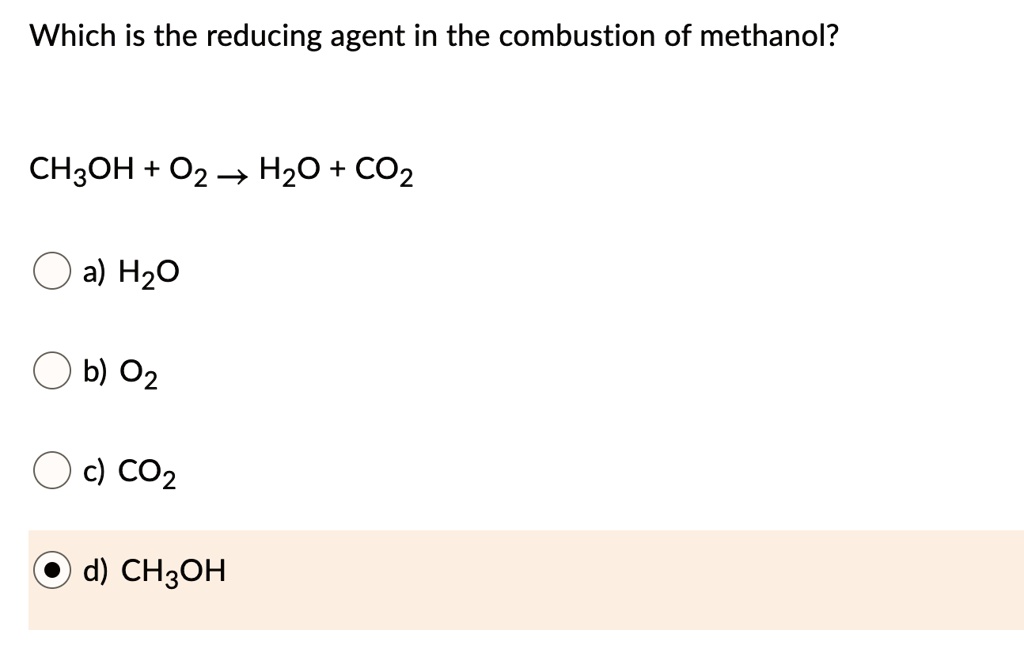
Which Is The Reducing Agent In The Combustion Of Methanol
Alright, let's chat about something that sounds super science-y but is actually happening all around us, even when we're just trying to get that morning caffeine fix. We're talking about combustion, specifically, the combustion of methanol. Now, don't let the word "combustion" scare you off like a surprise pop quiz in high school chemistry. Think of it more like a really enthusiastic hug between molecules, where stuff gets lit up and transformed. And at the heart of this fiery fiesta, we've got a star player – the reducing agent. It's like the unsung hero of the whole burning operation.
Imagine you're at a backyard barbecue. You've got the grill going, the burgers are sizzling, and the air is thick with delicious smoke. Now, the charcoal or the gas in your grill is basically the fuel, right? It's the stuff that's going to burn. But for it to burn, it needs something to team up with. It needs an oxidizer. In our backyard scenario, that's usually the oxygen from the air. The oxygen is like the enthusiastic cheer squad, pushing the fuel to give its all and unleash all that glorious heat and light.
So, in the case of methanol, which is a type of alcohol – think of the stuff they might use in some portable stoves or even as a fuel additive – it's also looking for its dance partner. And that dance partner, the thing that helps methanol burn and get all energized, is our good old friend, oxygen. But the question we're digging into isn't about the oxygen (which is actually the oxidizing agent, the stern but necessary instructor in this class). We're interested in the other guy, the one who gets a bit of a makeover during the process. That, my friends, is our reducing agent.
Let's break down this "reducing agent" thing. It sounds a bit like something is getting less of something, and in a way, it is! Think of it like a makeover. Someone comes in, maybe a bit rough around the edges, and through the whole process, they get cleaned up, changed, and sometimes even shed a few things. In a chemical reaction, a reducing agent is the molecule that donates electrons. Electrons are like tiny, energetic particles that are buzzing around. When one molecule hands over these electrons to another, it's like it's giving away some of its energy, its "stuff," if you will. And in return, it gets "reduced." Reduced, in this context, often means gaining hydrogen atoms or losing oxygen atoms. It's a chemical glow-up!
So, when methanol (which has the chemical formula CH₃OH, just a little heads-up) decides to have its big combustion party, what's it doing? It's reacting with oxygen (O₂). The methanol itself is the star of the show that's going to get burned. And during this burning, the methanol molecules are the ones that are giving up electrons. They are the ones that are being oxidized, in the grand scheme of things. But the term "reducing agent" refers to the molecule that causes reduction to happen to something else. It's a bit of a tongue-twister, I know! It’s like saying, "This guy is the coach who makes the other team lose points."

In the combustion of methanol, the methanol itself is the reducing agent. Why? Because it's the substance that readily gives up its electrons to the oxygen. Oxygen is greedy for those electrons! It's like oxygen is the picky eater at the buffet, and methanol is the generous soul offering up its most delicious morsels. When methanol hands over its electrons, it's getting oxidized (it's losing electrons), but in doing so, it's causing the oxygen to be reduced (the oxygen is gaining electrons).
Let's try another analogy. Imagine you're at a party, and there's a very popular game of musical chairs happening. The chairs are like the electrons. The oxygen is like the person who really, really wants a chair. Methanol is like someone who is happy to give up their chair (their electrons) to get the party going. When methanol gives up its "chair," it's the one getting "burned" (oxidized), but by doing so, it's enabling oxygen to "sit down" (be reduced). It’s a cooperative, albeit energetic, exchange!
The chemical reaction looks something like this: 2 CH₃OH (methanol) + 3 O₂ (oxygen) → 2 CO₂ (carbon dioxide) + 4 H₂O (water)
+Oxygen+(oxidizing+agent).jpg)
See that CH₃OH? That's our methanol. And that O₂? That's our oxygen. During the reaction, the carbon and hydrogen in the methanol are getting rearranged and ultimately form carbon dioxide and water. The methanol molecules are losing hydrogen and gaining oxygen (in the form of CO₂), which is a classic sign of oxidation. But the key here is that methanol is the one providing the electrons that oxygen is grabbing onto. Without methanol offering up its electrons, the oxygen wouldn't have anything to react with so enthusiastically. It would just be floating around, looking for a party to crash.
Think about it this way: if you have a wallet full of cash (electrons) and someone desperately needs money to buy a ticket to the hottest concert in town (to become reduced), you handing over your cash makes you the person who enables their "reduction" (in terms of their financial state, but in chemistry, it’s about electrons!). You are the one who caused them to get that ticket. So, methanol, by readily surrendering its electrons, is the reducing agent. It's the generous giver, the electron philanthropist, the molecule that kickstarts the chemical transformation by offering its goods.
It’s a bit of a funny twist of language, isn’t it? The thing that gets "burned" (oxidized) is also the "reducing agent." It's like the villain in a play who, by being so dramatically evil, actually makes the hero look even more heroic. Methanol is the fuel, the source of energy, the thing we want to burn to get heat. But in the chemical dance, it's also the one facilitating the other molecule's change by giving away its precious electrons. It’s a team effort, a give-and-take, a chemical love story where one molecule sacrifices a bit of itself to make the other one complete… or at least, to make it a different chemical compound!

So, next time you see a flame, whether it's a candle, a gas stove, or even a fancy methanol burner, remember the unsung hero. Remember the molecule that’s not just burning, but is actively participating in a chemical transformation by being the generous donor. It’s the methanol itself, the humble alcohol, that steps up to the plate, offers its electrons, and lets the fiery magic happen. It's a beautiful thing, really, this intricate ballet of electrons and atoms, all happening to bring us warmth, light, and sometimes, a perfectly seared steak.
We often focus on the bright, fiery part of combustion – the heat and light. But behind those dazzling displays are these quiet exchanges, these molecular handshakes. The reducing agent is crucial because it provides the "stuff" (electrons) that the oxidizer (oxygen) needs to complete its own transformation. Without the reducing agent, the oxidizer would just be standing there, like a chef with all the ingredients but no one to cook for. It needs that partner to get the culinary (or chemical) masterpiece going.
So, in the burning of methanol, don't be fooled by the dramatic flames. Look closer, and you'll see the methanol molecules bravely stepping forward, offering their electrons, and in doing so, becoming the essential reducing agent. It’s a testament to how even in the most energetic reactions, there’s a subtle give-and-take, a fundamental exchange that drives the whole show. It’s not just burning; it’s a carefully orchestrated chemical ballet, and methanol is one of its principal dancers, the one who graciously leads.

And if you ever find yourself explaining this to someone, you can use the barbecue analogy. "You know how the charcoal burns? Well, in the case of methanol, the methanol itself is like the charcoal, and it's giving something to the oxygen so they can have their big fiery dance. That 'something' is electrons, and the methanol is the reducing agent because it's the one giving them away." See? Totally relatable, totally makes sense!
It’s easy to think of chemistry as this abstract, disconnected thing, happening in sterile labs with bubbling beakers. But it’s not! It’s in your car engine, it’s in your body, and it's in the simple act of lighting a fire. And understanding concepts like reducing agents helps us appreciate the elegant simplicity and the complex beauty of these everyday occurrences. So, methanol, you magnificent molecule, we salute your electron-donating prowess!
The beauty of chemistry is in these little revelations. You might think of methanol as just a fuel, something that burns. But then you learn that it's also the reducing agent in that process, the one that facilitates the change. It’s a dual role, a secret life of a chemical compound. And that, my friends, is pretty darn cool. It's like finding out your quiet neighbour is actually a secret agent. The drama is all in the electrons!
