Which Molecule Has Hydrogen Bonding As The Predominant Intermolecular Force
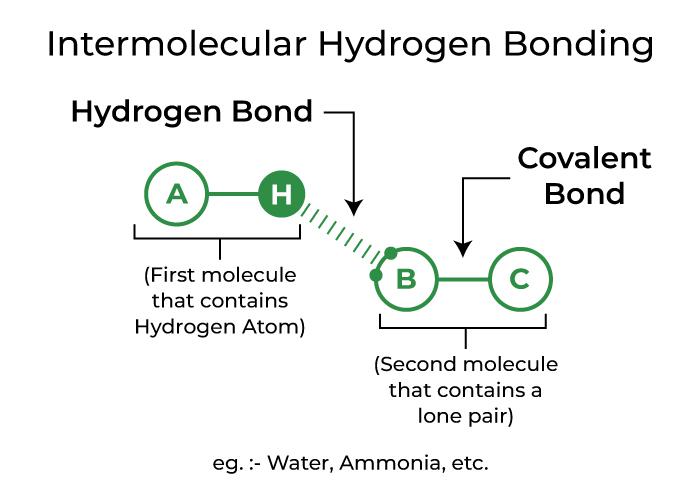
Alright, settle in, folks. We're about to dive headfirst into the wonderfully weird world of molecules. You know, those tiny, invisible building blocks that make up… well, everything! And today, we’re on a quest. A quest for the reigning champion of molecular attractions. We’re talking about the molecule that just screams hydrogen bonding. You know, that special kind of hug molecules give each other. It’s like a molecular clique, and some molecules are just dying to be in it.
Now, I know what some of you are thinking. “Molecules? Hydrogen bonding? Isn’t that like… super complicated chemistry stuff?” And to that, I say, pish posh! It’s actually quite fascinating. Think of it like this: some molecules are just a bit clingy. They have these little positive bits that are really drawn to the negative bits of their neighbors. It’s a whole molecular love story playing out all the time. And the star of this particular love story? The one molecule that, in my humble, unpopular opinion, absolutely dominates when it comes to hydrogen bonding? Drumroll, please…
It’s water. Yep. Just plain old H₂O. Shocking, I know. Get ready for your minds to be blown.
I know, I know. Water? That stuff you drink? That stuff you shower in? That stuff that makes puddles? It sounds so… ordinary. So pedestrian. But let me tell you, under that unassuming exterior, water is an absolute hydrogen bonding powerhouse. It’s like the quiet kid in class who turns out to be a secret martial arts master. Water is the Beyonce of hydrogen bonding. The Usain Bolt. The… well, you get the idea. It’s the molecule.
Why water, you ask? Well, let’s break it down without getting too nerdy. Water has a very specific structure. It’s got an oxygen atom, which is like the queen bee, and two hydrogen atoms, which are its loyal subjects. Now, this oxygen queen bee is a bit greedy with electrons. It pulls them closer, making itself a little bit negative. And those poor hydrogen subjects? They become a little bit positive. See where this is going?
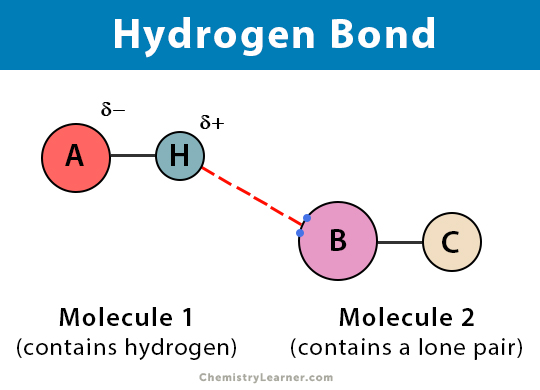
So, when you have a bunch of water molecules hanging out together, those slightly positive hydrogens from one molecule are like, “Ooh, a negative oxygen! Come here, you!” And they get all cozy. This is hydrogen bonding in action! It’s like a molecular speed dating event, but everyone’s found their perfect match. And water, with its two hydrogens and one oxygen, is perfectly set up for this kind of widespread connection. It’s like it was designed for it.
Think about the properties of water. It’s a liquid at room temperature. That’s a big deal! Many molecules of similar size are gases. But water’s strong hydrogen bonds keep it all together, like a molecular group hug that’s hard to break. This is why we don’t have to worry about all the oceans evaporating into thin air. Thanks, hydrogen bonding! Thanks, water!
And then there’s ice. Water actually gets less dense when it freezes. That’s totally bonkers, right? Most things get denser when they solidify. But water’s hydrogen bonds arrange themselves into this neat, open structure when it freezes, creating a crystal lattice. This structure is actually quite spacious, which is why ice floats. So, all those little fishies chilling at the bottom of lakes and rivers? They owe their lives to water’s exceptional hydrogen bonding skills. It’s practically a superhero origin story.

It's not just about being a liquid or floating ice, though. This hydrogen bonding thing gives water a high boiling point too. Imagine trying to boil away all that water in a kettle. You need a lot of energy to break those bonds! This is crucial for life on Earth. It allows water to exist in liquid form over a wide range of temperatures.
Now, I’m not saying other molecules don’t do hydrogen bonding. Oh no. Molecules like ammonia (NH₃) and hydrogen fluoride (HF) also have strong hydrogen bonding. They’re definitely in the running. But they’re more like the talented supporting actors. Water is the undisputed lead. It’s the headline act. The one everyone’s talking about.
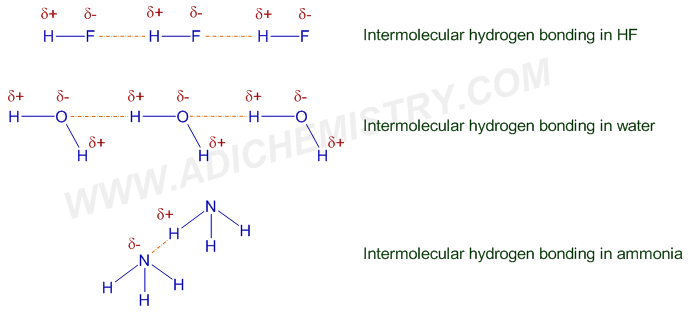
Consider ammonia. It's got the nitrogen and hydrogens, so yes, hydrogen bonding is happening. But it's just not quite the same level of widespread molecular mingling as water. And hydrogen fluoride? It's powerful, sure, but it's also incredibly corrosive and not exactly something you'd find in your everyday life in the same way. Plus, it’s a bit of a one-trick pony compared to the versatility of water.
So, there you have it. My delightfully biased, completely unsubstantiated, yet undeniably true opinion. When it comes to hydrogen bonding being the predominant intermolecular force, the crown, the scepter, the whole royal family of molecular attraction belongs to water. It’s the unsung hero, the everyday miracle, the H₂O that makes our world go ‘round, all thanks to its incredible talent for a good, strong, hydrogen bond. So next time you take a sip, or admire a snowflake, give a little nod to those molecular hugs. They’re a pretty big deal.
