Which Is An Indication That Water Is Entering Cells

Hey there, friend! Ever wonder what’s really going on inside your body, like, at a microscopic level? It’s pretty mind-blowing stuff, and today we’re diving into one of the coolest, and frankly, most essential processes: how we know when water is making its grand entrance into our cells. Yep, those tiny powerhouses that keep you running are constantly taking sips of this precious liquid, and there are some awesome clues that tell us this hydration party is in full swing.
So, grab a comfy seat (maybe with a glass of water, hint hint!) because we’re about to spill the tea on cellular hydration. No fancy jargon, no boring lectures, just a friendly chat about how your body works its magic. Think of this as a backstage pass to the microscopic world, where water is the VIP guest!
The Tiny Doors: Aquaporins Are Your Cell's Water Welcome Wagon
Okay, first things first. Our cells aren't just sitting around waiting for water to magically appear. They’ve got little, shall we say, entryways. And these aren't just any old doors; they're super specialized, incredibly efficient water channels called aquaporins. You can think of them as the cell's personal water valet service. Pretty neat, right?
These aquaporins are like tiny tunnels that water molecules can zoom through. Without them, water would have a really hard time getting inside. It’s like trying to get a whole parade through a tiny peephole – not going to happen! So, the very presence and activity of these aquaporins is a huge indication that water is being welcomed in.
When your body needs to hydrate cells, or when there’s more water available outside the cell than inside, these aquaporins are like, "Alright team, open for business!" They start working overtime, letting that refreshing H2O flow in. It's a beautifully orchestrated dance, and aquaporins are the lead dancers.
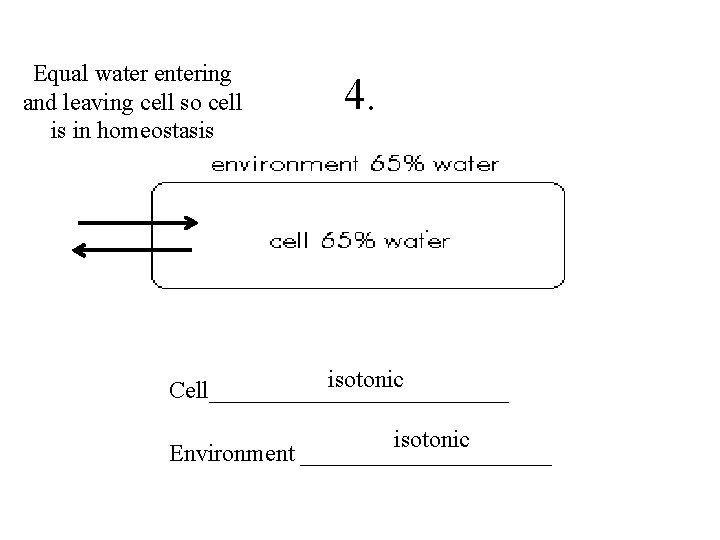
Spotting the Signs: When Cells Get Plump and Happy
So, how do we see this water entry in action? Well, it’s not like you can pull out a microscope and watch it happen in real-time (though wouldn't that be cool?!). But scientists have figured out some pretty clever ways to tell. One of the most obvious signs is a change in the cell's volume.
Imagine a tiny, deflated balloon. Now, imagine filling it with water. What happens? It gets bigger, right? It plumps up! Cells are kind of like that. When water enters them, they swell. This swelling is a direct, visual cue that water is indeed making its way inside.
This is a really fundamental concept. Cells are like little water balloons, and their size can tell us a lot about their hydration status. If cells are consistently shrinking, it’s a sign they’re dehydrated. But if they’re maintaining a healthy, plump volume, it’s a good bet they’re getting their fill of water. It’s like seeing happy, full cheeks on a person – you just know they’re doing well!
Osmosis: The Silent Force Behind Water's Journey
Now, you might be asking, "Why does water even want to go into the cell?" Great question! It's all thanks to a super important scientific principle called osmosis. Don't let the fancy word scare you; it's actually quite logical.
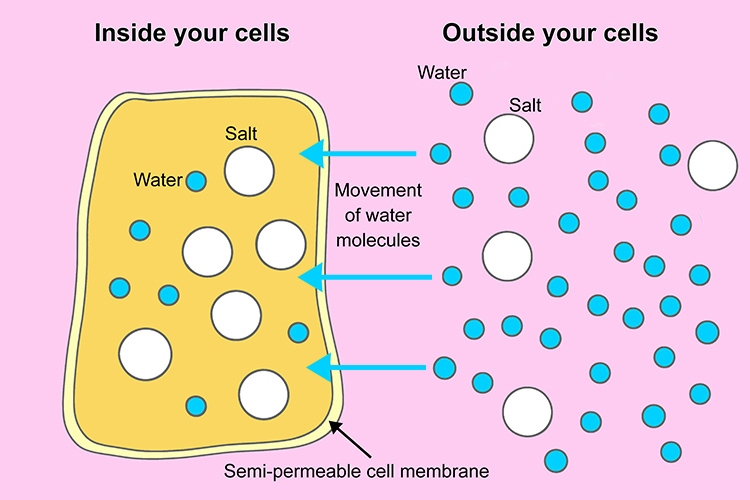
Think of it like this: imagine you have two rooms separated by a special curtain. This curtain lets water pass through, but not bigger things like salt or sugar. In one room, you have lots of salt, and in the other, you have very little salt. What do you think will happen to the water?
The water will naturally move from the room with less salt to the room with more salt. Why? Because it's trying to even things out! It's trying to dilute the concentrated salt solution and make the salt concentration the same on both sides of the curtain. This movement of water across a semipermeable membrane (like the cell membrane) from an area of lower solute concentration to an area of higher solute concentration is the essence of osmosis.
Cells are just like those rooms. The inside of a cell has a certain concentration of dissolved substances (salts, sugars, proteins, etc.), and the outside environment has a different concentration. When there's a higher concentration of solutes inside the cell, water will be drawn in through osmosis to try and balance things out. So, this internal tug-of-war of solute concentration is a major driver for water entering cells.
Concentration Gradients: The Cell's Internal Compass
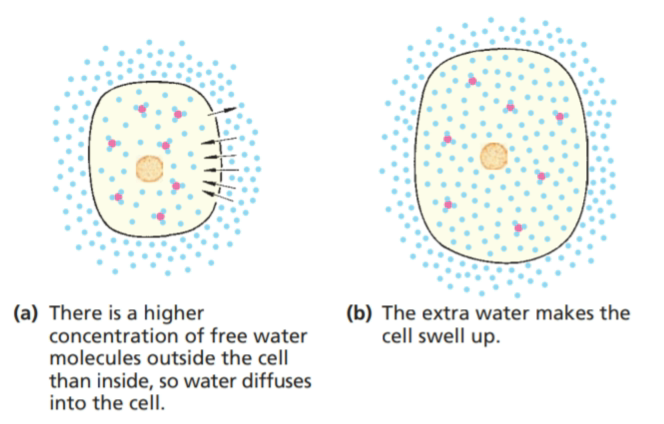
This concept of concentration differences is what we call a concentration gradient. It's like a hill that water "wants" to flow down. If the concentration of dissolved stuff (solutes) is higher inside the cell than outside, water will move in to try and reduce that difference. Conversely, if the solute concentration is higher outside the cell, water will move out.
So, measuring or observing these solute concentrations inside and outside the cell can be a huge indicator of water movement. When scientists see that the internal "soup" of the cell is more concentrated than the surrounding fluid, they know water is likely to be entering. It’s like knowing which way the wind is blowing by looking at the leaves on a tree.
This isn't just some abstract idea; it's the fundamental principle governing how our cells get their water. Our bodies are constantly managing these gradients to ensure cells get the right amount of hydration, not too much, not too little. It’s a delicate balancing act, and the concentration gradient is the conductor of this aqueous orchestra.
Changes in Cell Membrane Potential: The Electrical Whisper
Now, things get a little more technical, but stick with me! Sometimes, water entering or leaving a cell can actually cause a slight change in the cell's electrical charge, which we call the membrane potential. It’s like a tiny electrical whisper that can signal water movement.
Cells have a delicate balance of charged particles (ions) both inside and outside. When water moves, it can sometimes drag some of these charged particles with it, or it can alter the concentration of ions in a way that shifts the overall electrical charge across the cell membrane. This change, however subtle, can be detected by sensitive instruments and serves as another clue.
Think of it like this: imagine a busy street with cars (ions) and pedestrians (water molecules). If a lot of pedestrians suddenly start crossing, they might indirectly affect the flow of cars. In a similar way, water movement can subtly influence the electrical environment of the cell. It’s not the primary way we detect water entering, but it’s a fascinating secondary indicator that scientists can use.
Ion Channels: The Gatekeepers of Charge
This is where the role of ion channels comes into play. These are also special protein channels in the cell membrane, but instead of letting water through, they let specific ions pass. The opening and closing of these ion channels are tightly regulated, and they are crucial for maintaining the cell's electrical balance.
When water moves in or out, it can sometimes trigger the opening or closing of these ion channels, which then alters the membrane potential. So, by observing the activity of ion channels and the resulting changes in membrane potential, scientists can get indirect evidence of water transport. It's like seeing smoke and knowing there's a fire, even if you can't see the flames directly.
It’s a testament to how interconnected everything is within a cell. A simple influx of water can have ripple effects on the electrical landscape, and by studying these ripples, we learn more about the water's journey. Pretty sophisticated for something so small!
Cellular Swelling: The Obvious (and Sometimes Not-So-Obvious) Sign
Let's circle back to something we touched on earlier: cellular swelling. This is arguably the most straightforward indicator. When water enters a cell, the cell's internal pressure (called turgor pressure in plant cells, but the principle is similar) increases, causing it to expand. This expansion is a direct consequence of water uptake.
You can visualize this by imagining a dry sponge. When you soak it in water, it absorbs the liquid and expands. A cell doing the same thing. If you have a group of cells, and they all start to swell, it's a pretty clear sign that water is entering them. It's like seeing a whole field of flowers perk up after a good watering.
However, sometimes this swelling can be subtle. Cells are microscopic, so you're not going to see them visibly puff up with the naked eye. Scientists use advanced microscopy techniques, or measure changes in cell density or light scattering, to detect these subtle volumetric changes. They have their tricks up their sleeve!
Cellular Density and Light Scattering: The Sciencey Way to See Swelling
So, how do scientists actually measure this swelling if they can't see it with their own eyes? Well, they get a bit creative. One way is to look at the density of the cells. When a cell swells with water, its overall density decreases because it has more volume for the same amount of "stuff" inside. Think of a beach ball versus a bowling ball – the beach ball is much less dense.
Another clever technique involves looking at how light interacts with the cells. When cells swell, their physical structure changes, and this can affect how they scatter light. Instruments can measure these changes in light scattering, giving scientists an indirect but reliable way to infer water uptake. It's like feeling the air move to know something is happening, even if you can't see the wind itself.

These methods are incredibly useful for studying cellular processes in a controlled environment, allowing us to understand the fundamental mechanisms of hydration. It’s a bit like being a detective, piecing together clues to solve a microscopic mystery!
The Big Picture: Why Does This Matter?
You might be thinking, "Okay, cool, water goes into cells. But why should I care?" Well, my friend, this is literally the stuff of life! Every single process happening in your body relies on cells being properly hydrated.
From your brain cells firing off thoughts to your muscle cells contracting for that awesome workout, to your skin cells keeping you looking radiant – they all need water to function. When water enters cells, it's not just about filling them up; it's about providing the medium for countless chemical reactions to occur, transporting nutrients, and removing waste.
Think of your cells as tiny factories. Water is the essential lubricant, the coolant, and the transport system that keeps everything running smoothly. Without it, the whole operation grinds to a halt. So, that refreshing glass of water you’re sipping? It’s fueling a microscopic marvel!
Keeping Your Body Humming: Hydration is Key
So, the next time you feel thirsty, or you’ve just chugged some water after exercise, take a moment to appreciate what’s happening on a cellular level. You’re actively helping your aquaporins do their job, your osmosis working its magic, and your cells to swell with life-giving hydration.
This fundamental process is what allows your body to perform all its amazing feats, from thinking deep thoughts to running marathons. It's the silent, everyday miracle that keeps you going. So, here’s to keeping those cells happy, plump, and perfectly hydrated. Cheers to your incredible, microscopic world – may it always be overflowing with life!
