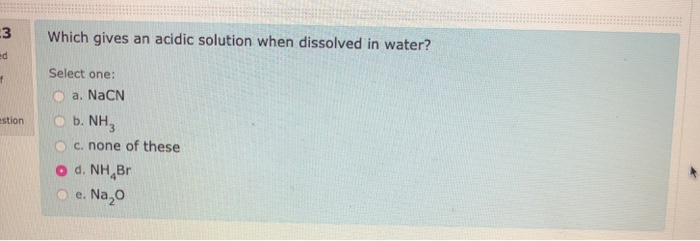
Which Gives An Acidic Solution When Dissolved In Water

Ever wondered why some of your favorite treats make your mouth pucker up like a prune? It’s all thanks to a little chemistry happening right inside them! These everyday heroes, often hiding in plain sight, are masters at creating that delightful tang we all know and love.
Think about your morning coffee. That satisfying jolt isn’t just caffeine; there’s a hint of something else at play. And those juicy lemons you squeeze into your water? Oh boy, they’re practically shouting their acidic secret!
It turns out, a whole cast of characters from your pantry and garden are secretly making your drinks and foods a little more… lively. They’re not trying to be difficult; they’re just doing what they do best when they meet water.
The Zesty Gang
Let’s start with the superstars of the sour world: citrus fruits. Lemons, limes, grapefruits, and oranges are loaded with a special ingredient. When these sunny delights get a splash of water, they unleash their inner zing!
The star player here is a compound called citric acid. It’s like a tiny, energetic sprite that loves to dance in water. This is what gives that intense, refreshing tartness to your lemonade or limeade.
But it’s not just about the taste. This little acid is also a champion at preserving food. It’s part of the reason why jams and jellies can sit on your shelf for so long without going bad. Pretty neat, right?
A Berry Good Story
Moving on to the berry patch, we find more friends contributing to that delicious tang. Think of those raspberries or cranberries. They offer a delightful tartness that balances their sweetness.
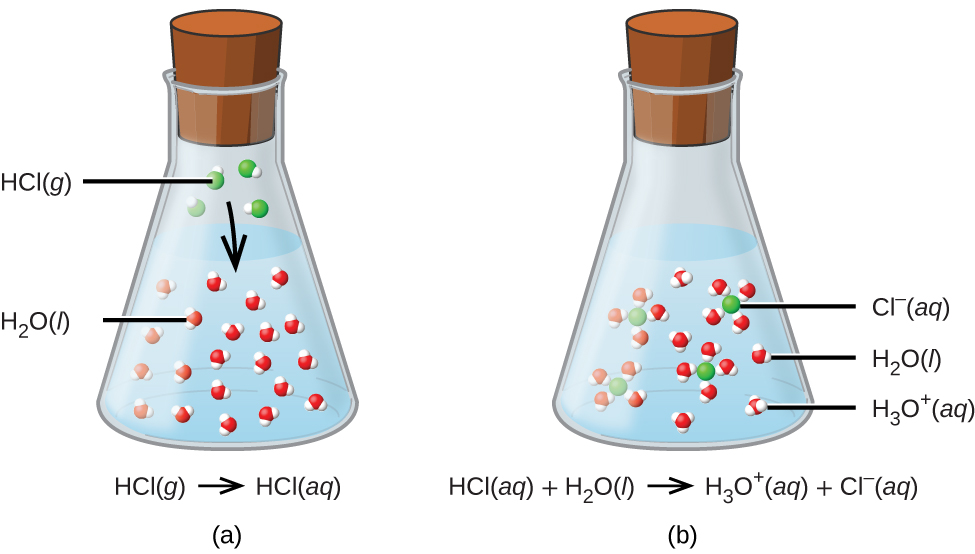
+ions:+HCl+¨+H%2B+and+Cl-.jpg)
These fruits are also packing a punch with their own unique acids. For instance, malic acid is often found in fruits like apples and cherries, but it also plays a role in some berries.
The exact blend of acids varies, making each berry a unique taste experience. It's a little symphony of sour notes that make our taste buds sing.
The Fermentation Funhouse
Now, let’s get a little more adventurous. Have you ever enjoyed a creamy dollop of yogurt or a tangy pickle? These are the results of a fascinating process called fermentation, and acids are the VIPs of this party.
Tiny, invisible friends called bacteria get to work, munching on sugars. As they do their thing, they produce… you guessed it, acids! It’s a delicious transformation that happens behind the scenes.
In yogurt, the main acid produced is lactic acid. This is what gives yogurt its characteristic creamy texture and slightly sour taste. Without lactic acid, yogurt would just be… well, milk!
Pickles get their satisfying crunch and tang from a similar process, often involving acetic acid, which we also know as vinegar. It’s a classic combo that’s been delighting people for centuries.
Vinegar: The Ultimate Pucker-Up Powerhouse
Speaking of vinegar, this liquid gold is practically synonymous with acidity. It’s a staple in kitchens worldwide for a reason. From salad dressings to marinades, vinegar adds that essential zing.
The primary acid in most vinegars is acetic acid. This is a relatively simple molecule, but its impact on flavor is huge. It’s the backbone of so many culinary delights.
It’s fascinating to think that this powerful ingredient starts with something as simple as alcohol being transformed by friendly bacteria. It’s like a magic trick for your taste buds!
The Soapy Secret
Now for a surprising twist. Ever wondered why some soaps can feel a bit harsh or why certain cleaning agents work so well on grease? Acids play a role here too, though perhaps not in the way you’d expect for consumption!
When we talk about acidic solutions in a general sense, it’s important to remember the broader chemical context. Some substances, when dissolved in water, create solutions that are acidic, and these can have very different properties.
Think about the chemical reactions involved in cleaning. Acids can help break down stubborn grime and lift away dirt. It's a powerful partnership between water and the right chemical properties.
The Unexpected Sweetener
And what about that sugary delight, honey? While it’s known for its sweetness, honey also contains small amounts of various acids. These contribute to its complex flavor profile and help with its natural preservation qualities.

The acids in honey, along with its high sugar content, create an environment that’s not very friendly to spoilage-causing microbes. It’s nature’s own little preservative, sweet and tangy all at once.
So, the next time you enjoy a spoonful of honey, remember that there’s a subtle hint of acidity working its magic!
A World of Tang and Pucker
It’s truly amazing how these common ingredients, when dissolved in water, create these vibrant, tangy solutions. They add zest to our food, help preserve our goods, and even contribute to cleaning.
From the sunny burst of a lemon to the subtle tang of yogurt, acidity is a fundamental part of our everyday lives. It’s a reminder that even simple things can have complex and wonderful properties.
So, embrace the pucker! These acidic wonders are what make our world a little more flavorful, a little more interesting, and a whole lot more delicious. Cheers to the unsung heroes of the acidic solution!
