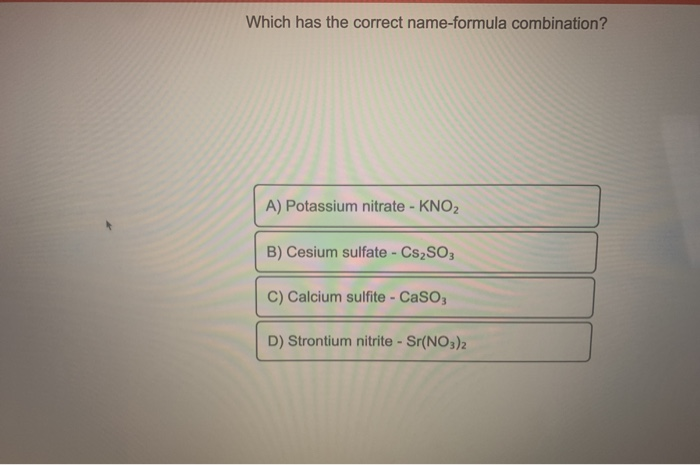
Which Has The Incorrect Name-formula Combination

Alright, gather 'round, my chemically curious comrades! You ever look at a chemical name and its formula and feel like you’ve just walked into a costume party where everyone forgot to tell the clown he’s supposed to be a mime? Yeah, me too. It’s like a cosmic clerical error, a scientific slip-up of epic proportions that leaves us scratching our heads and wondering if the universe has a sense of humor. Today, we’re diving headfirst into the wonderfully weird world of incorrect name-formula combinations. Prepare for gasps, giggles, and maybe a tiny bit of existential dread about the reliability of textbooks.
So, what are we even talking about? Basically, every chemical compound has a name – like “water” or “table salt” – and a formula, which is its secret handshake of atoms, like H₂O or NaCl. Most of the time, these two things are best buds, perfectly matched like peanut butter and jelly. But sometimes, someone, somewhere, apparently after a particularly rough night at the lab, decided to slap a name onto a formula that just… doesn’t fit. It’s like calling a poodle a rhinoceros. Not even close, buddy!
The Case of the Misleading Moniker
Let’s kick things off with a classic that’ll make you question everything you thought you knew about basic chemistry. We’re talking about things that sound totally innocent but are hiding a scandalous secret. Think of it as a chemical Jekyll and Hyde situation.
Take, for example, “Dihydrogen Monoxide.” Sounds fancy, right? Like something you’d find in a very expensive, probably European, bottled water. You’re imagining sparkling water with a hint of cucumber and maybe a fairy’s tear. But this, my friends, is where the deception truly begins. The formula for Dihydrogen Monoxide is H₂O. Yes, you read that right. That impossibly complex, vaguely intimidating name is just a ridiculously long way of saying… water. The stuff you drink, the stuff that falls from the sky, the stuff that makes up most of you. They basically gave the most common, everyday substance on Earth the scientific equivalent of a superhero origin story. I imagine the chemist who came up with this was just having a really, really slow day and decided to spice things up.
And the craziest part? This isn't even a wrong combination in the sense that H₂O isn't water. It's wrong in the sense that the name is designed to sound terrifying and complex when it’s actually the most mundane thing imaginable. It's the ultimate prank. Scientists have even used this to prank people into signing petitions to ban "dihydrogen monoxide" because of its supposed dangers (it can cause erosion, suffocation, and is a major component of acid rain, all true, but also true of… well, water!). It’s a testament to the power of scientific jargon and a great reminder that sometimes the most dangerous-sounding things are just… normal.

When Formulas Go Rogue
But it's not just about overly complicated names for simple things. Oh no, that would be too easy. Sometimes, the formula itself is the imposter, masquerading as something it's not, while its name is perfectly innocent. It’s like a wolf in sheep’s clothing, or in this case, a really reactive element in a gentle-looking formula.
Consider this: you’ve got sodium chloride. We all know that one, right? The king of the kitchen counter, the reason your fries are edible. Its formula is NaCl. Simple, elegant, correct. But what if, hypothetically, someone mistakenly wrote the formula for sodium chloride as, say, Na₂Cl? Suddenly, you’ve gone from your everyday table salt to a chemical compound that might spontaneously combust in your salt shaker. It’s the difference between a friendly wave and a karate chop. The name says "nice to meet you," but the formula is screaming "get away from me!"

The implications are huge, people! Imagine you’re a chef, meticulously measuring out ingredients, and your recipe calls for NaCl. You grab what you think is salt, but it’s actually Na₂Cl. Your entire kitchen could go up in smoke, quite literally. It’s the kind of mistake that would make Gordon Ramsay weep into his Michelin stars. Thankfully, the scientific community is usually pretty good at catching these slip-ups before they cause a culinary apocalypse.
The Case of the "Confused" Compound
Here’s where things get really juicy. Sometimes, the name and formula are almost right, but there’s a tiny, infuriating detail that’s off. It’s like a perfect painting with a single, glaring smudge. This is where the real detective work begins.
Let’s talk about carbon monoxide. Its formula is CO. Pretty straightforward, deadly gas, best avoided. But what if, in a moment of profound cosmic confusion, someone insisted that the formula for carbon monoxide was CO₂? Now, that’s a mismatch made in… well, not heaven. CO₂ is, of course, carbon dioxide. The stuff we exhale, the stuff that makes soda fizzy, and the primary greenhouse gas. Carbon monoxide (CO) is the one that famously, tragically, kills people in poorly ventilated spaces because it binds to your blood and stops you from breathing. So, confusing CO for CO₂ is like confusing a little paper cut for a full-blown amputation. Both involve a loss of blood, sure, but one is significantly more problematic.
This isn't just a theoretical "oopsie." Misidentifying these gases can have life-or-death consequences. A faulty carbon monoxide detector is bad enough; a faulty detector that's also mislabeled due to a name-formula mix-up? That’s a recipe for disaster. It’s a stark reminder that in the world of chemistry, precision isn't just important; it's a matter of survival. They’re practically identical twins, but one is a charming conversationalist, and the other is a silent, deadly assassin.
Why Does This Even Happen?
You might be wondering, how in the name of all that is holy do these mix-ups occur? Well, it’s a combination of factors, really. Sometimes, it’s pure human error. We’re all just complex biological machines, prone to occasional glitches. Think of it as a typo in the grand cosmic code.
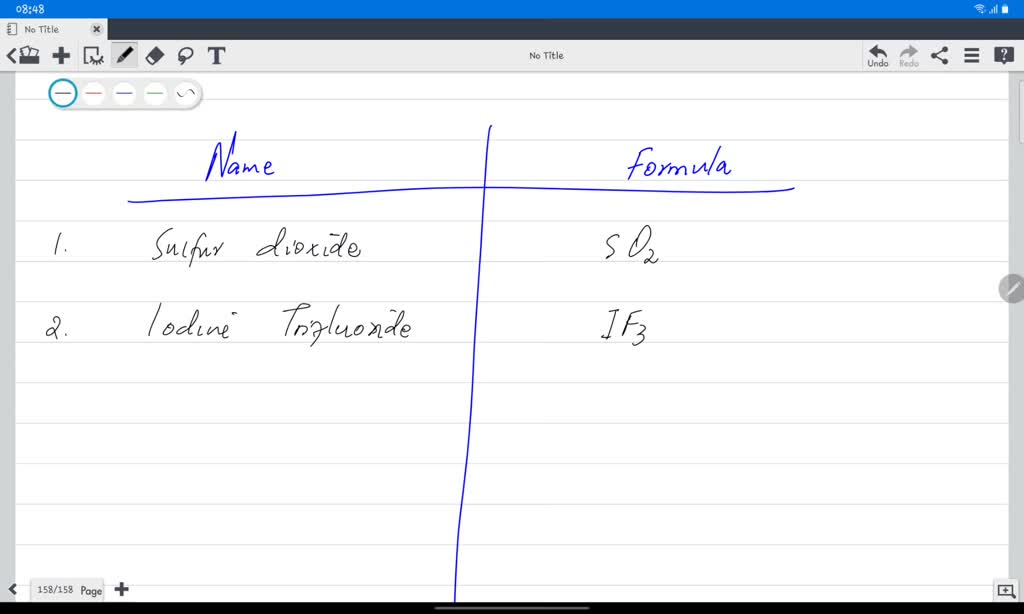
Other times, it’s about historical naming conventions. Before we had standardized IUPAC nomenclature (that’s the fancy scientific naming system), chemists had all sorts of quirky names for things. They’d name compounds after their discoverer, their source, or even their perceived properties. This led to a glorious, chaotic mess that sometimes makes it hard to tell what’s what. It’s like trying to navigate a city where every street has three different names depending on who you ask.
And let's not forget the sheer complexity of chemistry. There are millions of compounds out there. It’s bound to happen that a few slip through the cracks, a few names get tangled with their formulas, and a few atoms decide to play hide-and-seek. It’s a testament to how much we don't know, and how much there is still to discover, and occasionally, to misname.
So, the next time you’re staring at a chemical equation, remember: behind every formula and every name, there's a story. And sometimes, that story involves a scientist who was just trying their best, a historical quirk, or perhaps, a mischievous atom that decided to mess with the system. It's a reminder that even in the structured world of science, there's always room for a little bit of delightful, and sometimes dangerous, confusion.
