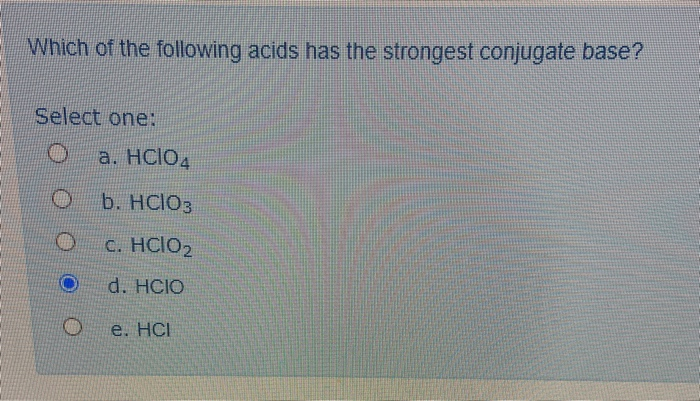
Which Of The Following Acids Has The Strongest Conjugate Base

Imagine a grand party where all the acids are invited, and they’re all dressed up in their finest chemical attire. Each acid brings along its trusty sidekick, a conjugate base. Think of these conjugate bases as the party favors, the little gifts each acid leaves behind after it’s had its fun. Today, we're looking for the acid whose party favor is the most… well, let's just say the most clingy and eager to stick around!
It’s like asking which of your friends is the most likely to crash on your couch after a long night. The acid that leaves behind the strongest conjugate base is the one that’s really good at letting go of its hydrogen. And when we say “strongest conjugate base,” we don’t mean it’s going to win a bodybuilding competition. We mean it's the most unwilling to give up that hydrogen back!
Let’s meet our contenders! We have some famous faces in the acid world. There’s the everyday hero, acetic acid, the one you find in your vinegar. It's a pretty chill acid, not too dramatic.
Then we have hydrochloric acid, a powerhouse often found in your stomach acid. This one is definitely a strong personality, always ready to make a splash!
And let’s not forget sulfuric acid, the king of the strong acids. It’s like the VIP guest at our chemical party, very important and a bit intimidating.
Now, each of these acids, when it decides to donate a proton (that's like a tiny hydrogen atom with a positive attitude), leaves behind its conjugate base. This conjugate base is like the acid’s slightly less adventurous twin.
The question boils down to this: which of these acids is the most generous with its proton, to the point where its conjugate base is just begging for it to come back? It’s a bit of a paradox, isn’t it?

Think of it this way: a strong acid is like a very popular celebrity. It readily sheds its entourage (the proton) because it knows it will always be in demand. Its conjugate base, therefore, is the less desirable member of the entourage, the one who is very keen to get back into the celebrity’s good graces.
So, if a strong acid is eager to get rid of its proton, its conjugate base is going to be desperate to get it back. This means the conjugate base of a strong acid is a weak base. It's like a shy admirer who is too timid to make a move unless practically forced.
Conversely, a weak acid is like a celebrity who’s a bit stingy with their attention. They don’t give up their proton easily. When they do finally give it up, their conjugate base is like a super-fan, really, really happy to have that attention and unlikely to give it back.
Therefore, the acid with the weakest conjugate base is the strongest acid. This is where the fun twist comes in! We’re looking for the acid whose conjugate base is the least likely to try and snatch that proton back.

So, to find the acid with the strongest conjugate base, we are actually looking for the weakest acid among our choices. It’s a bit like a riddle, isn't it?
Let's revisit our guests. Acetic acid, the vinegar friend, is a classic example of a weak acid. It’s a bit hesitant, like someone who always brings a plus-one to a party you thought was just for individuals.
Hydrochloric acid, the stomach acid star, is a very, very strong acid. It’s the life of the party, donating its proton without a second thought. Its conjugate base is the person who politely waits by the door, hoping for a chance to chat again.
And sulfuric acid? Oh, that’s the ultimate show-off. It’s so strong, it practically throws its proton into the crowd! Its conjugate base is the most subdued, the one who’s just happy to have been a part of the event.
So, if we want the acid that leaves behind the strongest conjugate base, we need to find the acid that is the weakest at giving away its proton. This acid's conjugate base will be the most eager to accept that proton back and become the acid again.
Think of it as a game of musical chairs. The strong acids are so good at grabbing a chair (donating their proton) that the person left standing (the conjugate base) is already a bit sad. But the weak acids are a bit slower to the punch. When they finally give up their proton, their conjugate base is absolutely thrilled to sit back down!
Among our friends, acetic acid is the most hesitant to part with its proton. It holds onto it a little tighter than its stronger counterparts.
Because acetic acid is a weaker acid, it means its conjugate base, the acetate ion, is a stronger base. The acetate ion really wants that proton back to reform acetic acid.
In contrast, the conjugate bases of very strong acids like hydrochloric acid (the chloride ion) and sulfuric acid (the bisulfate and sulfate ions) are extremely weak bases. They are so weak, they barely react. They’re like that guest who leaves the party early and doesn't even look back.

So, the acid that has the strongest conjugate base is the one that is itself the weakest acid. It's a bit of a backward compliment!
And in the lineup of acetic acid, hydrochloric acid, and sulfuric acid, our friend acetic acid wins the award for leaving behind the most tenacious, the most eager-to-reunite, the strongest conjugate base.
It’s a reminder that sometimes, the gentlest approaches lead to the most devoted friendships, even in the world of chemistry. The acetate ion is just waiting for its chance to be reunited with its proton, making it the star of our conjugate base party!
So next time you’re enjoying a salad with vinaigrette, you can appreciate the humble acetic acid and its loyal companion, the acetate ion, holding hands in the chemical world.
It’s a heartwarming thought, really. Even in the world of electrons and protons, there’s a bond that’s hard to break, a love that’s hard to let go of. The story of the strongest conjugate base is a story of loyalty and a little bit of chemical romance!
