Which Has The Highest Boiling Point Ccl4 Cf4 Or Cbr4
Ever found yourself staring at a label on a cleaning product, or maybe wondering about those little packets that keep electronics dry? Sometimes, the world of chemistry, with its jumble of letters and numbers, can feel a bit like trying to decipher a secret code. But what if I told you that understanding a tiny bit of chemistry could actually be… well, kinda cool? And even more importantly, that it might explain why some things in your life behave the way they do? Today, we’re going to peek behind the curtain at three molecules, all siblings in a way, but with surprisingly different personalities. We’re talking about CCl4, CF4, and CBr4. Now, before your eyes glaze over, stick with me! We’re going to make this as easy-going as a Sunday morning coffee.
Imagine you have three siblings. Let’s call them Carl (CCl4), Fiona (CF4), and Brenda (CBr4). They all share the same basic family trait: they’re made of one carbon atom at the center, like the sturdy trunk of a tree, and then they have four other atoms hanging off them, like branches. The only difference is the type of atom that makes up those branches. Carl has four chlorine atoms, Fiona has four fluorine atoms, and Brenda has four bromine atoms. Simple enough, right? They’re all part of the same chemical family, the halogenated methanes.
Now, when we talk about boiling points, we’re essentially talking about how much oomph you need to give something to turn it from a liquid into a gas. Think about boiling a pot of water on the stove. You turn up the heat, the water starts to bubble, and poof! It turns into steam, those invisible wisps you see rising. The boiling point is that specific temperature where this transformation happens.
So, what makes one liquid decide to become a gas at a lower temperature than another? It all comes down to how much the little molecules, like our Carl, Fiona, and Brenda, like to stick together. If they’re really cozy and attached, you’ll need a lot more heat – and therefore a higher boiling point – to pry them apart and send them off as individual gas molecules.
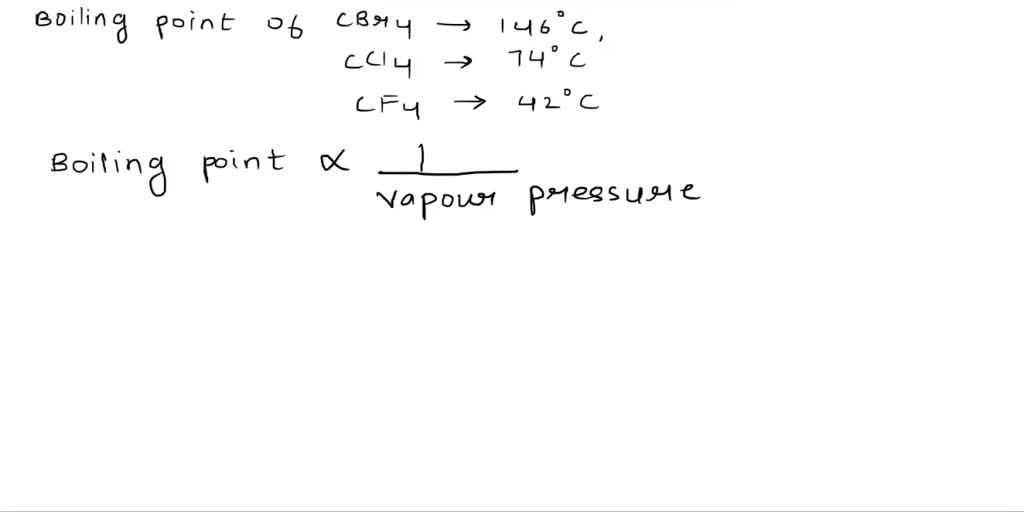
In the case of our carbon siblings, the size and type of the "branches" – the chlorine, fluorine, and bromine atoms – play a big role in how tightly they hold onto each other. Let’s start with Fiona (CF4). Fluorine atoms are the smallest and lightest of the bunch. Because they’re so tiny and don’t have a lot of stuff to them, they don’t really create a big fuss or exert much pull on their neighboring Fiona molecules. It’s like having four very polite and distant cousins at a family gathering. They don’t engage in big hugs or hold hands for too long. This means Fiona is quite happy to become a gas at a very, very low temperature. In fact, it’s so low that at room temperature, Fiona is already a gas! You wouldn’t even see it as a liquid to boil it.

Next up is Carl (CCl4). Chlorine atoms are a bit bigger and heavier than fluorine atoms. Think of them as slightly more boisterous cousins. They have a bit more presence, a bit more ability to interact with each other. This means the CCl4 molecules have a stronger grip on each other than the CF4 molecules. It’s not a super-tight grip, mind you, but it’s enough that you need to add a bit more heat to get them to separate and become a gas. Carl, or CCl4, has a boiling point that’s much higher than Fiona’s (which, remember, is already a gas at room temperature). If you were to hold a container of liquid CCl4, it would feel cool, but it would certainly require some warmth to make it bubble and steam away.
And finally, we have Brenda (CBr4). Bromine atoms are the biggest and heaviest of the three halogen atoms. Imagine these as the really enthusiastic cousins who give big, hearty hugs. Because bromine atoms are so large and have more electrons – those tiny negatively charged particles that orbit atoms – they create a stronger attractive force between the CBr4 molecules. It’s like everyone in the Brenda family is constantly giving each other a friendly squeeze. This means it takes a lot more energy, and therefore a lot more heat, to overcome these strong attractions and make Brenda turn into a gas. Brenda (CBr4) has the highest boiling point of our three siblings. It needs a significant amount of heat to go from liquid to gas.
So, to recap: Fiona (CF4) is the tiny, light one, already a gas. Carl (CCl4) is the middle child, needing a moderate amount of heat to boil. And Brenda (CBr4) is the big, hefty one, requiring the most heat to change from a liquid to a gas.

Why should you, the everyday reader, care about this?
Well, it’s not just about abstract chemistry! These molecules, or molecules very much like them, pop up in our lives in surprising ways. For instance, compounds similar to CCl4 were once widely used as dry-cleaning solvents. Imagine your favorite shirt getting a mysterious stain. A dry-cleaning fluid, which is a liquid, needs to be able to dissolve the stain without damaging the fabric. Then, it needs to evaporate easily afterwards, leaving your shirt clean and stain-free. The boiling point is crucial here! If it evaporated too slowly, your clothes would be left damp and smelling funny. If it evaporated too quickly at too low a temperature, it might not do a good enough job of dissolving tougher stains. Understanding boiling points helps scientists and engineers choose the right tools for the job, whether it's cleaning your clothes or creating specific products.
Think about refrigeration. The cool air in your fridge doesn’t come from magic! It comes from a special liquid that circulates, absorbing heat from inside the fridge and then releasing it outside. This liquid needs to be able to easily turn from a liquid to a gas and back again, a process that relies heavily on its boiling point. If the boiling point is too high, it won’t absorb enough heat. If it’s too low, it might leak out as a gas before it can do its job. So, the careful selection of these chemicals, with their specific boiling points, is what keeps your milk cold and your ice cream frozen!

Even something as simple as safety is involved. Certain CCl4 compounds, for example, were once used in fire extinguishers because they don’t conduct electricity and can smother flames. However, their use was phased out due to environmental and health concerns. But the principle remains: knowing how a substance behaves when heated – its boiling point – is vital for understanding its potential applications and its risks.
Ultimately, understanding these seemingly small differences in molecules helps us appreciate the intricate world around us. It’s about how tiny, invisible particles interact to create the world we see and experience. From the air we breathe to the food we eat, chemistry is at play. And sometimes, just knowing that Carl, Fiona, and Brenda have different boiling points because of the size of their "branches" can unlock a little bit of wonder about the science that shapes our everyday lives.
So, the next time you see a product that seems a bit mysterious, or wonder how something works, remember our three siblings: Carl, Fiona, and Brenda. Their differing boiling points are a perfect little illustration of how the smallest details in chemistry can have a big impact on the world. And that, my friends, is pretty neat!
