Which Information Did Millikan Contribute To The Understanding Of Electrons

Alright, settle in folks, grab your biscotti, and let me tell you about a guy named Robert Millikan. This dude was basically the Sherlock Holmes of the microscopic world, but instead of dusty magnifying glasses, he was using fancy contraptions to track teeny-tiny, invisible things. And what was he tracking? You guessed it, the elusive, the infamous, the utterly essential electron!
Now, before Millikan came along and did his thing, electrons were kind of like that awkward cousin at a family reunion. Everyone knew they were there, doing something, but nobody could quite nail down their characteristics. It was all a bit of a muddle. Scientists had hints, whispers in the atomic ether, but no solid evidence. They were like detectives with a blurry footprint and a vague description of a suspect – exciting, but not exactly closing the case.
So, what exactly did our man Millikan uncover that got us all jazzed about electrons? Well, he basically did two major things. Think of them as the killer one-two punch that knocked out the ignorance surrounding these fundamental particles. And trust me, this was a big deal. Before this, understanding the atom was like trying to understand how a car works by just looking at a pile of its parts. You knew there were pieces, but the how and the why were missing.
First off, Millikan figured out the electric charge of a single electron. Yes, you read that right. He measured the charge of one of these minuscule mischief-makers. Imagine trying to measure the weight of a single grain of sand on a balance that’s so sensitive, a sneeze could throw it off. That’s the kind of precision we’re talking about here. It’s like he invented a microscopic scale that could weigh the tiniest speck of dust in the universe.
How did he pull off this microscopic marvel? With his famous oil-drop experiment. Picture this: a little chamber, a sprayer like you’d use for perfume (but way fancier), and some tiny oil droplets. He’d spray these droplets, and some of them, through a bit of atom-brained magic (okay, ionization from X-rays, but let’s keep the mystique alive!), would pick up an electric charge. Then, he’d use electric fields to levitate these charged droplets. It was like a microscopic ballet of charged particles, with Millikan as the conductor.
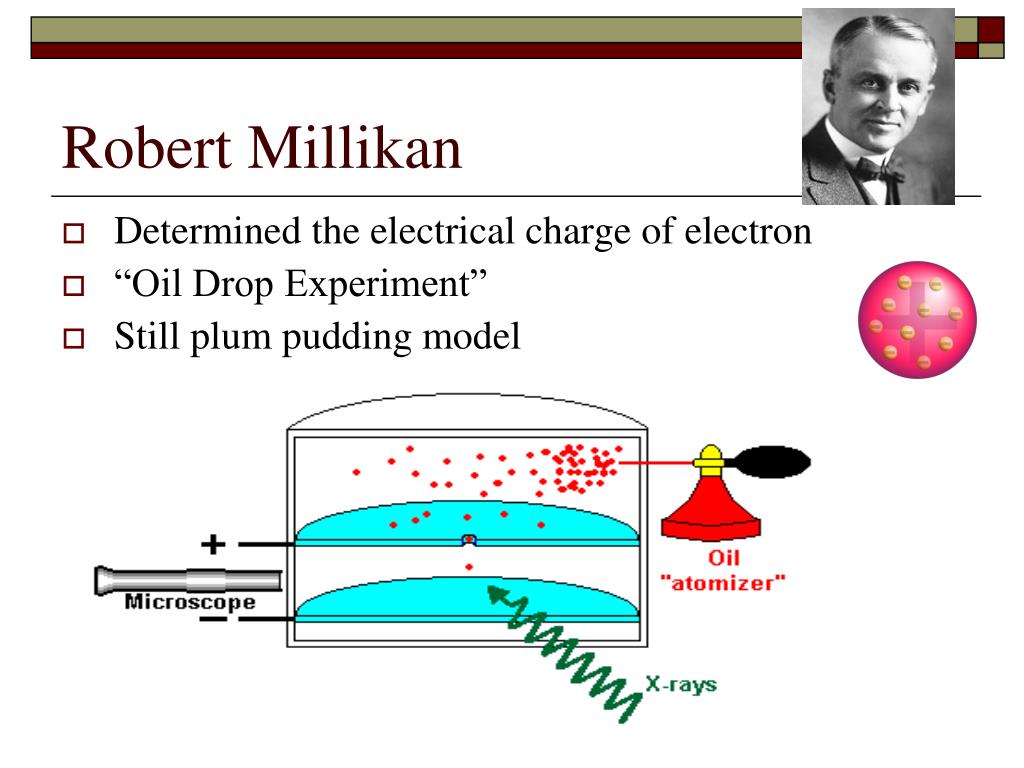
He’d adjust the electric field, watching these droplets dance. Some would drift down, others would be held perfectly still, and some might even drift up. By carefully observing how these droplets responded to different electric forces, he could calculate the total charge on each one. And here’s the mind-blowing part: he noticed that the charges on these droplets were always, always, multiples of a specific, tiny number. It was like finding out that all the different ways to pay for a gumball were just combinations of nickels and dimes, never a weird seven-cent coin. This specific, tiny number? That was the charge of a single electron. Boom! Mic drop.
Think about the implications! This wasn't just some abstract scientific curiosity. Knowing the charge of an electron was like finding the fundamental building block of electrical interactions. It was the key that unlocked so many doors in physics and chemistry. Suddenly, we could start building a coherent picture of how electricity worked, how atoms bonded, and even how light behaved.
But wait, there’s more! Millikan didn’t just stop at the charge. He also figured out the mass of the electron. Now, if measuring the charge was like weighing a grain of sand, measuring the mass was like trying to weigh a dust mite that’s been on a diet. It’s incredibly small. He used his clever oil-drop experiment, combined with some other physics know-how, to deduce the mass of this phantom particle.

He essentially used the charge he’d already measured and combined it with other known physics laws (like how fast things accelerate in certain fields). By comparing how a charged droplet moved under gravity versus how it moved under an electric field, he could make an educated guess, a very educated guess, about its mass. And surprise, surprise, it was ridiculously, laughably small. We’re talking about a mass so tiny that if you put a million electrons on a feather, you probably wouldn’t even notice the difference. It’s like trying to measure the weight of a single thought. It’s that minuscule.
So, to recap, Millikan gave us two absolute gems: the fundamental unit of electric charge (the electron’s charge) and the mass of the electron. These weren’t just facts; they were foundational pillars. Without these measurements, our entire understanding of electricity, matter, and pretty much everything would be wobbly and uncertain, like a Jenga tower after a rogue elbow.

His work was so important, so groundbreaking, that he won a Nobel Prize for it. And let me tell you, winning a Nobel Prize for measuring something so small is like winning an Oscar for perfectly mimicking a cricket chirp. It’s a testament to incredible skill and dedication. It proves that even the tiniest things can have the biggest impact.
Before Millikan, electrons were just fuzzy concepts. After Millikan, they were tangible, measurable entities. He took them from the realm of theoretical speculation and firmly planted them in the world of empirical science. It’s like he turned a ghost into a documented, fingerprint-able individual. He gave us the exact properties of the tiny particle that makes your phone light up, your toaster toast, and your entire world hum with energy.
So, the next time you flip a light switch or scroll through your social media feed, spare a thought for Robert Millikan. He’s the guy who, with a bit of oil, some electricity, and a whole lot of brainpower, helped us understand the tiny, powerful force that makes it all possible. He truly was a giant in the microscopic realm!
