Which Nuclide Is Listed With Its Half-life And Decay Mode

Okay, so you know how some things just… disappear? Like that sock in the dryer? Or your motivation on a Monday morning? Well, in the wild and wacky world of atoms, things disappear too. But it's not magic, it's radioactivity! And sometimes, scientists are super cool and tell us exactly how fast this disappearing act happens.
We're talking about nuclides here. Think of them as tiny, unstable particles that can't quite make up their minds. They're like the indecisive millennials of the atomic world. And when they decide to change, they go through something called radioactive decay. It's their way of saying, "Peace out, I'm gonna be something else now!"
But here's the kicker: each of these little guys has its own special timer. It's called a half-life. Imagine you have a perfectly ripe avocado. Its half-life is how long it takes for half of that avocado to turn brown and questionable. Same idea, but with atoms! Some half-lives are super, super fast. Others are ridiculously, mind-bogglingly long. Like, longer than you've been alive. Longer than your great-great-great-great-great-grandma was alive. We’re talking geological timescales here, folks!
So, What's the Deal with the "Which Nuclide Is Listed With Its Half-life And Decay Mode" Thing?
Basically, it's a fun little quiz the universe gives us. When scientists are messing around with these unstable atoms, they gotta keep track. So they'll often say, "Alright, we've got this nuclide here. It's called [insert funny-sounding name here], and guess what? It's gonna half its radioactivity in [insert super short or super long time here]. And how's it gonna go out? Oh, it's doing a beta decay! How dramatic!"
It's like a little atomic dating profile. "Hi, I'm Carbon-14. My half-life is 5,730 years. I mostly do beta decay. Looking for stable isotopes to settle down with." Okay, maybe not exactly like that, but you get the vibe. It’s about identifying these specific atomic personalities.
Why Is This Even Fun to Talk About?
Because it’s full of surprises! You think atoms are just… there? Nope! They’re doing little dance routines, emitting particles, and transforming like tiny shape-shifters. It’s a constant performance happening all around us, even if we can't see it.

And the decay modes? Oh boy. We're not just talking one way to fizzle out. There's alpha decay, where it’s like the atom is shedding a tiny, helium-like chunk of itself. It’s like it’s saying, "Too heavy, gotta lighten the load!"
Then there's beta decay. This one’s a bit more nuanced. It can be a beta-minus decay, where a neutron basically turns into a proton and spits out an electron. Poof! New element, courtesy of a rogue electron. Or it could be a beta-plus decay, where a proton turns into a neutron and ejects a positron (the electron's anti-matter twin – mind-bending, right?). It's like the atom's internal plumbing is getting a little… creative.
And don't forget gamma decay. This one's like the atom is just too excited and needs to let off some steam. It’s not changing its identity, just shedding excess energy in the form of a high-energy photon. Think of it as the atomic equivalent of a toddler after too much sugar – lots of wiggles and jitters!

Each nuclide has its own preferred method of saying goodbye to its unstable state. It’s like picking your favorite way to exit a party – some sneak out the back, others make a grand pronouncement. Some nuclides are just… divas of disintegration!
Quirky Facts and Funny Details to Tickle Your Atomic Fancy
Did you know that Carbon-14, the one with that 5,730-year half-life? That's the stuff used for radiocarbon dating! It’s how we figure out how old mummies or ancient pottery are. So, the next time you’re looking at an ancient artifact in a museum, you can thank Carbon-14's predictable decay for telling you its story. Pretty neat, huh?
And then there’s Uranium-238. This bad boy has a half-life of 4.5 billion years! That's older than the Earth itself! It’s still hanging around, slowly decaying, contributing to the heat deep within our planet. It’s like the ultimate grandparent of the elements, still around to tell its ancient tales.

What about something with a super short half-life? Take Polonium-214. Its half-life is a mere 164 microseconds. That's 164 millionths of a second! It decays faster than you can say "radioactivity." It's the Usain Bolt of the atomic world, gone in a flash. It’s so fleeting, it’s almost like a magic trick that happens too fast to catch.
And the decay modes can be a bit of a family affair. Sometimes, one nuclide decays into another, which then decays into another. It’s a whole chain reaction! Like a radioactive domino effect. It's a testament to how interconnected and dynamic the atomic realm truly is.
It’s these specific pairings – the nuclide, its half-life, and its decay mode – that give us so much information. It’s not just random atomic chaos. It’s a predictable, yet endlessly fascinating, system. It’s the universe’s way of saying, "Here's a puzzle, have fun with it!"
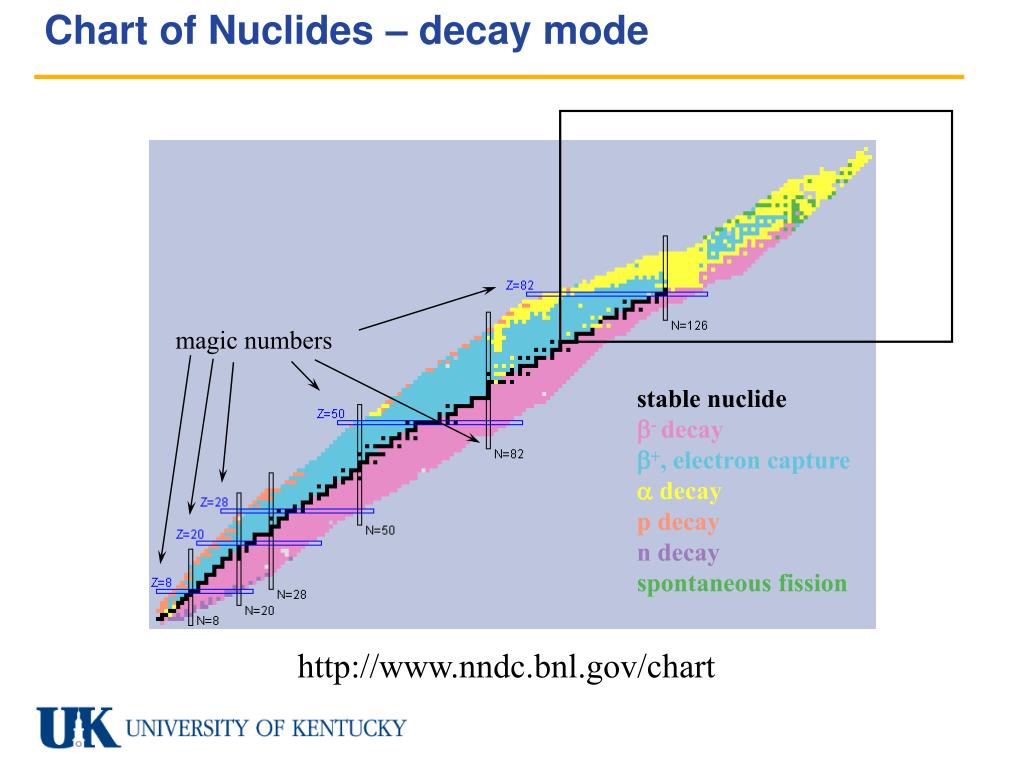
Why This Topic Should Spark Your Curiosity
Because it’s everywhere! Radioactivity is a fundamental part of our universe. It’s in the rocks beneath our feet, in the air we breathe, and even in some of the food we eat (in tiny, harmless amounts, don't worry!). Understanding these nuclides and their habits helps us understand everything from the age of the Earth to how nuclear power works.
Plus, it's just plain cool. The idea that tiny particles are constantly transforming, emitting energy, and following these precise rules is mind-blowing. It’s like a secret language the universe speaks, and scientists are slowly but surely learning to translate it.
So next time you hear about a nuclide with its half-life and decay mode, don't just nod and pretend you understand. Lean in! Ask what kind of decay it is. Wonder about how long that half-life is. Because behind those scientific terms is a universe of tiny, energetic characters with some pretty wild stories to tell. It’s a reminder that even the smallest things can have the biggest impacts, and that the universe is always, always changing.
