Which Lewis Electron-dot Diagram Represents The Bonding In Potassium Iodide
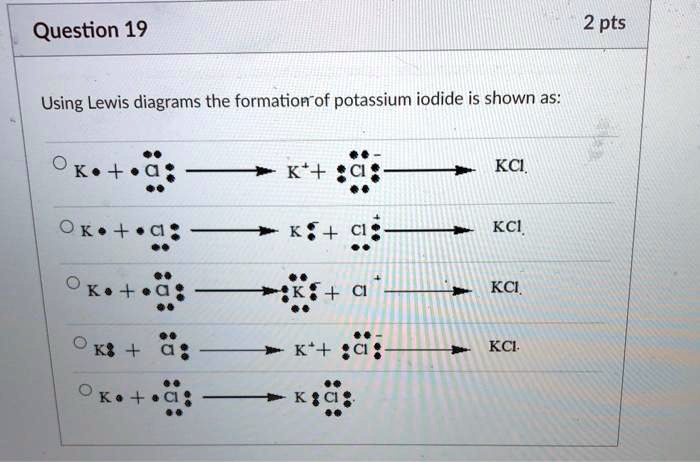
Hey there, fellow humans! Ever looked at something and thought, "Wow, that's neat!"? Today, we're going to peek at something super neat, even if it sounds a bit science-y at first. We're talking about the magical little dance that atoms do when they decide to become friends. Specifically, we're diving into the bonding in a compound called Potassium Iodide. Don't worry, it's not as complicated as deciphering your teenager's text messages!
Think of atoms as tiny, tiny LEGO bricks. They're the fundamental building blocks of everything around us – your comfy chair, that delicious cookie you had (or are planning to have!), and even the air you're breathing. These LEGO bricks have little "sticky bits" on their outsides, which are called electrons. These electrons are like the connectors on the LEGOs, and they're really, really important for how atoms join up.
Now, imagine some LEGO bricks are super happy with the number of sticky bits they have, while others are desperately trying to get more or get rid of a few. It's a bit like us, right? Sometimes we feel like we have too much on our plate and want to give some away, and other times we wish we had a few more helping hands. Atoms are the same way with their electrons!
When atoms decide to bond, they're essentially trying to achieve a state of happy stability. They want their outer "sticky bit" layer to be full, kind of like a full deck of cards or a perfectly organized toolbox. This is where our friend, the Lewis Electron-dot Diagram, comes in. It's like a little cartoon of these atoms, showing us exactly where their important outer electrons are and how they're interacting.
So, what is Potassium Iodide? It's a compound, meaning it's made of two different types of atoms that have decided to stick together. In this case, we have Potassium (symbolized as K) and Iodine (symbolized as I). Think of them as two different kinds of LEGOs that found each other and decided to build something awesome together.
Let's break down our players. Potassium (K) is a metal. Metals are often like those generous friends who have a spare electron they're just itching to share. Potassium has one extra electron in its outer shell. It's like having one extra ticket to a concert – you'd be happy to give it away to someone who really wants it!
On the other hand, we have Iodine (I). Iodine is a non-metal. Non-metals are often a bit like those friends who are always looking for an extra hand. Iodine has seven electrons in its outer shell and is just one electron short of being perfectly happy and full. It's like needing just one more ingredient to bake that perfect cake.
Now, here's where the magic happens! Potassium, with its extra electron, looks at Iodine, which is desperately needing one. It's a perfect match, like peanut butter and jelly, or socks and sandals (okay, maybe not that last one!). Potassium thinks, "Hey, Iodine, you look like you could use this extra sticky bit. Take it! I'll be happier without it, and you'll be happier with it!"

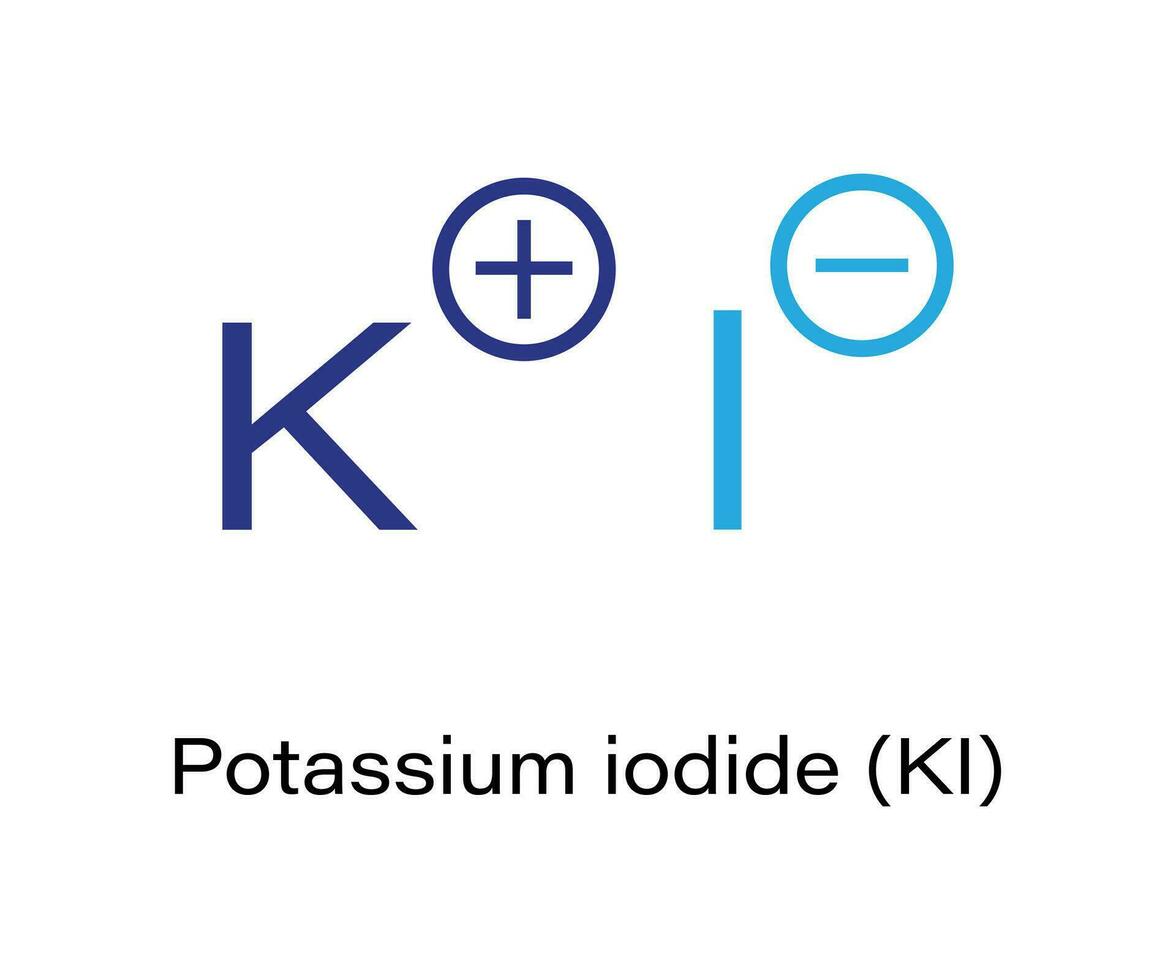
So, Potassium gives its one outer electron to Iodine. This act of giving and taking is a super common way for atoms to bond, and it's called ionic bonding. It's like a handshake, but with electrons! When Potassium gives away its electron, it becomes a positively charged ion (think of it as having a little "+" sign because it lost a negative charge). And when Iodine accepts that electron, it becomes a negatively charged ion (it gets a little "-" sign because it gained a negative charge).
Now, you've got a positive Potassium and a negative Iodine. What happens when you have opposite charges? They stick together! It's like how the positive and negative ends of a magnet attract each other. This strong attraction between the positive Potassium ion and the negative Iodine ion is what holds Potassium Iodide together.

A Lewis Electron-dot Diagram shows us this perfectly. You'll see the Potassium atom represented with its symbol (K) and its one outer electron. Then, you'll see the Iodine atom with its symbol (I) and its seven outer electrons. When they bond, the diagram will show the Potassium with its electron GONE, often represented with a positive charge (K+). And the Iodine will be shown with eight outer electrons, and a negative charge (I-).
Imagine a little cartoon: Potassium is a cheerful character with a single, glowing orb (its electron) that it happily hands over. Iodine is a character with seven orbs, looking a bit stressed, and when it receives the orb from Potassium, it now has eight glowing orbs and a big, contented smile (symbolized by the negative charge).
Why should you care about this electron-dot diagram thing? Well, this simple way of showing bonding helps us understand why things behave the way they do. Potassium Iodide, for example, is a common salt used in things like animal feed and even in some medical treatments to prevent thyroid problems. Understanding how it's put together, at this atomic level, is the first step to figuring out its properties and uses.

It's like understanding how a lock and key work. You see the shape of the key (the electron configuration and how they bond), and you understand how it fits into the lock (the resulting compound's properties). Without that understanding, the lock just looks like a confusing metal object.
So, when you see a Lewis Electron-dot Diagram for Potassium Iodide, you're looking at a visual story of an atom giving a little something away and another atom happily receiving it, leading to a strong, stable partnership. It's a fundamental principle of chemistry, and it explains so much of the world around us. It's the tiny, invisible dance that makes our solid world possible!
Next time you encounter a Lewis Electron-dot Diagram, don't be intimidated! Think of it as a secret code, a helpful little drawing that tells a fascinating story about atoms making friends. It's a beautiful illustration of how even the smallest things in the universe are constantly interacting and creating the complex and wonderful world we live in. Pretty neat, right?
