Which Has The Higher Boiling Point Hf Or Hcl Why

Ever found yourself staring at two mysterious chemical symbols, like Hf and HCl, and wondered about their secret lives – specifically, their boiling points? It might sound like something straight out of a mad scientist's lab, but understanding why one substance likes to stay liquid longer than another is actually a blast! It’s like being a detective, but instead of solving crimes, you’re uncovering the hidden forces that keep molecules together. This isn't just trivia for your next pub quiz; it's a little peek into the fundamental nature of the world around us, from the water you drink to the air you breathe. Pretty cool, right?
Unlocking the Secrets of Molecular Merriment
So, what’s the big deal about boiling points? Think of it as a measure of how much a substance really wants to stay put in its liquid form. When you heat something up, you’re essentially giving its molecules a burst of energy, making them wiggle and jiggle until they can break free from their liquid bonds and float off as a gas. A higher boiling point means those molecules are holding on tighter, requiring more energy – a hotter temperature – to get them dancing into the gas phase. Understanding this helps us in all sorts of ways:
- Industrial Processes: In factories, precise control over boiling points is crucial for separating different chemicals, purifying substances, and making sure reactions happen just right.
- Everyday Science: Why does oil and water behave so differently? Boiling points are a piece of that puzzle!
- Scientific Discovery: For researchers, knowing these properties is like having a key to understand new materials and predict how they'll act.
It’s all about the invisible forces at play between tiny particles, and it's surprisingly engaging to figure out who’s the stronger "holder-on."
The Showdown: Hf vs. HCl
Now, for the main event! We’re pitting two very different contenders against each other: Hafnium (Hf) and Hydrogen Chloride (HCl). On the surface, they might seem like just letters and numbers, but their boiling points tell a fascinating story about their inner workings.
So, which one throws a bigger party before it turns into a gas? Drumroll, please…
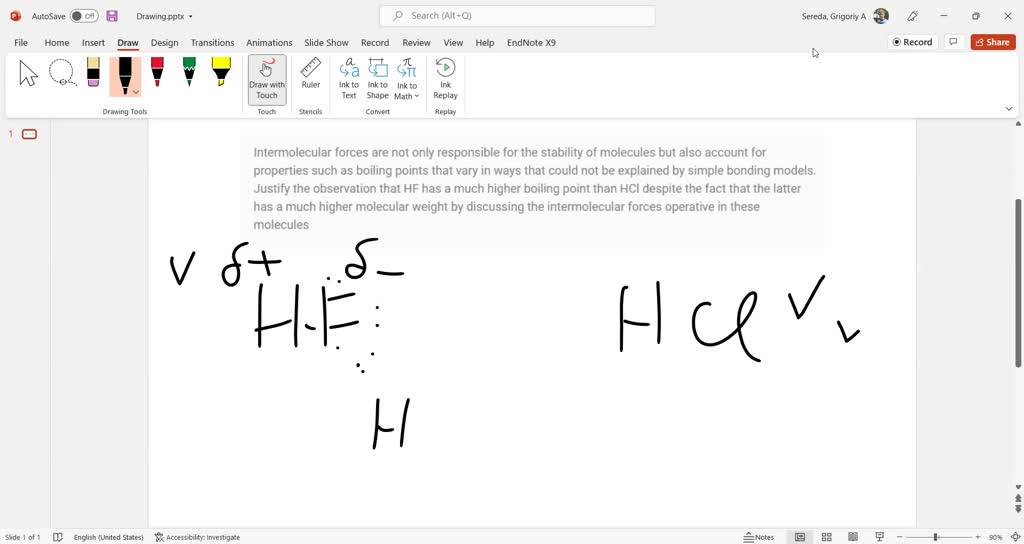
SOLVED: The boiling point of HF is higher than HCl due to ion-dipole
It's Hafnium (Hf) that boasts the significantly higher boiling point. While Hydrogen Chloride (HCl) boils away at a relatively chilly -85.05 °C (-121.09 °F), Hafnium (Hf) doesn't even start to vaporize until a scorching 4603 °C (8317 °F)! That’s a difference so massive, it’s like comparing a mild spring day to the surface of the sun.
The "Why": A Tale of Two Forces
The dramatic difference in their boiling points boils down to the types and strengths of the intermolecular forces holding their particles together.

Let’s start with Hydrogen Chloride (HCl). This is a molecule, made up of hydrogen and chlorine atoms bonded together. Because chlorine is much more electronegative than hydrogen, it pulls the shared electrons closer to itself. This creates a slight, permanent imbalance of charge across the molecule – one end is a tiny bit negative, and the other is a tiny bit positive. This is what we call a polar molecule. These polar molecules are attracted to each other through something called dipole-dipole interactions. Think of it like tiny magnets gently sticking to each other. There are also weaker London dispersion forces at play, which are temporary attractions that happen when electrons are momentarily unevenly distributed. While these forces are present, they are relatively weak. So, it doesn't take a huge amount of energy to overcome these attractions and turn HCl into a gas.
Now, let’s turn our attention to Hafnium (Hf). Hafnium, unlike HCl, is a metal. Metals exist as a giant, organized lattice of positively charged metal ions surrounded by a "sea" of delocalized electrons. The forces holding these metal ions together are incredibly strong. This type of bonding is called metallic bonding. Imagine a vast, interconnected network where positively charged nuclei are held tightly in place by a shared cloud of mobile electrons. This extensive network of strong attractions requires an enormous amount of energy to disrupt. The electrons are not localized to individual atoms but are free to move throughout the entire structure. This "sea of electrons" effectively glues the positive metal ions together with immense force.

Because metallic bonding in Hafnium is so much stronger than the dipole-dipole and London dispersion forces in Hydrogen Chloride, Hafnium atoms are held together much more tightly. Consequently, it takes a vastly higher temperature to give those Hafnium atoms enough energy to break free from their metallic lattice and become a gas.
So, the next time you see those symbols, remember the fascinating story they tell about the strength of their connections! It's a little bit of science magic, revealing why some things boil easily and others are incredibly stubborn.