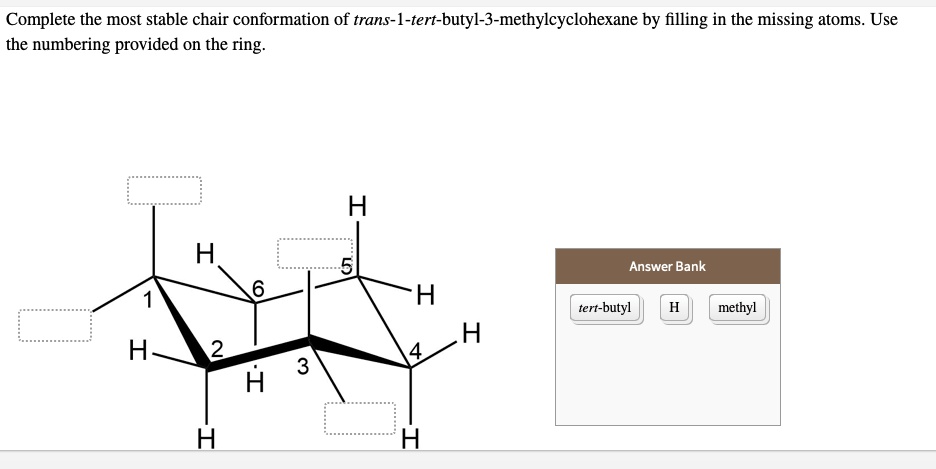
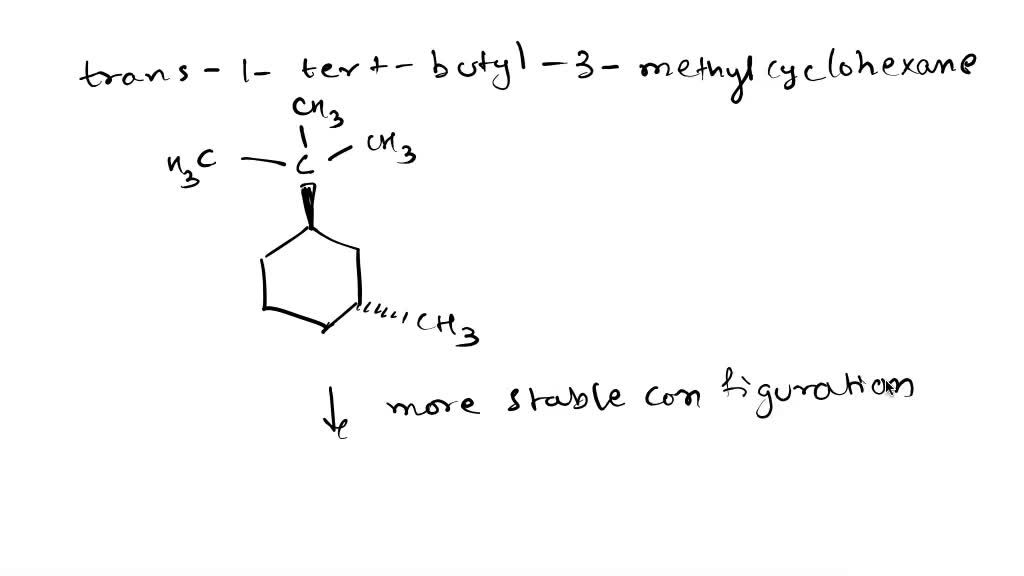
Which Is The Most Stable Chair Form Of Trans-1-tert-butyl-3-methylcyclohexane
Ever wondered about the secret lives of molecules? You know, those tiny building blocks of everything around us? Well, get ready for a little adventure into the surprisingly fascinating world of cyclohexane! Specifically, we're going to chat about a molecule called trans-1-tert-butyl-3-methylcyclohexane. Sounds like a mouthful, right? But stick with me, because this little guy has a bit of a personality, and we're going to uncover which of its chair forms is the king of chill.
Now, when we talk about "chair forms" of cyclohexane, think of it like this: the molecule itself is a ring of six carbon atoms. This ring isn't flat like a dinner plate. Oh no, it loves to bend and twist! The most common and stable way it does this is by folding into a shape that looks remarkably like a comfy armchair. Hence, the "chair form." It's like the molecule is saying, "Ah, this is the best way for me to relax!"
But here's where it gets interesting. Our special molecule, trans-1-tert-butyl-3-methylcyclohexane, has some extra bits attached to its ring. We've got a big, bulky group called a tert-butyl group and a smaller methyl group. These guys love to hang out, and their position on the ring matters. The "trans" in its name tells us how these two groups are positioned relative to each other. Imagine you're looking at the chair. If one group is pointing up, the other is pointing down. They're on opposite sides of the ring's general plane.
So, our molecule can exist in two main chair forms. Think of these as two different ways our armchair-shaped molecule can settle into its comfortable position. In one form, the tert-butyl and methyl groups might be pointing in certain directions. In the other form, they flip around, and their directions change. It's like the armchair can be positioned in slightly different ways, and our guests (the tert-butyl and methyl groups) adjust their seating accordingly.
Now, the big question: which of these two chair forms is the most stable? In the world of molecules, "stable" means they like being in that form the most. It's the most comfortable, the least stressed-out arrangement. It's like finding your favorite spot on the couch – you just naturally gravitate towards it.
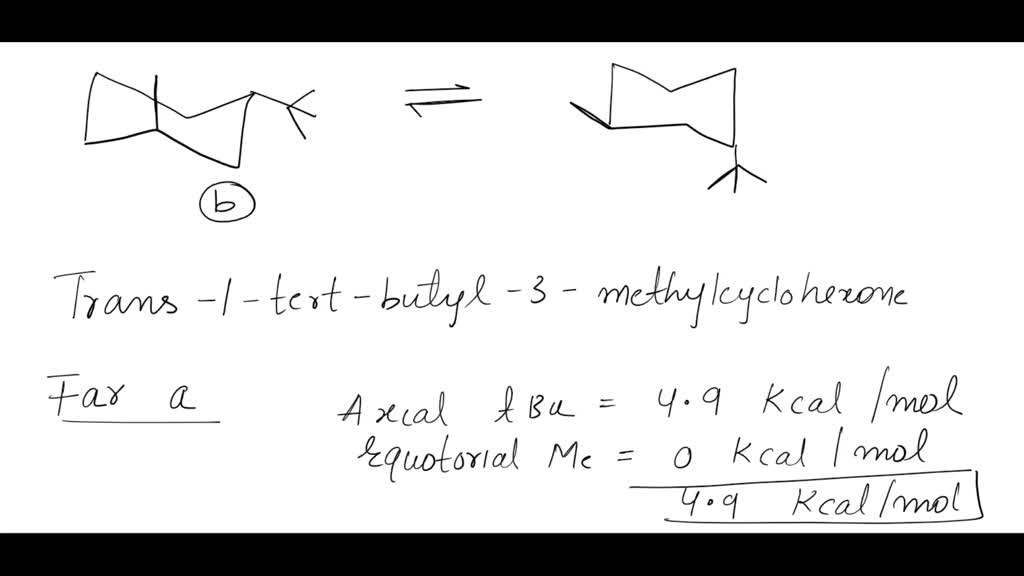
The answer to our molecular puzzle is the chair form where the tert-butyl group is in the equatorial position. What's "equatorial"? Imagine the armchair again. Some parts of the ring are sticking out more or less straight sideways, and some are pointing more up and down. The equatorial positions are those that are sticking out more sideways, like the arms of the chair. The other positions, pointing up and down, are called axial.
So, why is the tert-butyl group preferring the equatorial spot so important? Well, that tert-butyl group is a real heavyweight. It's quite large and takes up a lot of space. When it's in an axial position, it bumps into other parts of the molecule, causing a bit of a crowd and making the whole molecule feel a bit tense and unhappy. It's like trying to squeeze a big cushion into a tight spot on the couch – it just doesn't feel right.
But when that big tert-butyl group is in an equatorial position, it gets to spread out. It has plenty of room to relax, and it doesn't cause as much stress within the molecule. It's like letting that big cushion rest comfortably on the arm of the couch – everyone's happy.
Our little methyl group also plays a role, but it's much smaller. Think of it as a smaller cushion. While it also has preferences for equatorial or axial positions, its influence is less dramatic than that of the giant tert-butyl group. In the most stable chair form, the tert-butyl group is happily in its equatorial spot, and the methyl group also finds a comfortable arrangement, often also equatorial because it's next door and the bulky tert-butyl group helps push it there, but the tert-butyl is the main driver of this stability.
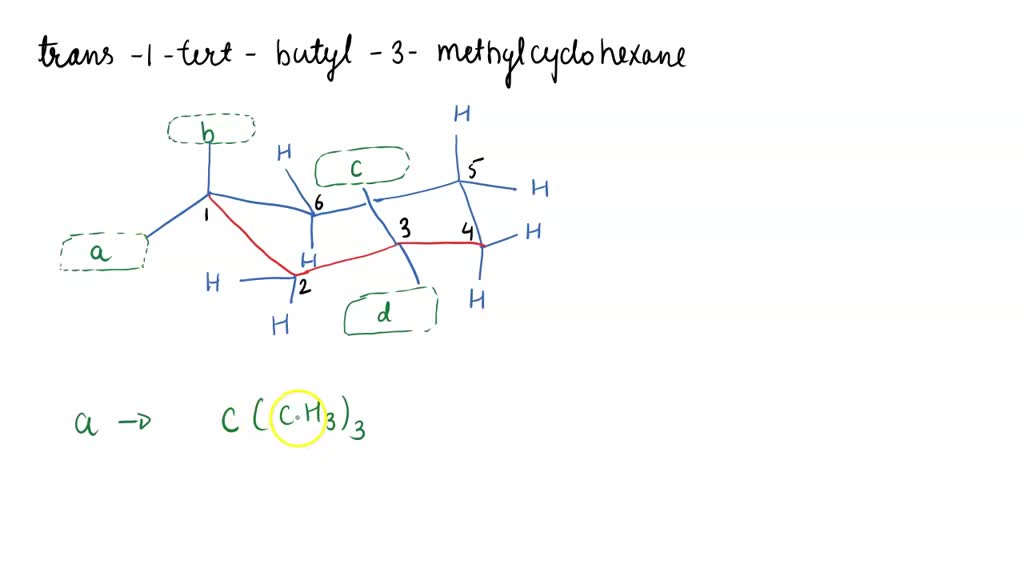
So, to sum up our molecular drama: the most stable chair form of trans-1-tert-butyl-3-methylcyclohexane is the one where the bulky tert-butyl group gets to stretch out in an equatorial position. It’s the molecule’s ultimate comfort zone! This preference for the equatorial position by the largest substituent is a fundamental principle in organic chemistry, and it’s a neat little peek into how molecules arrange themselves to be as happy and relaxed as possible.
Isn't it cool that even these tiny, invisible things have preferences and shapes that we can understand? It’s like having a whole world of tiny, invisible furniture arrangements to discover. Who knew chemistry could be so much about finding the most comfortable seat in the house?
The next time you see a molecule name that looks a bit intimidating, remember our friend trans-1-tert-butyl-3-methylcyclohexane. It's a reminder that behind every complex name is a simple story about shape, space, and finding the perfect, stable position. It's a little molecular drama playing out, and the star of the show is always the one who finds the best place to sit!
It’s all about finding that sweet spot where everyone can relax!
This preference for the equatorial position by large groups is a big deal in understanding how molecules behave. It helps scientists predict how reactions will happen and what shapes molecules will take. So, while it might seem like just a naming convention, it's actually a key to unlocking a lot of chemical secrets. It’s like having a secret handshake that tells you all about the molecule’s personality.
So, if you ever feel like your furniture arrangement could be more comfortable, just remember our cyclohexane friend. It’s always striving for that most stable, most relaxed posture. And in the case of trans-1-tert-butyl-3-methylcyclohexane, that means giving the tert-butyl group the prime equatorial real estate. It’s a tiny triumph of molecular engineering, all for the sake of ultimate comfort!
