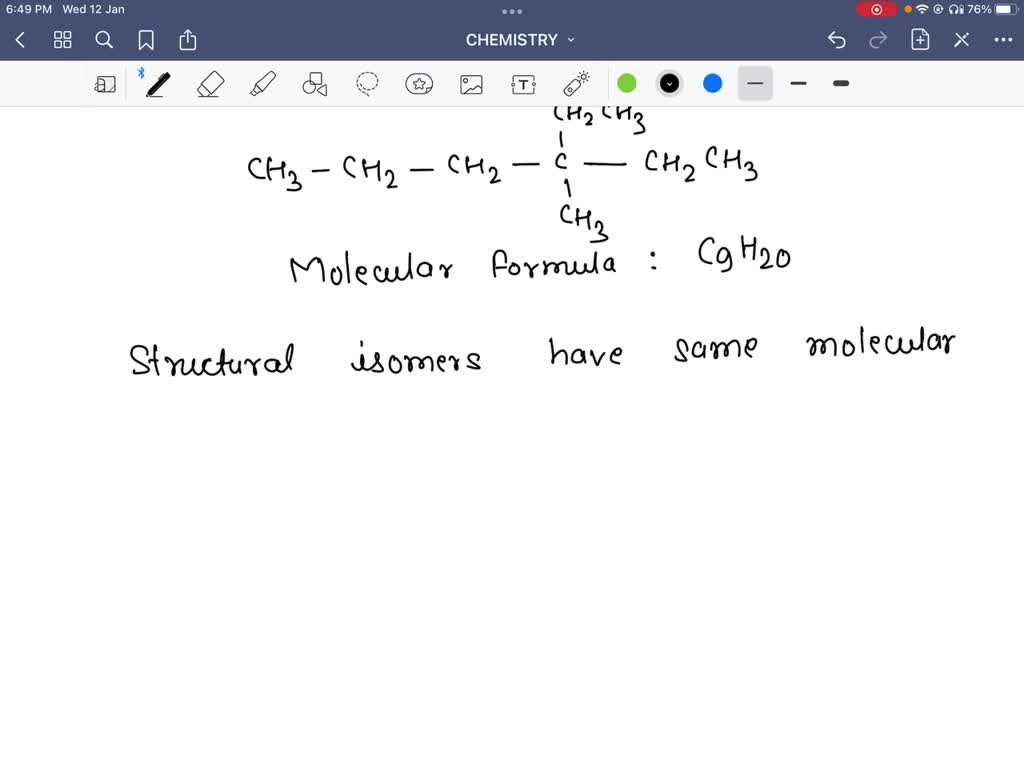
Which Is A Structural Isomer Of 3 Ethyl 3 Methylhexane

Imagine a world of tiny building blocks, the kind that chemists use to construct all sorts of amazing things – from the fizz in your soda to the comfortable fabric of your favorite t-shirt. These blocks are called atoms, and they love to link up in chains and rings, forming molecules. Now, sometimes, these atoms get a little creative with how they arrange themselves. It’s like having the same set of LEGO bricks but building slightly different, yet still awesome, structures. That’s where our story begins, with a molecule that sounds a bit like a fancy cocktail: 3-Ethyl-3-methylhexane.
Now, “hexane” tells us our main chain has six carbon atoms lined up, like a little parade. Then, “ethyl” and “methyl” are like little decorations hanging off this parade. “Ethyl” is a group of two carbon atoms, and “methyl” is a single carbon atom. The numbers “3” tell us exactly where these decorations are attached – right in the middle of our six-carbon parade, on the third carbon from either end. So, picture this: a six-person line, and right in the middle, on the third person, we have two friends joining hands – one friend is a pair (ethyl), and the other is flying solo (methyl).
This particular arrangement, 3-Ethyl-3-methylhexane, is like the original masterpiece. It’s got its own personality, its own way of behaving, and its own unique smell (though we won't be sniffing it today!). But here's the fun part, the little twist in our story: what if those same LEGO bricks, those same atoms, decided to connect in a slightly different order? What if they wanted to surprise us? This is where we meet its structural isomers.
Think of it like this: you’ve got a recipe for a delicious cake. The ingredients are all the same – flour, sugar, eggs, butter. But what if you decide to bake them in a muffin tin instead of a cake pan? Or what if you add a swirl of frosting on top that wasn't in the original plan? The cake is still made of the same stuff, but it looks and tastes a little different, right? That’s exactly what structural isomers are doing. They’re the same ingredients (atoms) but assembled in a different order, giving them a new form.
So, what’s a structural isomer of our 3-Ethyl-3-methylhexane? It’s another molecule made of exactly the same atoms, but they’re linked up in a different pattern. Imagine our six-carbon parade again. Instead of having those two friends (ethyl and methyl) attached to the exact same spot, what if one of them decided to hang out a little further down the line? Or what if the main parade decided to bend and form a little loop instead of a straight line?

Let’s introduce a contender, a molecule that’s like a sibling to 3-Ethyl-3-methylhexane, but with a slightly different outlook. This sibling is called 3,4-dimethylhexane. Now, that sounds a bit similar, doesn’t it? “Hexane” still means our six-carbon parade. But instead of an ethyl and a methyl group, we have two methyl groups. And instead of both hanging off the third carbon, they’ve decided to spread out a bit – one on the third carbon, and the other on the fourth. It’s like our parade has two solo friends, but they’re standing on adjacent shoulders instead of both on the same shoulder.
It’s a subtle change, really. Like a tiny tweak in a dance move that changes the whole rhythm of the performance.
SOLVED: Score: 755/2900 0f 29 Which is a structural isomer of 3-ethyl-3
And that’s the magic! Even though 3,4-dimethylhexane uses the exact same number of carbon and hydrogen atoms as 3-Ethyl-3-methylhexane, its different arrangement means it might have slightly different properties. It could evaporate at a slightly different temperature, or react a little differently in a chemical reaction. It’s like two twins who look almost identical but have completely different personalities and hobbies. One might love to read, while the other is always out playing sports. Same genes, different expressions.
Finding these isomers is like a fun detective game for chemists. They’re looking for all the possible ways to connect these atoms, like solving a giant molecular puzzle. And each piece, each isomer, has its own story to tell. It’s a reminder that even with the same basic ingredients, the universe can create an astonishing variety of forms and functions.
So, the next time you hear a fancy chemical name, don't be intimidated! Think of it as a blueprint for a tiny, intricate structure. And remember that sometimes, the most interesting variations happen when the building blocks get a little creative. 3,4-dimethylhexane is just one of the many clever ways nature can rearrange the same atoms to create something new, something that shares a lineage but charts its own unique course in the molecular world. It’s a delightful demonstration of the subtle beauty and endless possibilities found in the very fabric of matter, a little dance of atoms that keeps on surprising us.