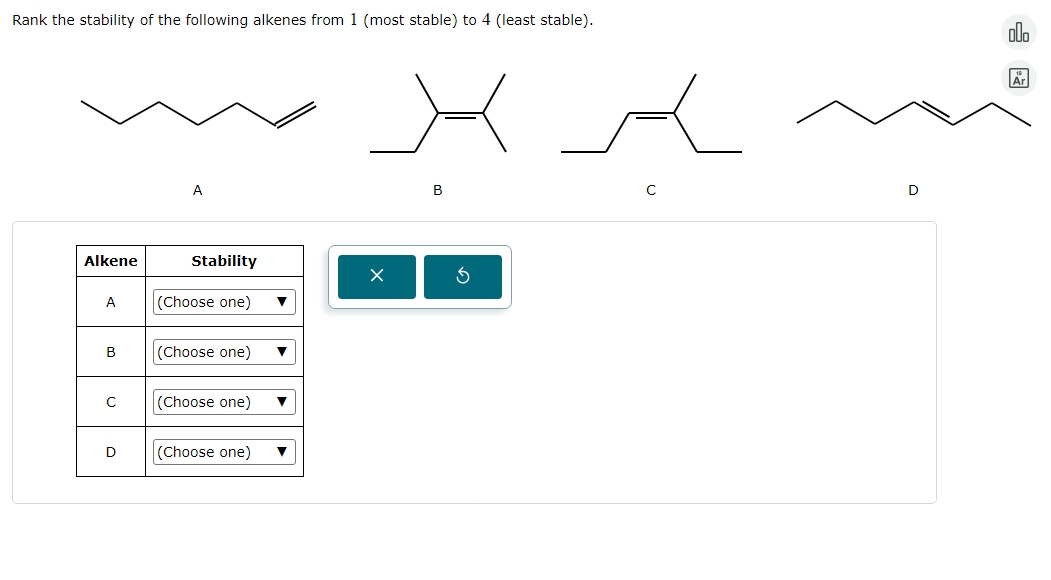
- Ethene (no substituents)
- Propene (one substituent)
- 1-butene (one substituent)
- 2-butene (two substituents, and here we have a choice: cis-2-butene and trans-2-butene)
- 2-methylpropene (two substituents, but arranged differently)
Based on what we've discussed, ethene is the least stable. Propene and 1-butene are more stable than ethene because they have one alkyl group. Now, comparing 2-butene and 2-methylpropene, both are disubstituted. But how are they substituted? In 2-butene, the two methyl groups are attached to the carbons of the double bond. In 2-methylpropene, one carbon of the double bond has two methyl groups attached, and the other has only hydrogens. This arrangement in 2-methylpropene is what we call a geminal disubstitution (meaning both substituents are on the same carbon).
Here’s a fun detail: while more alkyl groups generally mean more stability, the arrangement can sometimes be more nuanced. For our specific comparison, and considering the common trends, we usually see that a trans disubstituted alkene is more stable than a cis disubstituted alkene. And when comparing different substitution patterns, the fully substituted double bond (like in tetrasubstituted alkenes) tend to be the most stable due to the cumulative effect of hyperconjugation and minimized steric strain. However, in the context of typical general chemistry examples, the focus often sharpens on the cis vs. trans debate among disubstituted alkenes, or comparing alkenes with increasing numbers of alkyl substituents.
So, if our options included cis-2-butene and trans-2-butene, the trans-2-butene would be the more stable one. It's got that extra space, that relaxed vibe. It's not feeling the squeeze!

What about that 2-methylpropene? It's got two methyl groups attached to one carbon of the double bond. This is a geminal disubstituted alkene. It's got a different kind of "crowding" going on. While it's more stable than monosubstituted alkenes, it’s often less stable than a trans disubstituted alkene like trans-2-butene. Why? Because the two methyl groups on the same carbon create a bit of repulsion, even though they're not directly bumping into each other across the double bond.
The ultimate winner in terms of stability in most common scenarios, especially when comparing simple alkenes, is usually the one with the most alkyl substituents arranged in the most favorable way. If we were presented with a list that included a tetrasubstituted alkene (where all four positions on the double bond are occupied by alkyl groups), that would likely be the king or queen of stability in that group. Think of it as a double-decker bus of alkyl groups, all snug and stable!
So, to answer the ultimate question, without a specific list of alkenes, it's hard to point to one definitive molecule. But the principles are clear! More alkyl groups attached to the double bond? More stable. Trans arrangement over cis? More stable. It's a fun puzzle to solve, like a chemical Sudoku. And the more you learn about these little molecular differences, the more you realize how much cool stuff is happening at the atomic level. It's not just boring chemicals; it's a whole world of interactions and preferences!
So next time you hear about alkenes, remember the party! Some are wallflowers, some are disco divas, and some are just chilling with their friends. And stability? It's all about who's got the best dance moves and the most personal space. Pretty cool, right?
