Which Molecule Has A Structure That Is Most Like Aspirin
Ever wondered about the tiny building blocks that make up everything around us? It's a bit like a giant LEGO set, but with molecules! And today, we're going to have some fun exploring which molecule has a structure that's a bit like our old friend, Aspirin. This isn't just for science buffs; understanding these molecular shapes can be surprisingly useful and even a bit of a brain teaser!
For those just dipping their toes into the world of chemistry, figuring out molecular structures is like learning to recognize faces. You start to see patterns and similarities. For families, it's a fantastic way to spark curiosity in kids. Imagine explaining that the medicine you take for a headache shares a structural family with other things! Hobbyists, especially those interested in things like home brewing or even gardening, might find that a little molecular knowledge can explain a lot about how certain processes work.
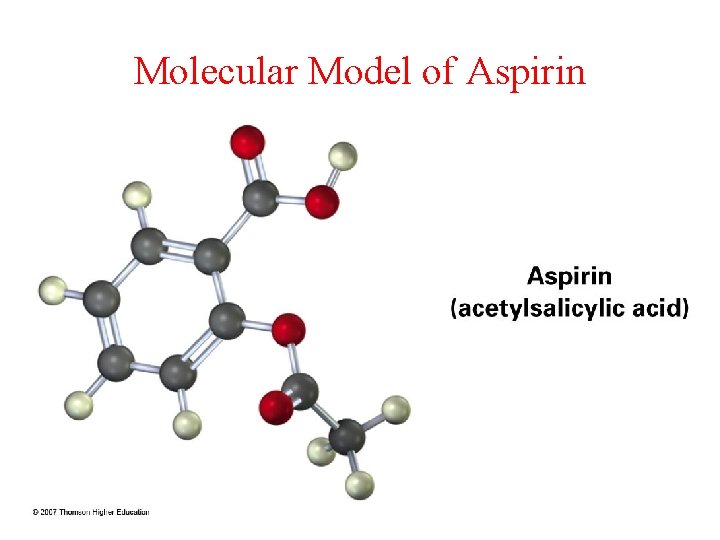
So, what's so special about Aspirin's structure? Aspirin, or acetylsalicylic acid, has a few key features. It has a benzene ring – a hexagon-shaped arrangement of carbon atoms – with some interesting bits attached. When we look for molecules that are "most like" Aspirin, we're often looking for other molecules that also contain this benzene ring and have similar functional groups attached. Think of it like finding a sibling in the molecular family – they share common traits!
One of the closest relatives you might find is salicylic acid itself. Surprised? Salicylic acid is what Aspirin is made from. It's very similar, just missing a small piece called an acetyl group. Another example, though a bit more distant, could be other phenols or compounds with similar aromatic rings. These molecules might not be medicines, but their structural similarities can influence how they behave and interact with other substances.

Getting started with this is easier than you think! You don't need a lab coat. A great first step is to look up images of molecular structures online. Websites like Wikipedia or educational chemistry sites have vast libraries. Search for "aspirin structure," "salicylic acid structure," and then try searching for "benzene ring" to see the basic hexagonal shape. You'll start to notice the common elements. You can even find 3D models that let you rotate and zoom in, making it feel like you're holding the molecule in your hands!
It's a simple exercise, but it opens up a fascinating window into the organized chaos of the molecular world. Seeing how even small changes in structure can lead to different properties is incredibly rewarding. So, next time you reach for that familiar white pill, remember the intriguing molecular family it belongs to. It's a fun little puzzle, and the more you look, the more connections you'll find!
