Which Monomer Units Combine To Form Nucleic Acids

Hey there, curious minds! Ever wondered what makes up the amazing building blocks of life? We're talking about the stuff that carries all your instructions, from what color your eyes are to how your body works. It’s pretty mind-blowing stuff, and today we're going to peek behind the curtain at how it all gets put together.
Think of it like a super-secret recipe. Just like you need flour, sugar, and eggs to make a cake, living things need special ingredients to build their instructions. These ingredients are called monomer units. They are the tiny, repeating pieces that link up to form much larger, more complex structures. Without these little guys, life as we know it just wouldn't happen!
The star players in our story are the nucleic acids. You might have heard of them! The most famous ones are DNA and RNA. These are the master blueprints of your body. They hold all the information passed down from your parents and tell every single cell what to do.
So, what are these amazing nucleic acids made of? They are like long chains, and the individual links in those chains are our special monomer units. Imagine a LEGO set – you have all these little bricks that snap together to build something big. Nucleic acids are just like that, but way, way more intricate and important.
The "Nucleotides": Our Tiny Stars
The specific name for the monomer units that make up nucleic acids is nucleotides. Say that ten times fast! These nucleotides are the fundamental building blocks, the absolute must-haves for DNA and RNA.
Now, what makes a nucleotide so special? It's not just one thing; it’s actually three parts working together perfectly. Think of it as a tiny team, each member having a vital role. When these three parts come together, voilà – you have a nucleotide!
Let’s break down this little team. First up, we have a sugar. But not just any sugar – it’s a special kind called a pentose sugar. Pentose just means it has five carbon atoms, which is a key structural feature. It's like the backbone or the frame of our nucleotide.
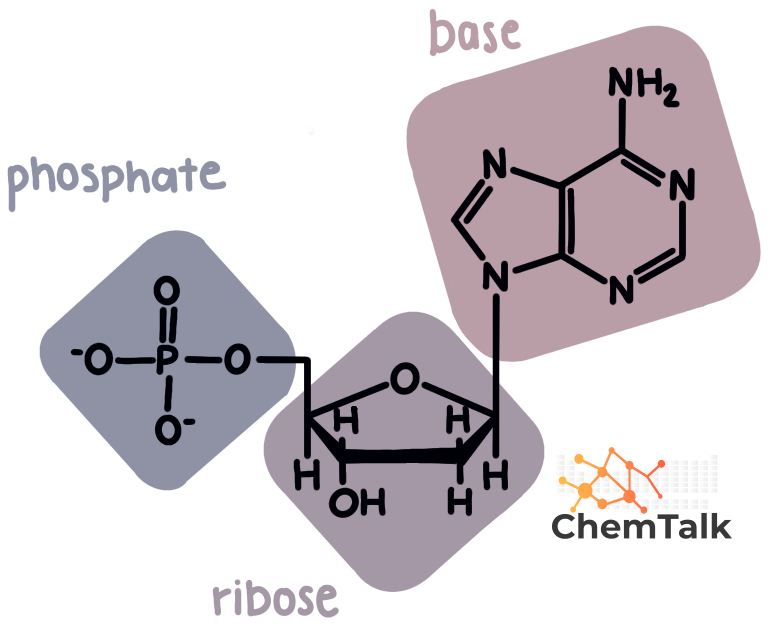
Next on the team is a phosphate group. This is a molecule containing phosphorus and oxygen. It’s pretty energetic and plays a crucial role in linking the nucleotides together. Think of it as the glue that holds our chain strong and stable. This phosphate group is what gives nucleic acids their "acidic" nature.
And finally, the most exciting part of the nucleotide team: the nitrogenous base. This is where things get really interesting and unique. These bases are made up of nitrogen atoms and carbon atoms, and they come in different flavors, each with its own personality.
The Colorful Cast of Nitrogenous Bases
The nitrogenous bases are the ones that carry the actual "code" or "letters" of our genetic information. They are like the alphabet of life, and there are five main characters in this play.
We have two main families of these bases. The first family is called purines. These are larger bases with a double-ring structure. The stars in this group are Adenine (A) and Guanine (G). They are like the bold, leading characters in our genetic story.
Then we have the second family, called pyrimidines. These bases are a bit smaller, with a single-ring structure. The key players here are Cytosine (C), Thymine (T), and Uracil (U). They are just as important, but they have a different structural style.

Here's a fun little twist! In DNA, you’ll find Adenine (A), Guanine (G), Cytosine (C), and Thymine (T). They are the classic quartet for our DNA instructions.
But in RNA, things change ever so slightly. RNA uses Adenine (A), Guanine (G), and Cytosine (C), just like DNA. However, instead of Thymine (T), it uses Uracil (U). It's like a slightly different dialect of the same language, perfectly suited for its job.
So, to recap, a nucleotide is made of a sugar, a phosphate group, and a nitrogenous base. And these bases are our A, T, C, G, and sometimes U!
How They Link Up: The Amazing Chain Reaction
Now for the really cool part: how do these nucleotides get together to form those long chains of DNA and RNA? It's a bit like zipping them up, but with a chemical bond.
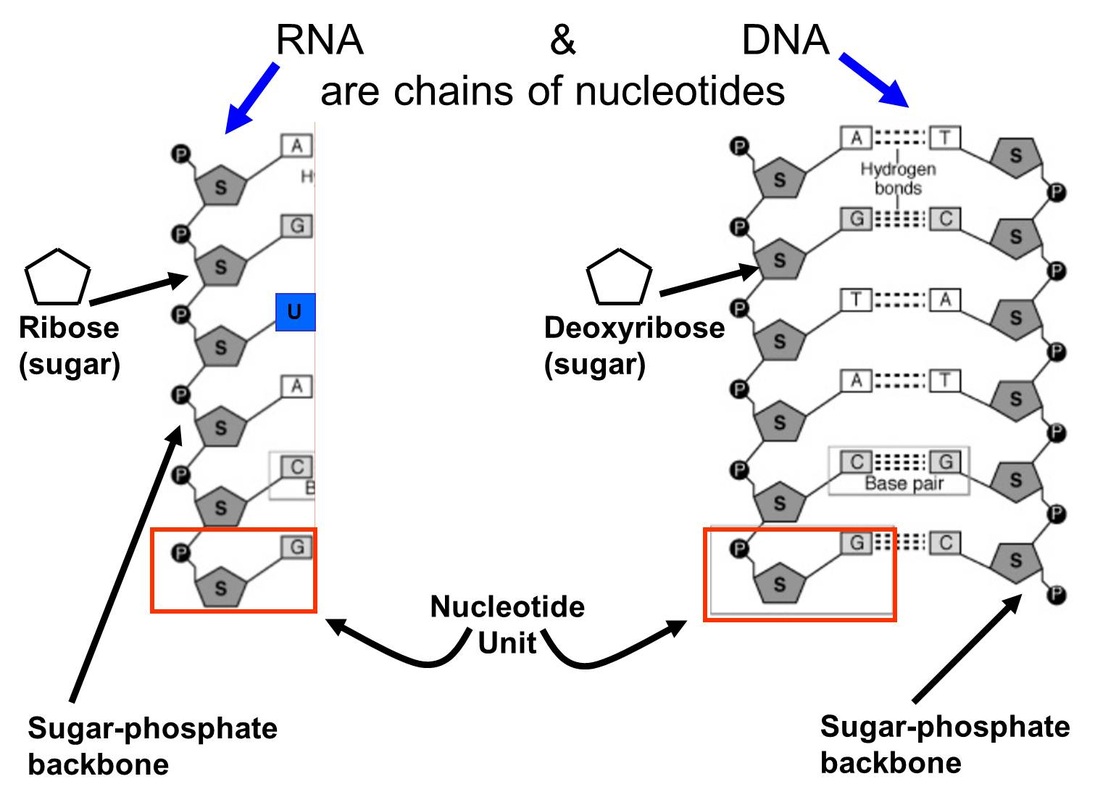
The phosphate group from one nucleotide connects to the sugar of the next nucleotide. This creates a strong, repeating backbone. Think of it as a long string of pearls, where the pearls are the nucleotides and the string is made of those linked sugars and phosphates.

This linkage is called a phosphodiester bond. Fancy name, right? It's a very strong chemical bond that ensures the DNA and RNA chains stay intact. They are built to last, carrying their precious information reliably.
And the nitrogenous bases? They hang off this backbone like little flags. In DNA, these flags on one strand pair up with specific flags on another strand. Adenine (A) always pairs with Thymine (T), and Guanine (G) always pairs with Cytosine (C). It's a very specific and organized dance.
This precise pairing is what allows DNA to be a double helix, like a twisted ladder. The sugar-phosphate chains form the sides of the ladder, and the paired bases form the rungs. It’s a structure of incredible elegance and efficiency.
RNA, on the other hand, is usually a single strand. It doesn't always need a partner in the same way DNA does. This flexibility allows RNA to perform a wider variety of roles within the cell, like carrying messages or helping to build proteins.
Why It's So Special and Entertaining!
What makes this all so entertaining? It’s the sheer ingenuity of nature! Think about it: simple little molecules, just a few types of each part, coming together in specific sequences to create the incredibly complex instructions for every living thing on Earth.
It’s like a universal language. All life, from the tiniest bacteria to the largest whale, uses these fundamental nucleotide units to build its genetic code. It’s a testament to the power of simplicity and repetition in creating complexity.
And the variations! With just four main letters in DNA (A, T, C, G) and four in RNA (A, U, C, G), the number of possible combinations is astronomically huge. This allows for all the diversity of life we see. It's like a massive, infinite library, all built from the same basic alphabet.
The way these bases pair up is also fascinating. It's like a lock and key mechanism. A only fits with T (or U in RNA), and G only fits with C. This specificity is crucial for accurate copying of genetic information when cells divide.
Understanding how these monomer units, the nucleotides, combine to form nucleic acids, like DNA and RNA, is like understanding the fundamental code of life itself. It’s the basis of heredity, evolution, and all biological processes.
So, the next time you hear about DNA or RNA, remember these amazing little building blocks: the nucleotides, with their sugary core, phosphate glue, and the wonderfully diverse nitrogenous bases. They are the silent architects of life, and their story is one of the most entertaining and important tales in the universe!
