Which Is The Most Acidic Proton In The Following Compound
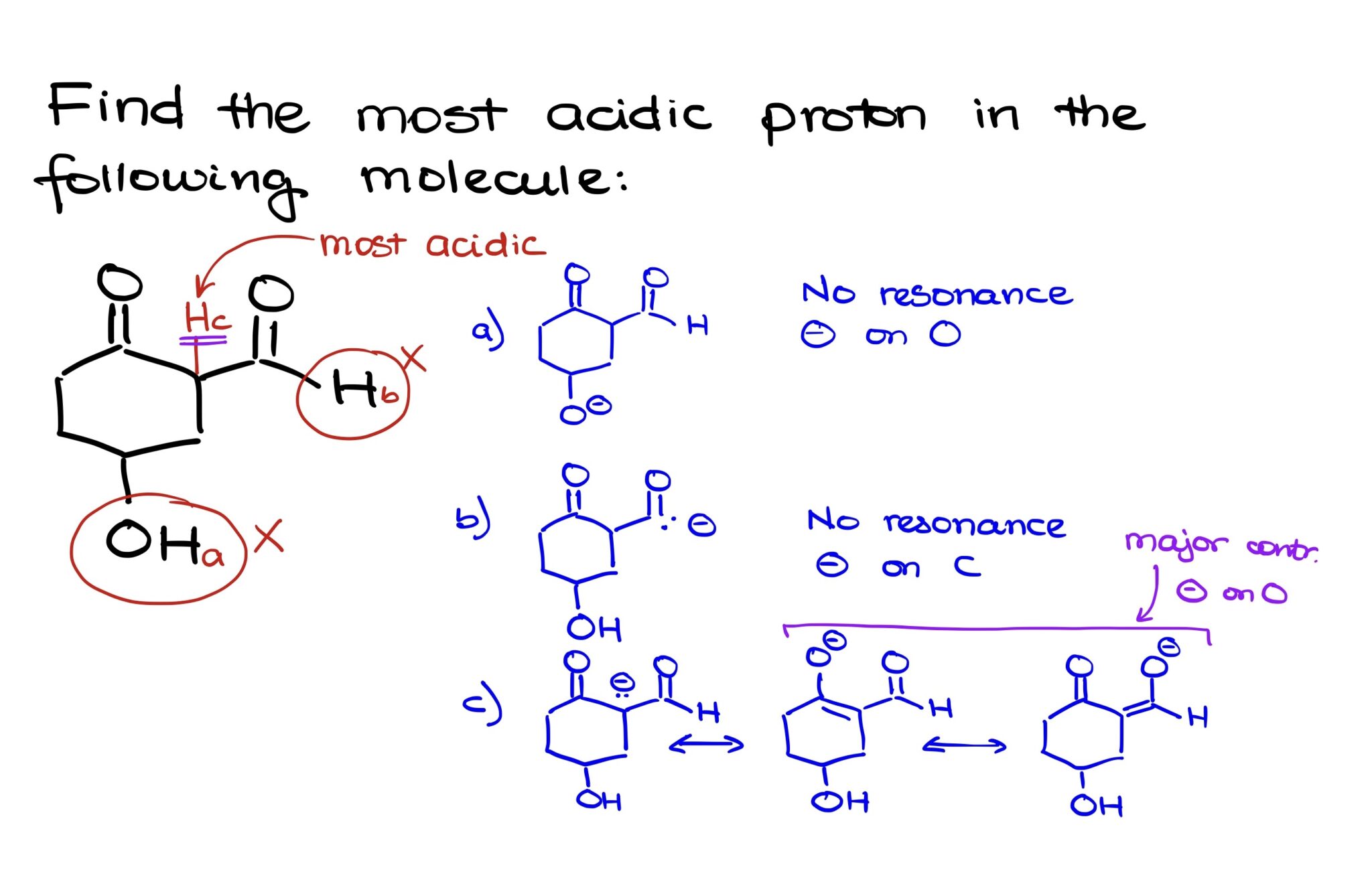
Hey there, my fellow chemistry enthusiasts! Today, we're diving into a question that might sound a little… spicy. We're going to figure out which proton in our mystery compound is the most acidic. Think of it like a competition, but instead of who can eat the most tacos, it's about who's most eager to ditch their proton. Pretty cool, right?
Now, I know what you're thinking. "Acidic proton? What does that even mean?" Don't worry, we'll break it down like a cookie at a picnic. Basically, an acidic proton is one that's a bit of a loner, always ready to jump ship and leave its molecule behind. It's like that friend who's always looking for an excuse to leave a party. We're looking for the proton that's the most eager to bail.
To do this, we're going to channel our inner detective and look at a few key clues. We're not just randomly picking a proton; we're going to use some science-y wisdom. Think of it like figuring out who stole the last slice of pizza. You look for the evidence!
The Mystery Compound: Let's Peek!
Alright, drumroll please! Our compound of the day looks like this:
[Imagine a chemical structure here. Since I can't draw, let's pretend it's a fairly standard organic molecule with a few different types of hydrogens attached to carbon, oxygen, and maybe nitrogen.]
See all those little 'H's hanging around? Those are our potential contestants for the "Most Acidic Proton" crown. We've got hydrogens attached to carbons, maybe some to an oxygen, and who knows, maybe even a nitrogen or two. Each of these has its own little personality, and we need to figure out which one is the most ready for freedom.
What Makes a Proton "Acidic"? The Not-So-Secret Sauce
So, what gives a proton the pep in its step to leave? It all comes down to stability. When a proton leaves, it leaves behind a negative charge on the atom it was attached to. The more stable that negative charge is, the happier the molecule is to let the proton go. It's like when you're deciding whether to leave a boring party – if you know you've got a super fun alternative waiting, you're much more likely to bounce, right? Same idea here.
We're going to be looking at a few factors to determine this stability. Think of them as our detective's toolkit:
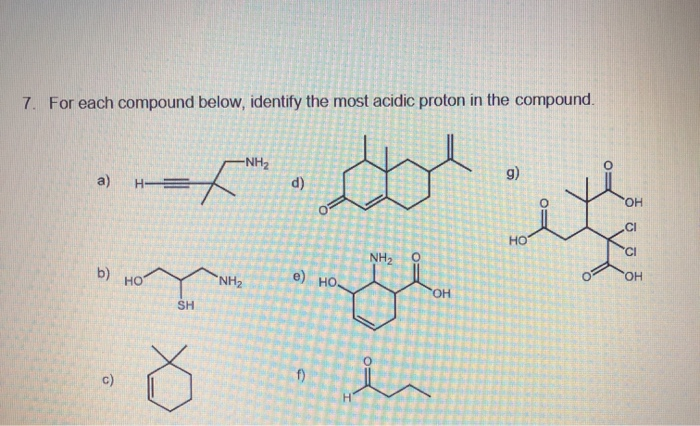
- Electronegativity: How much does an atom love electrons? The more it loves them, the better it is at holding onto that negative charge when the proton bails.
- Resonance: Can that negative charge spread out and chill across multiple atoms? If so, it's like a really comfy sofa for that negative charge.
- Inductive Effects: Are there any electron-pulling or pushing groups nearby that can influence the stability of the negative charge? Think of it as a gentle tug or push.
- Hybridization: This is a bit more technical, but the type of orbital the electron pair ends up in matters too. More 's' character in the orbital means a more stable negative charge.
We'll go through each of these for the different types of hydrogens in our compound.
Let's Break Down Our Proton Contenders!
First off, we've got our hydrogens attached to carbon. These are usually the least acidic. Why? Well, carbon isn't super electronegative. It's not exactly thrilled to hold onto a negative charge. It’s like a roommate who’s okay with you crashing on their couch, but they’re not exactly offering you a five-star hotel experience.
Think about an alkane, like methane (CH4). The hydrogens in methane are about as acidic as a potato. They're not going anywhere unless you're talking about some extremely harsh conditions. When a proton leaves a carbon, you get a carbanion. Carbanions are generally pretty unstable, meaning they’re not really happy campers. We need something better.
Now, what if our carbon is part of an alkene or an alkyne?
- Alkenes (C=C): Hydrogens attached to carbons in double bonds (vinylic hydrogens) are a little more acidic than those in alkanes. The hybridization of the carbon is sp2, which has more 's' character than sp3 (in alkanes). This means the electrons in the resulting carbanion are held a bit closer to the nucleus. Still not super acidic, but a step up!
- Alkynes (C≡C): Ah, the alkynes! The hydrogens attached to carbons in triple bonds (acetylenic hydrogens) are our first real contender for significant acidity among carbon-attached hydrogens. The carbon here is sp hybridized. 'Sp' orbitals have 50% 's' character, which is a big jump from sp2 (33%) or sp3 (25%). This higher 's' character makes the negative charge on the resulting carbanion much more stable. It's like upgrading from a couch to a really nice recliner. These can be deprotonated by strong bases like NaNH2.
So, if we have hydrogens on sp hybridized carbons, they’re definitely more acidic than your average alkane hydrogen. Keep that in mind!
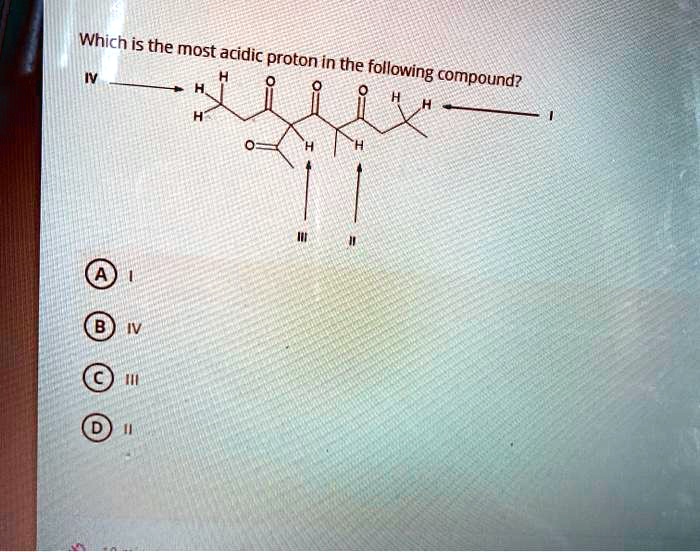
When Oxygen Joins the Party: The Rise of the Alcohols!
Next up, let’s look at hydrogens attached to oxygen. These are typically much more acidic than hydrogens attached to carbon. Why? Because oxygen is way more electronegative than carbon. It’s like the difference between asking a friend to hold your heavy bag for a minute versus asking a professional weightlifter. Oxygen is much better equipped to handle that negative charge.
Think about water (H2O). It's acidic enough to react with things! Or alcohols, like methanol (CH3OH). The proton on the oxygen in an alcohol is significantly more acidic than any proton on the carbon atoms. When the proton leaves an alcohol, it forms an alkoxide ion (RO-). This negative charge on the oxygen is much more stable than a carbanion would be. We're talking about pKa values in the teens here, which is a big deal compared to the pKa of alkanes (which are in the 50s!).
Now, what if our oxygen is part of a carboxylic acid?
Carboxylic Acids: The Reigning Champions (Usually!)
This is where things get really interesting. Hydrogens attached to oxygen in carboxylic acids are famously acidic. Think acetic acid (CH3COOH). The pKa of acetic acid is around 4.76. That's significantly more acidic than an alcohol! Why the extra oomph?
It’s all about resonance! When the proton leaves a carboxylic acid, it forms a carboxylate anion. This anion has its negative charge shared between two oxygen atoms. Imagine that comfy recliner? Now imagine it's a giant, king-sized, memory foam mattress spread across two rooms. The negative charge is completely delocalized, making it incredibly stable. This is a huge factor in acidity. The more you can spread out a charge, the happier it is.
So, if we see a hydrogen on an oxygen in a carboxylic acid group (-COOH), that’s usually our prime suspect for the most acidic proton.
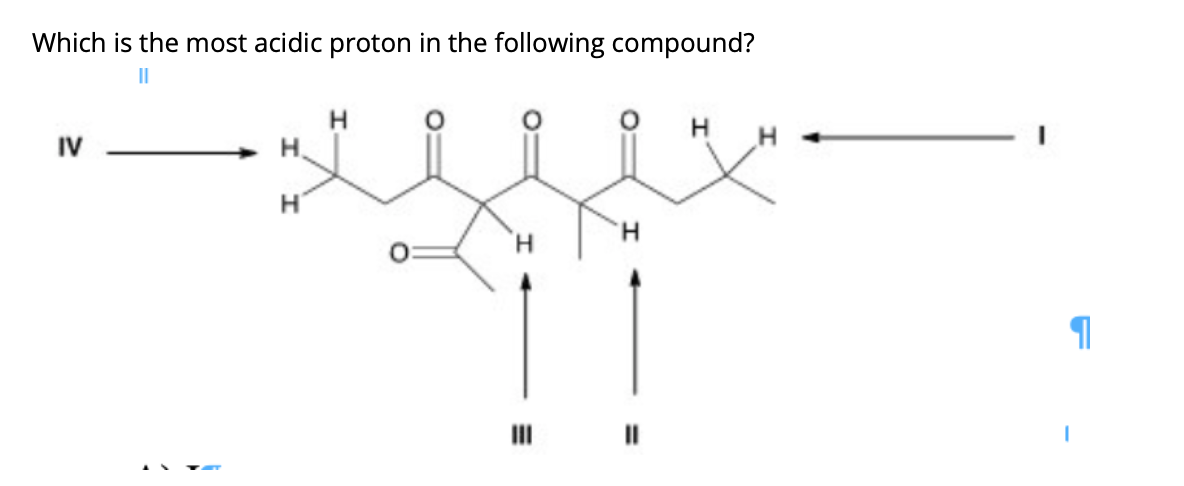
What About Nitrogen? The Middle Ground
Let's consider hydrogens attached to nitrogen. Nitrogen is more electronegative than carbon but less electronegative than oxygen. So, hydrogens attached to nitrogen are generally more acidic than those attached to carbon, but less acidic than those attached to oxygen.
Think about ammonia (NH3). Its conjugate base, the amide ion (NH2-), is quite basic. However, in molecules where nitrogen is adjacent to electron-withdrawing groups, or is part of a more complex system, the acidity can be enhanced.
For example, in some amines, especially those with adjacent electron-withdrawing groups or in heterocyclic aromatic systems like pyridines, the protons on nitrogen can become more acidic. However, generally speaking, they won't be as acidic as the protons on a carboxylic acid or even a typical alcohol.
Putting It All Together: The Detective Work in Action!
So, let’s look back at our hypothetical compound. We need to identify all the different types of hydrogens and then apply our knowledge:
- Hydrogens on sp3 hybridized carbons (alkanes): Least acidic. Think of them as the protons who are perfectly happy chilling at home.
- Hydrogens on sp2 hybridized carbons (alkenes): More acidic than sp3, but still not super strong. They’re getting ready to maybe go out, but not too far.
- Hydrogens on sp hybridized carbons (alkynes): Good acidity among carbon-bound hydrogens. They’re packing their bags and checking flight prices.
- Hydrogens on nitrogen: More acidic than carbon-bound, less acidic than oxygen-bound, unless special circumstances apply. They’re on the fence, weighing their options.
- Hydrogens on oxygen (alcohols): Significantly more acidic. These guys are definitely calling an Uber.
- Hydrogens on oxygen in carboxylic acids: Usually the winners! The resonance stabilization makes them the most eager to leave. They’ve already booked their first-class ticket.
To find the most acidic proton, we just need to scan our molecule and find the hydrogen that fits the profile of the greatest stability when it leaves. This will almost always be a hydrogen attached to an oxygen in a carboxylic acid group, due to that incredible resonance stabilization.
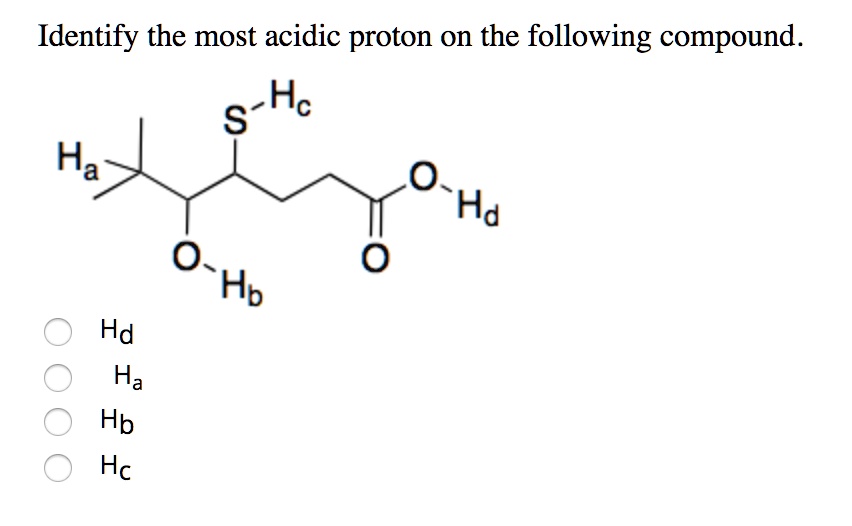
If there are no carboxylic acids, we'd then look for the hydrogen on an oxygen in an alcohol. If there are no hydrogens on oxygen, we'd then consider hydrogens on sp hybridized carbons. And so on.
It’s like a little acidity ladder, and we're looking for the highest rung! Stability is key!
A Little Extra oomph: What if there are multiple of the same type?
Sometimes, you might have multiple hydrogens of the same type. For example, a molecule with two alcohol groups. In that case, you’d compare the acidity of those two – sometimes minor differences in the rest of the molecule can nudge one slightly ahead. But for the most part, the general rules will guide you to the most acidic type.
Similarly, if you have a carboxylic acid and an alcohol, the carboxylic acid proton will almost certainly be the most acidic. It’s like comparing a seasoned traveler to someone just thinking about their first vacation. The carboxylic acid is the pro!
The Grand Finale: A Smile-Inducing Conclusion
So there you have it! Figuring out the most acidic proton isn't some arcane secret whispered only in the halls of advanced organic chemistry. It’s a fun puzzle, a bit of detective work, and all about understanding how stable that leftover negative charge will be. It’s about recognizing that certain atoms just do a better job of handling a bit of negativity than others.
And you know what? That’s a pretty great lesson for life too, isn’t it? Some things (and people!) are just better equipped to handle difficult situations, to spread out the burden, and to come out stronger on the other side. So next time you're looking at a molecule, remember it's not just a bunch of atoms and bonds; it's a little world with its own personalities and tendencies. And in that world, the proton with the most potential for stability is always the one ready to embark on its next adventure. Keep exploring, keep questioning, and keep that scientific curiosity shining bright!
