Which Lewis Electron Dot Diagram Is Correct For Co2
Hey there, science explorers! Ever looked at a molecule and wondered, "What's going on in there?" Like, what are those atoms really up to? Well, today we're diving into a super fun puzzle: the Lewis electron dot diagram for carbon dioxide, or CO2. Sounds fancy, right? But trust me, it's like a little detective story for your brain, and figuring out the right one can be incredibly satisfying. Think of it as a tiny, microscopic game of Lego, where the bricks are electrons!
You know, sometimes in science class, things can feel a bit… dry. Lots of rules, lots of memorizing. But when you get to the heart of it, it's all about understanding how the universe works. And what's cooler than that? So, when we talk about Lewis structures, we're really just talking about how atoms share their outer electrons to become happy and stable. It’s like they’re holding hands, forming little chemical friendships. And CO2? It’s a classic! Let's crack the code together.
The CO2 Conundrum: Which Picture is Worth a Thousand Bonds?
So, we've got carbon (C) and two oxygens (O). The big question is, how do they arrange their electrons to be the most stable? We've got options, and that’s where the fun begins! Imagine you’re trying to arrange little candies. You have a certain number, and you need to make them look just right. That’s essentially what we’re doing with electrons.
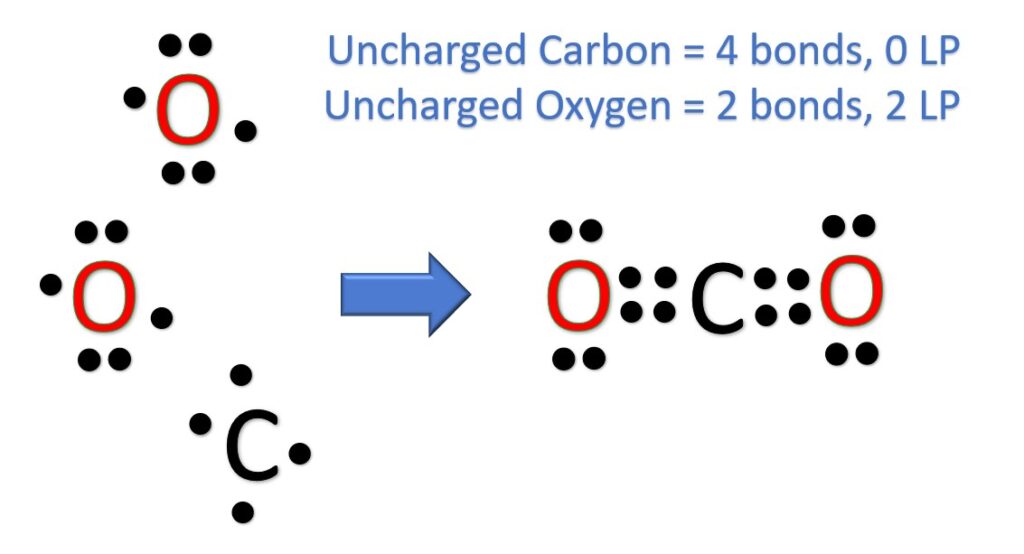
First things first, let’s count our valence electrons. These are the electrons in the outermost shell, the ones that do all the bonding action. Carbon is in Group 14, so it’s got 4 valence electrons. Oxygen is in Group 16, so it brings 6 valence electrons to the party. Since we have two oxygens, that’s 2 * 6 = 12 electrons from the oxygens. Add in the carbon's 4, and voilà, we have a grand total of 16 valence electrons to play with!
Now, the basic idea of a Lewis structure is that each atom wants to have a full outer shell, usually meaning 8 electrons (the octet rule, a.k.a. the "happy electron club"). It’s like they’re all aiming for that perfect score. So, we need to arrange these 16 electrons in a way that satisfies everyone.
Option 1: The "Too Much Sharing" Scenario
Let’s say we try to give carbon a single bond to each oxygen. So, O-C-O. That uses up 4 electrons (two for each single bond). We have 12 electrons left. We can then try to give each oxygen 6 more electrons to complete their octet. That uses up another 12 electrons. Uh oh! We've used all 16 electrons, but look at carbon – it only has 2 electrons from each bond, so it’s only got 4. Not a happy camper at all!
This is where you’d say, "Hmm, that doesn't look right." And you'd be absolutely correct! This arrangement simply doesn't fulfill the octet rule for carbon. It’s like trying to build a tower with not enough blocks at the base – it’s just not going to stand up.

Option 2: The "One Double, One Single" Shuffle
Okay, let's try something a bit more adventurous. What if we make one bond a double bond? Let's try O=C-O. This uses 6 electrons (4 for the double bond, 2 for the single bond). We have 10 electrons left. Let's give the double-bonded oxygen 4 more electrons to complete its octet. It now has 4 + 4 = 8. Good! Now, let's give the single-bonded oxygen 6 more electrons. It now has 2 + 6 = 8. Also good! But look at carbon. It has 4 electrons from the double bond and 2 from the single bond, totaling 6. Still not a full octet for carbon. This is better, but not the best.
This option feels closer, doesn't it? You can almost feel the atoms getting a little happier, but there's still a slight unease. Like when you’re almost done with a puzzle, but there’s one piece missing. It’s a near miss!

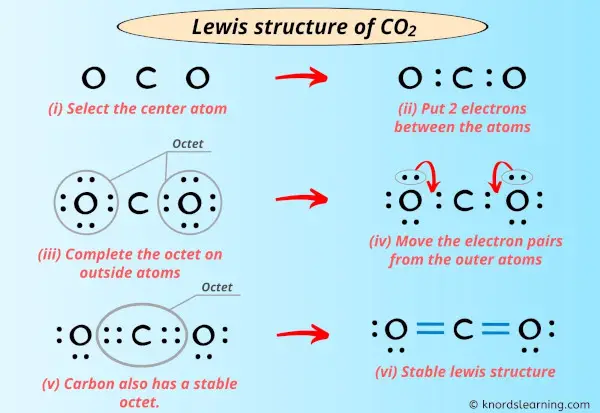
Option 3: The "Perfectly Balanced" Duet (of Doubles!)
Now for the grand finale! What if we try making both bonds double bonds? That means O=C=O. This uses up 8 electrons (4 for each double bond). We have 16 - 8 = 8 electrons left. Let's distribute these remaining electrons. We give 4 electrons to each oxygen atom. So, each oxygen now has 4 electrons from the double bond and 4 lone pair electrons, totaling 8. Perfect! And carbon? It has 4 electrons from the left double bond and 4 electrons from the right double bond, totaling 8. Hooray!
This is it! This is the winning formula. Both oxygens have their octets, and carbon has its octet. All 16 valence electrons are accounted for, and everyone is as stable and happy as can be. This is the Lewis electron dot diagram that accurately represents the bonding in CO2. It's a beautifully symmetrical arrangement, showing that carbon is sharing its electrons equally with both oxygen atoms through double bonds.
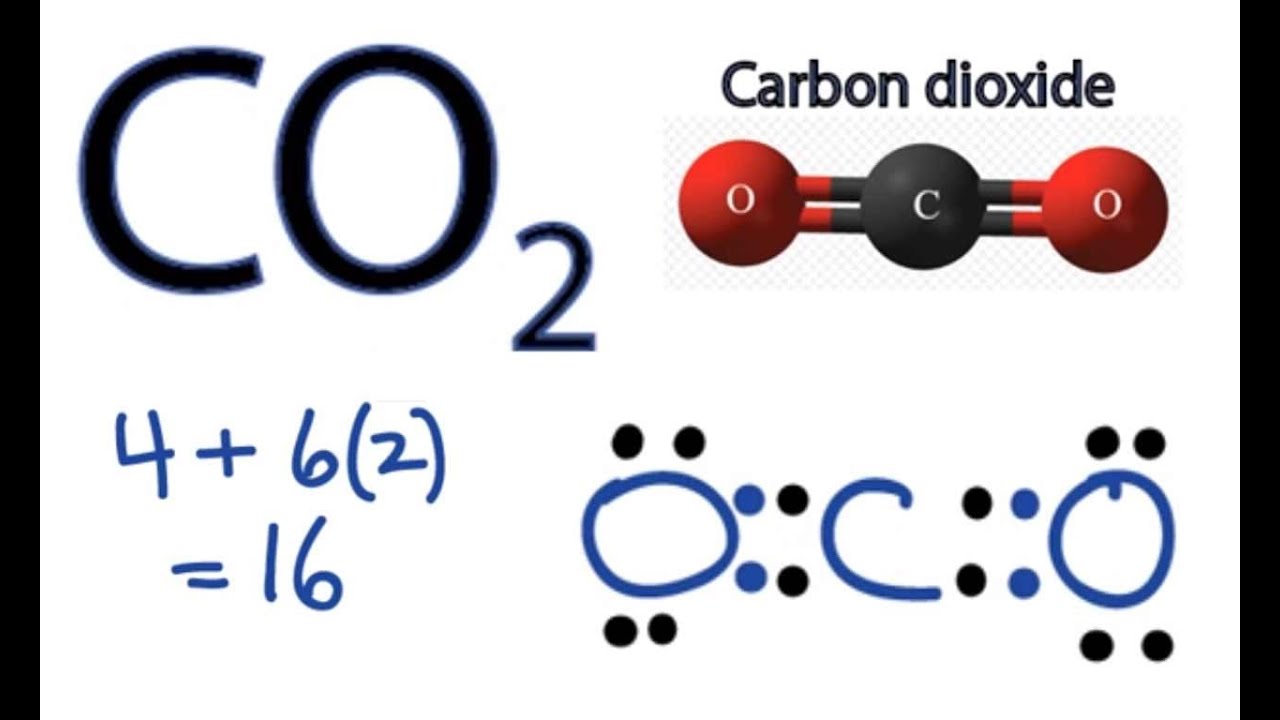
Why This Stuff is Actually Fun!
See? It’s not just about drawing dots. It’s about solving a little chemical puzzle! Each molecule has its own unique personality, defined by how its atoms arrange their electrons. And understanding these arrangements helps us predict how molecules will behave, how they'll react, and what amazing things they can do. From the air we breathe (CO2 is a big part of that, believe it or not!) to the fuels that power our world, Lewis structures give us a peek into the invisible architecture of matter.
Think about it: every time you see a beautiful flower, every time you take a breath, you're experiencing the results of these tiny electron dances. It’s a constant, unseen ballet of atoms and electrons, and learning about Lewis structures is like getting a backstage pass to the most incredible show in the universe!
So, the next time you encounter a molecule, don't just see it as a jumble of letters. See it as an opportunity for discovery. See it as a chance to understand the fundamental forces that shape our reality. Embrace the challenge, enjoy the process, and remember that even the smallest electron can make a big difference. Keep asking questions, keep exploring, and you'll find that the world of chemistry is not only fascinating but also incredibly inspiring!
