Which Observation Illustrates The Law Of Conservation Of Mass

Have you ever watched a baker knead dough for a loaf of bread, or a gardener lovingly plant tiny seeds, only to see them transform into something entirely new? It’s like magic, right? Well, sort of. Science has its own kind of magic, and one of its most fascinating spells is called the Law of Conservation of Mass. It sounds a bit fancy, but it’s actually incredibly down-to-earth, and you probably see it happening every single day without even realizing it.
Imagine you have a big, juicy watermelon. You slice it up, enjoy it on a hot summer day, and then… what happens to the rind? You might toss it in the compost bin, or maybe your dog nibbles on it (though we don't recommend that!). Now, let’s think about that rind. It doesn't just vanish into thin air, does it? Nope. It goes on a grand adventure. It breaks down, gets mixed with other bits and pieces, and over time, it helps new plants grow. That watermelon, in its entirety – the delicious pink flesh and the tough green rind – didn't magically lose any weight. It just changed its form, its personality, you could say.
This is where our superhero, the Law of Conservation of Mass, swoops in. It basically tells us that no matter what happens to something, the total amount of “stuff” in it stays the same. Think of it like your favorite toy set. You can take apart all the LEGO bricks and build a spaceship, then a castle, then a robot. You're still using the same number of bricks, they’ve just been rearranged into different shapes. Nothing has been added, and nothing has disappeared.
Let's picture something a little more… explosive! Not a bomb, mind you, but a really fun science experiment. Imagine you mix two liquids together in a clear glass. Maybe one is bubbly and clear, and the other is a bit murky. You pour them together, and poof! A new substance appears. It might fizz, it might change color, it might even start to glow a little (okay, maybe not glow, but it's fun to imagine!). If you were to carefully weigh the two liquids before you mixed them, and then weigh the resulting mixture, you'd find that the total weight is exactly the same. The atoms and molecules from the original liquids just decided to hold hands and dance together in a new way, creating something different but still made of the original ingredients.
Consider the humble candle. You light it, and the flame dances, melting the wax, creating light and warmth. Over time, the candle gets shorter and shorter, and eventually, it’s all gone. It seems like it vanished! But if you could capture all the smoke, all the gases, and all the tiny bits of wax that have evaporated, and add them back together, you'd find you have the exact same amount of "stuff" as you did when the candle was whole and new. The wax didn't disappear; it just turned into something else – mostly gases that floated away into the air. It’s like a disappearing act, but the magician never actually makes anything disappear, they just hide it really, really well!
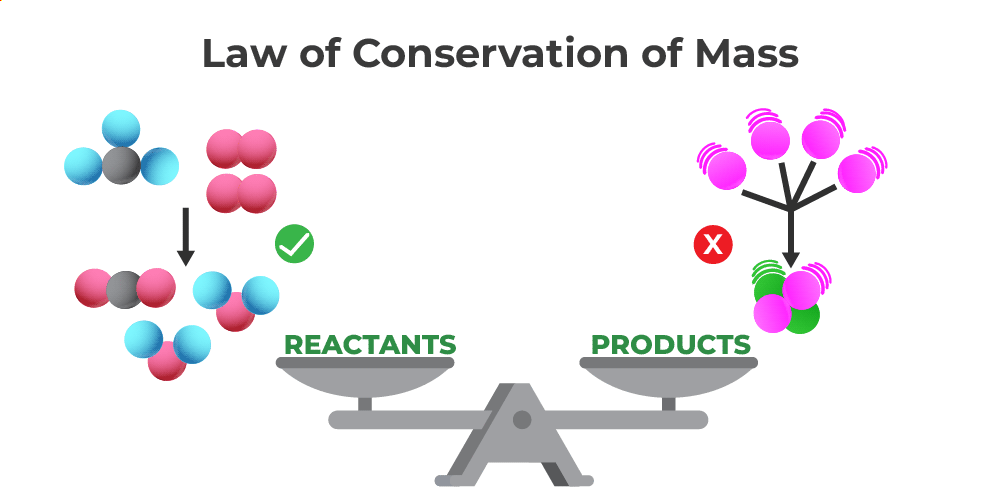
The Law of Conservation of Mass is like the universe's ultimate "nothing is lost, only changed" policy.
Think about cooking. When you bake a cake, you start with flour, sugar, eggs, and butter. You mix them all up, put them in a hot oven, and out comes a fluffy, delicious cake. Did all that sugar and flour just evaporate? Of course not! They’ve undergone a chemical transformation. The heat from the oven caused the ingredients to react with each other, changing their structure and creating the wonderful texture and taste of the cake. But if you could gather up every crumb, every whiff of steam, and every molecule, you’d have the original ingredients back in terms of sheer mass.

Even in our own bodies, this law is at play. When you eat food, your body breaks it down, rearranges it, and uses it for energy and to build new cells. The food doesn't just disappear. It becomes part of you. And when you exhale, you're releasing gases that were once part of that food, transformed. It’s a constant cycle of change, but the fundamental amount of matter involved remains constant. So, that yummy cookie you enjoyed? Its atoms are still around, just in a different configuration, helping you run, jump, and learn!
It’s pretty mind-blowing when you stop and think about it. The Law of Conservation of Mass reminds us that nothing truly disappears. It’s a comforting thought, isn't it? Like knowing that even when something ends, its essence, its "stuff," continues on, just in a new form. It’s the universe’s way of saying, "Don't worry, nothing goes to waste!" And that, my friends, is a kind of magic that’s happening all around us, all the time.
