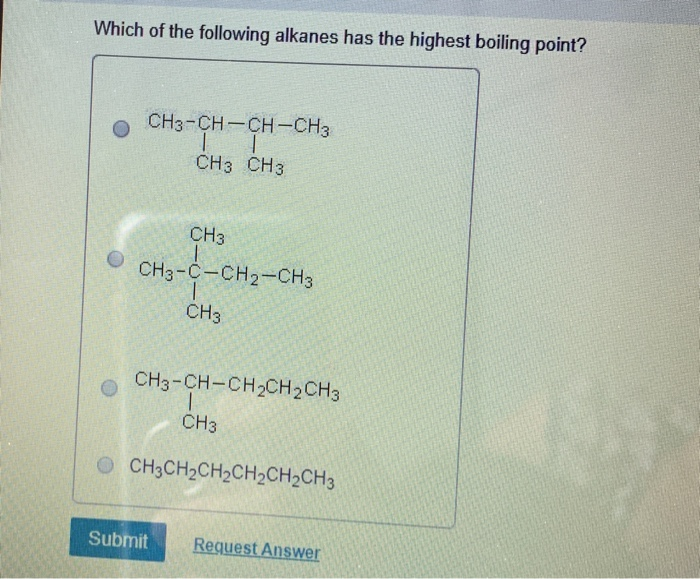
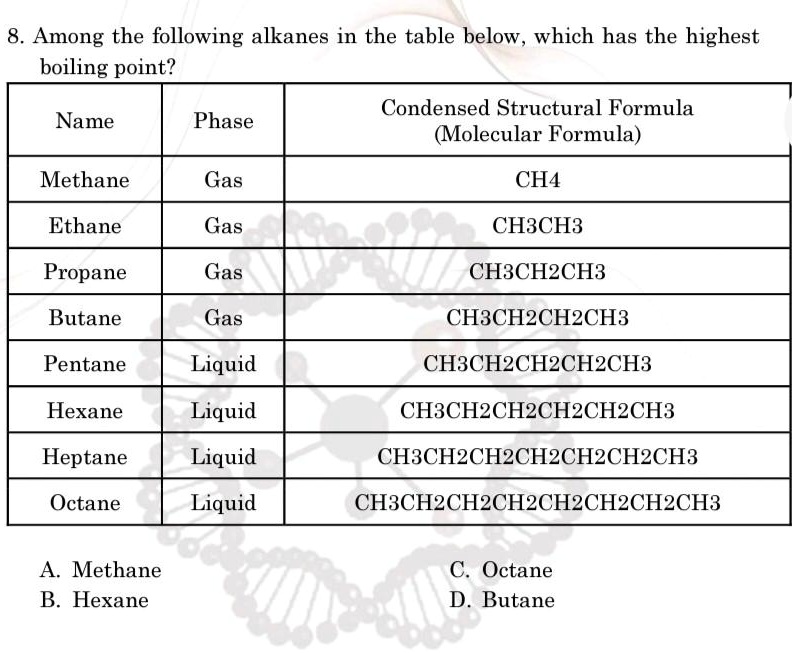
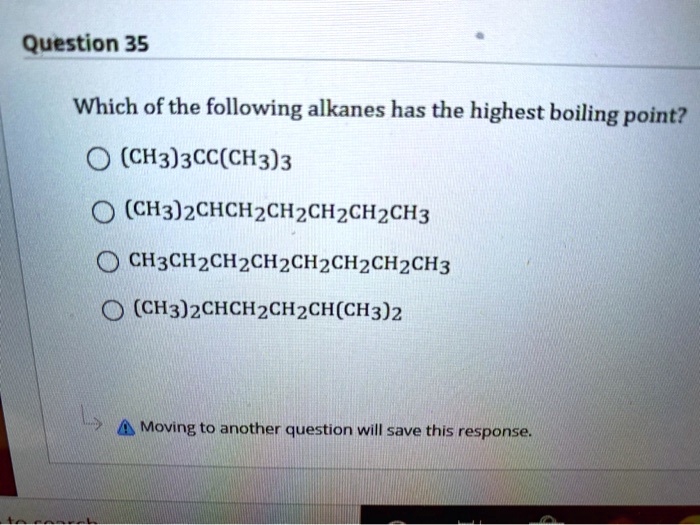
Which Of The Following Alkanes Has The Highest Boiling Point

Get ready for a super fun science adventure, folks! We're diving into the exciting world of alkanes and figuring out which one throws the best boiling point party. Think of them as tiny, but mighty, building blocks of so many things around us.
Imagine you're at a summer barbecue, and you've got a bunch of friends chilling. Some are super chill and ready to jump into the grill's heat (that's like a low boiling point!), while others are a bit more hesitant, needing a serious sizzle to get going. We're looking for the ultimate reluctant toaster among our alkane pals!
Now, let's meet our contenders. We have methane, the super lightweight of the group. It’s like a little firefly, zipping around with barely any effort. Then there's ethane, a slightly beefier friend, who’s got a bit more oomph. Next up, we have propane, the king of your backyard grill adventures, always ready to bring the heat!
And let's not forget about butane! This little guy is famous for his role in those handy lighter things, sparking up smiles and light. Each of these guys is a simple hydrocarbon, made of just carbon and hydrogen atoms, all linked up like a friendly chain.
But here's the kicker: even though they’re all related, their boiling points are as different as night and day! Some need just a gentle nudge to turn from a liquid into a gas, while others are absolute stubborn mules, demanding a full-on fiery embrace before they’ll budge.
So, which of our amazing alkane friends is the ultimate heat-seeker? Which one needs the most intense party to start bubbling and frothing like a mad scientist's potion?
Let's start with our littlest champ, methane. This guy is practically a gas already at room temperature. It's like trying to boil water that's already steaming – it just doesn't need much convincing!
Methane, with its single carbon atom, is the epitome of a lightweight. It’s so tiny and so simple that its molecules barely even notice each other. They’re like a few people trying to hold hands in a giant ballroom – not much grip!
So, methane boils at a frigid -161.5 degrees Celsius. Brrr! That’s colder than a polar bear's picnic! You wouldn't want to be trying to cook with methane at that temperature, unless you were aiming for ice cream.
Next up, we have ethane. Ethane has two carbon atoms linked together. It's a bit like methane bringing along a buddy. Now there are two tiny things to keep track of, and they’re a little bit more interested in hanging out together.
Because there are two carbon atoms in ethane, its molecules have a bit more 'stickiness' to them. They're like two friends holding hands a bit tighter than just a brief clasp. This means it takes a tiny bit more energy to pull them apart and turn them into a gas.
Ethane needs a temperature of -88.5 degrees Celsius to start its boiling party. Still super cold, but a noticeable jump up from methane! It’s like going from an ice cube to a very, very chilly slushie.
Now, let's introduce our grill master, propane! Propane is king of the barbecue for a reason. It’s got three carbon atoms all strung together in a neat little line. This means there are more molecules wanting to cling to each other, like a happy, tightly-knit family.
With three carbon atoms, propane molecules have a stronger pull on each other. They’re like a group of friends holding hands in a circle – much harder to break apart! This requires more heat to get them to dance into a gaseous state.

Propane’s boiling point is a much warmer -42 degrees Celsius. Now we’re getting somewhere! This is where things start to get interesting, and you can imagine the sizzle of your burgers starting to happen.
And finally, let’s talk about our final contender, butane. Butane is the seasoned veteran of this group, boasting a chain of four carbon atoms. That’s a whole lot of atoms linked up, giving these molecules a significant amount of 'stickiness'.
Butane’s molecules are like a whole group of friends holding hands in a big, robust circle. They are very reluctant to let go and become individual gas molecules. This requires a substantial amount of heat to overcome their attraction.
Butane boils at a respectable -0.5 degrees Celsius. We're practically at room temperature now! This is why butane is so useful in lighters and camping stoves – it's ready to be a gas without needing extreme cold.
So, let’s recap our contestants and their boiling point superpowers:
- Methane: -161.5 °C (The tiny firefly)
- Ethane: -88.5 °C (The buddy system)
- Propane: -42 °C (The grill king)
- Butane: -0.5 °C (The seasoned veteran)
Looking at these numbers, it’s crystal clear! The alkane that needs the most heat to start its boiling party, the one that is the most stubborn to turn into a gas, is butane!
Think about it like this: imagine trying to get a single feather to fly away versus trying to get a whole pile of feathers to scatter. The pile needs a much bigger gust of wind, right? The more carbon atoms, the bigger the 'pile' of molecules, and the more 'wind' (heat) you need to break them apart.
So, the next time you’re enjoying a barbecue with propane, or flicking a lighter with butane, you can appreciate the amazing science behind it. These simple alkanes, with their varying numbers of carbon atoms, have vastly different appetites for heat!
It’s a fun little puzzle, isn't it? We’ve gone from chilly ice ages to toasty barbecue temperatures just by looking at a few simple molecules.
Remember, as the carbon chain gets longer in these alkanes, the forces holding the molecules together get stronger. It's like adding more and more glue between tiny LEGO bricks. You need a lot more force to pull them apart!
So, the champion of the high boiling point among our chosen alkanes is none other than butane. It’s the ultimate heat-lover in this particular family reunion!
This is just a tiny peek into the amazing world of chemistry. There’s so much more to discover, and it’s all around us, making our lives easier and more exciting!
Keep your eyes peeled for more fun science facts. You never know what cool things you’ll uncover next. Science is everywhere, and it’s always ready to put on a show!
So, next time you see a can of butane, give it a little nod of appreciation. It’s a testament to the power of molecular structure and a champion of the boiling point!
It’s amazing how a simple change in the number of atoms can make such a huge difference. Butane is a superstar in its own right, and now you know why!
We've crowned our winner, and it's all thanks to the magic of chemistry. So, go forth and share your newfound knowledge! You're now an alkane boiling point expert!
Isn't science fun? It’s like a treasure hunt for understanding the world around us, and today, we found a boiling point gem!
So, to answer our burning question: Butane has the highest boiling point among methane, ethane, propane, and butane. It's the rockstar that needs the most heat to get going!
