Which Halogen Has The Least Attraction For Electrons

Alright folks, gather 'round! We’re about to dive into a little scientific mystery that’s probably been keeping you up at night. Or, you know, not. But it’s fun anyway! We’re talking about the halogens. You know, those chatty members of the periodic table. They’re like the life of the party, always looking to make a connection. Specifically, we're going to ponder a burning question: which of these flashy fellows is the least keen on grabbing onto electrons?
Now, when I say "attraction for electrons," picture it like this. Imagine a really popular kid at school. Everyone wants to be their friend. Electrons are like the cool kids, and the halogens are the ones trying to make new buddies. Some halogens are super popular, like, mega-popular. They practically have a fan club for electrons. Others? Well, they’re a bit more… laid back.
We've got Fluorine. Oh, Fluorine! This guy is the superstar. If electrons were pop stars, Fluorine would be Beyoncé. They just flock to Fluorine. It's got that magnetic pull, that undeniable charm. You don’t want to mess with Fluorine’s electron-grabbing game. It’s the undisputed champ. Seriously, if you’re an electron and you have a choice, you’re probably heading straight for Fluorine’s doorstep. It’s like that exclusive club everyone wants to get into.
Then there’s Chlorine. Chlorine is pretty popular too. It’s like the really cool older sibling who throws awesome parties. Lots of electrons are drawn to Chlorine’s energetic vibe. It’s got a strong handshake when it comes to electrons. Not quite Beyoncé level, but definitely a headliner. You see Chlorine around a lot, making connections, forming bonds. It’s a real player in the electron game. It’s reliable, it’s effective, it gets the job done.
Next up is Bromine. Bromine is more like the popular friend of a popular friend. Still cool, still gets attention, but maybe not the absolute center of the universe. Electrons definitely find Bromine attractive. It’s got a good grip, a solid presence. It’s like the dependable actor in a blockbuster movie. You know you’re going to see Bromine involved in things. It’s not shy about its electron desires.
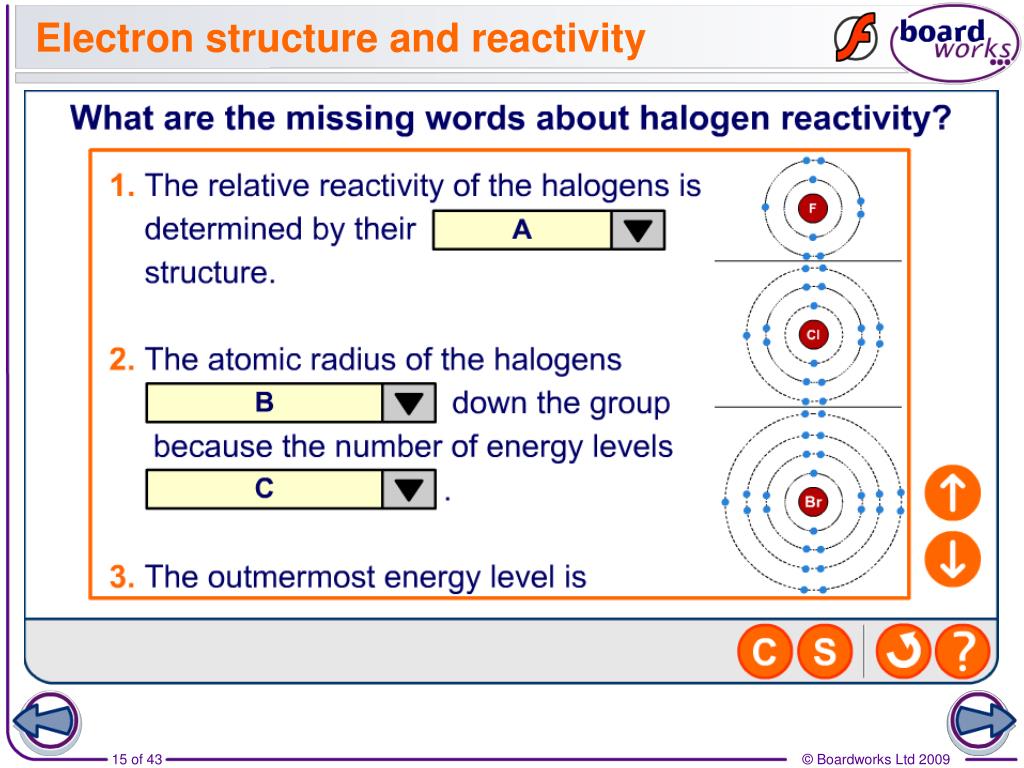
And then we have Iodine. Iodine is interesting. It’s like the wise, slightly more reserved elder statesman. Electrons still find Iodine appealing, but perhaps with a bit more… contemplation. It’s not as frantic as Fluorine, not as boisterous as Chlorine. Iodine has its charms, its own magnetic allure, but it's a more subtle kind of magnetism. It’s the kind of attraction that makes you pause and think, rather than just rush in.
But wait, there’s one more! The grand finale! The one we’ve been building up to. The halogen that, in my humble, and possibly unpopular, opinion, is the most chill when it comes to electrons. The one who’s a bit like, “Eh, electrons, whatever.”
I'm talking about Astatine.
Now, Astatine is a bit of a mystery. It’s rare, it’s fleeting, and it doesn’t stick around for long. Think of it as the eccentric artist of the halogen family. While its cousins are all about grabbing and holding electrons with all their might, Astatine? It’s more of a casual acquaintance. It might let an electron hang out for a bit, but it’s not like it’s going to tie them down. It’s got other, more pressing, radioactivity-related things to worry about, I suppose.
It’s like Astatine is saying, “Sure, you can borrow my electron for a bit, but don’t get too attached. I’m kind of busy disintegrating here.” This makes it, in my book, the halogen with the least intense attraction. It's not that it doesn't attract electrons at all, mind you. It absolutely does. But compared to the almost desperate, ravenous hunger of Fluorine, Astatine’s pull feels more like a gentle suggestion. It’s the quiet whisper in a room full of shouts.
So, while the other halogens are busy playing tug-of-war with electrons, Astatine is off in its own corner, contemplating the universe. It’s got a less aggressive approach. It’s the one who might offer you a cup of tea rather than try to drag you into a mosh pit. And that, my friends, is why, in my whimsical scientific musings, Astatine wins the award for being the halogen that’s the least, shall we say, electron-obsessed.
It’s not the most talked-about member of the family, and it’s definitely not the most stable. But in its own elusive way, Astatine offers a different perspective on electron attraction. It’s the quiet rebel, the laid-back loner. And sometimes, in the chaotic world of chemical bonding, a little bit of laid-back attitude is exactly what you need. So next time you think about halogens, give a nod to Astatine. The one who’s just not that into electrons.
