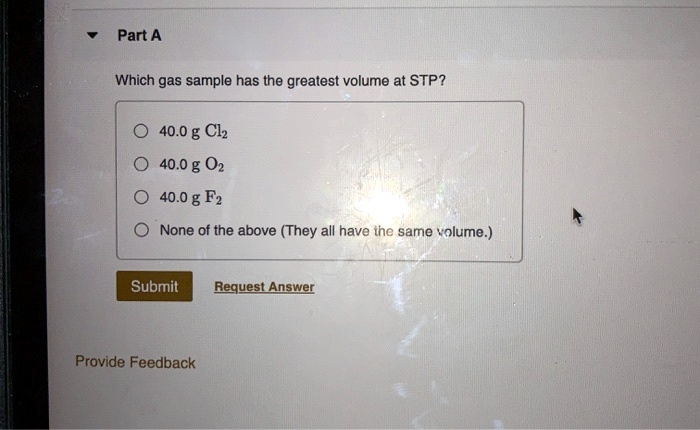
Which Gas Sample Has The Greatest Volume At Stp
Hey there, science curious folks! Ever wonder about those invisible gases all around us? They're more fascinating than you might think. Today, we're diving into a super fun question that might sound a bit like a riddle: Which gas sample has the greatest volume at STP? Sounds technical, right? But trust me, it’s a cool little puzzle with a surprisingly simple and delightful answer.
First off, let’s break down that fancy acronym: STP. It just stands for Standard Temperature and Pressure. Think of it as the VIP section for gases. It's a specific set of conditions where scientists like to compare how gases behave. It’s like having a universal playground where all the gas samples are playing under the same rules. So, when we say "at STP," we're just setting the stage for our gas party!
Now, for the big question: Which gas sample has the greatest volume? You might be picturing different gases like helium in balloons, oxygen we breathe, or maybe even something a bit more exotic. You might be thinking, "Does a heavier gas take up less space? Or does a lighter gas puff out more?" It’s tempting to guess based on what we see or what we know about everyday objects. We often associate size with how much "stuff" is inside, right? More stuff, bigger container. But gases? They play by slightly different rules.
Here's where the magic of gases at STP comes in. It turns out, when you have the same amount of different gases at the same temperature and pressure, they all take up the exact same amount of space! Mind-blowing, isn't it? It doesn't matter if it's helium, oxygen, nitrogen, or even a really heavy gas like xenon. If you have one mole of each, they will all occupy the same volume at STP. This is a fundamental concept in chemistry, and it's called Avogadro's Law. It’s like a universal gas pact!
So, why is this so cool? It means that the identity of the gas doesn't actually matter when we're talking about equal amounts at STP. It’s not about the weight or the type of gas. It’s about the number of particles. This law is incredibly powerful because it simplifies so many calculations and predictions about gases. Imagine if every gas behaved differently at the same conditions! Chemistry would be a chaotic mess. But thanks to Avogadro’s Law, things are wonderfully predictable.

Think of it this way: Imagine you have a bunch of identical boxes, all the same size. Now, imagine filling those boxes with different kinds of marbles – tiny little ones, medium-sized ones, and even slightly bigger ones. If you make sure you put the exact same number of marbles in each box, even though the marbles are different, each box will be "full" in the same way. The gases at STP are kind of like those marbles. The particles are different sizes, but if you have the same number of them (which we usually measure in moles), they push out and fill their container (their volume) to the same extent.
This might challenge your everyday intuition. We’re used to thinking that bigger things are heavier, and smaller things are lighter. But with gases at STP, the focus shifts from the individual particle's size or weight to the total number of particles. It's a subtle but crucial difference that makes the world of gases so intriguing.
The standard volume for one mole of any gas at STP is approximately 22.4 liters. Yes, you read that right. 22.4 liters! That’s a significant chunk of space. So, if you have one mole of hydrogen gas, it will occupy 22.4 liters. If you have one mole of carbon dioxide, it will also occupy 22.4 liters. And if you have one mole of argon? You guessed it – 22.4 liters!
This universality is what makes this concept so special. It’s a constant, a reliable fact that scientists can always count on. It’s like finding a secret key that unlocks understanding about all gases, regardless of their individual characteristics. It’s a testament to the elegant simplicity that can exist in the universe, even when dealing with something as unseen as gas.

So, to answer our fun little riddle: Which gas sample has the greatest volume at STP? The answer is: they all have the same volume, provided they have the same number of particles (i.e., the same number of moles)! It’s a delightful twist that emphasizes the power of collective behavior in gases. It’s a concept that’s not just confined to dusty textbooks; it has real-world applications in everything from understanding weather patterns to designing chemical processes.
Isn't that neat? The next time you see a balloon, or think about the air you breathe, remember this cool gas fact. It’s a small piece of science that opens up a big understanding of how our gaseous world works. It’s a reminder that sometimes, the most profound answers are the simplest ones, and that even invisible things can hold incredible, consistent wonders.
The key takeaway? At STP, if you have the same number of particles, you have the same volume! It's a universal truth for gases.
So, there you have it! A little journey into the captivating world of gases. It’s a bit like discovering a secret handshake for all gas particles. Pretty cool, right? It makes you wonder what other hidden simplicities are waiting to be discovered in the science all around us. Keep your eyes (and your mind!) open!
