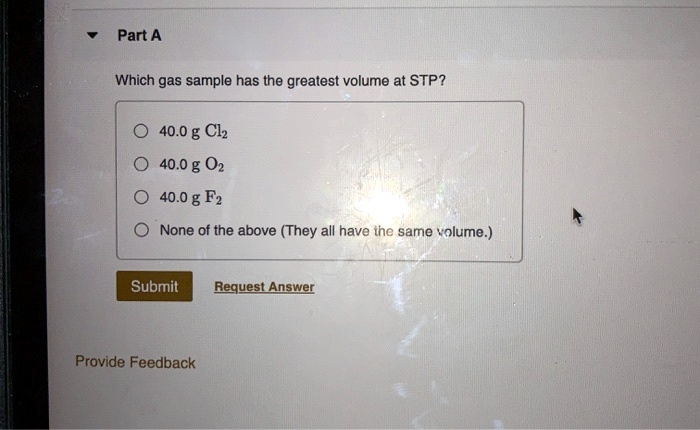
Which Gas Has The Greatest Kinetic Energy At Stp

Imagine a big party happening in a room full of tiny, bouncing balls. These aren't just any balls; they're gas molecules, zipping around and bumping into each other. We're talking about a special kind of party, held at what scientists call STP – that's a fancy acronym for a very specific chill-out zone for these gas molecules.
Now, each of these little partygoers is full of energy. This energy, the energy of their wild, zoomy dance, is called kinetic energy. Think of it as their enthusiasm for life, their "go-get-'em" spirit.
At this STP party, we have a bunch of different types of gas molecules. Some are big and clunky, like a group of enthusiastic golden retrievers. Others are sleek and speedy, like tiny, energetic hummingbirds. And some are just plain simple, like a single, perfectly formed droplet of dew.
The big question is: which of these gas molecules is having the most energetic fun? Who's dancing the fastest, bumping the hardest, and generally throwing the biggest energy party?
The Mystery of the Speedy Sprites
This is where things get a little surprising. You might think the biggest, heaviest molecules would have the most kinetic energy, right? They seem like they should be the kings and queens of the dance floor.
But, in the world of gases at STP, it's not always about size. It's more about how much "oomph" each individual molecule has. And that "oomph" is influenced by a few things, but at STP, a crucial factor is the temperature.
At STP, the temperature is set to a cozy 0 degrees Celsius, which is pretty cool, but not freezing. This temperature is like the master conductor of the molecular orchestra, making sure everyone plays at a certain tempo.
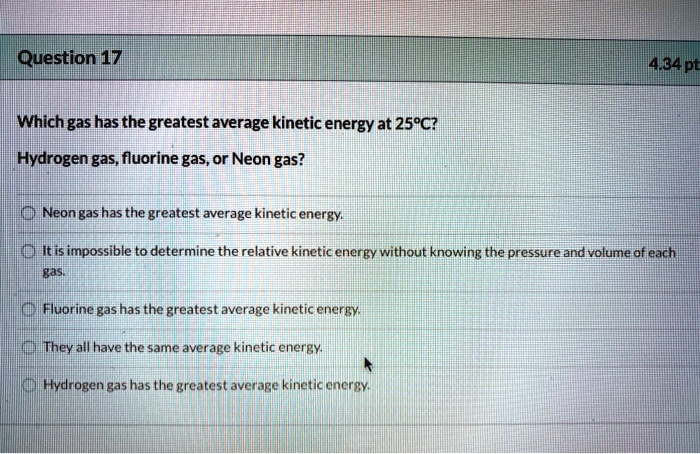
The Surprising Winner: It's All About the Average
Here's the really neat part. At STP, every type of gas molecule, no matter how big or small, actually has the same average kinetic energy. Yes, you read that right!
It's like at this particular party, the host has a rule: everyone gets the same amount of pizza slices, regardless of how many legs they have or how fluffy their fur is. Everyone gets the same share of energy!
But wait, there's a twist in this delightful tale. While the average kinetic energy is the same for all gases at STP, the speed at which individual molecules move can be quite different. This is where the size of the molecule really starts to shine.
"It's a bit like a playground race. Everyone gets the same amount of energy to run, but the smaller kids can zoom around much faster than the bigger ones covering the same distance with that energy."
So, while a big, heavy molecule like xenon might be lumbering along with its share of the energy, a light, zippy molecule like hydrogen will be practically a blur. It's using its assigned energy allowance to zip and zoom with all its might.
The Heartwarming Tale of Lightness and Speed
Think of hydrogen as the little engine that could. It's super light, almost like a tiny feather. So, when it gets its energy from the STP environment, it can convert that energy into a whole lot of speed.
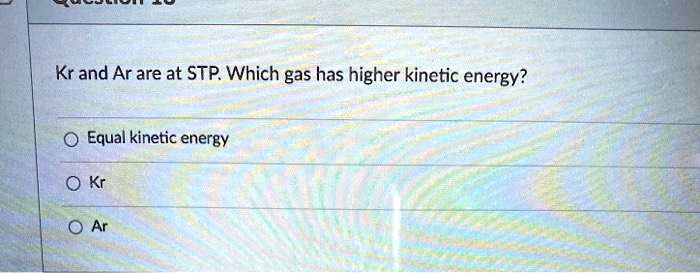
On the other hand, a gas like methane, which is a bit more substantial, will use the same amount of energy but won't reach those dizzying speeds. It's more like a steady jogger than a sprinter.
And then you have the really hefty gases, like radon, which is like the gentle giant of the gas world. It has the same energy allowance as hydrogen, but its sheer mass means it moves much, much slower.
So, if we're talking about individual molecules and how fast they're darting around, the gas with the greatest kinetic energy per molecule at STP is the one that's best at turning that energy into motion. That, my friends, is our speedy little hero, hydrogen!

It's a heartwarming thought, isn't it? That even the smallest, lightest among us can achieve incredible speeds and have the most exciting journey, all thanks to a little bit of shared energy. They might not be the biggest, but they're definitely the most enthusiastic dancers on the STP floor.
A World of Dancing Molecules
This dance of molecules is happening all around us, all the time. Whether it's the air we breathe, the helium in a balloon, or the steam rising from a kettle, these tiny particles are in constant motion.
Understanding their kinetic energy helps us understand so much about the world, from how engines work to why some gases are more flammable than others. It’s like peeking behind the curtain at a grand, invisible ballet.
So next time you think about gases, remember this party at STP. Remember the little hydrogen molecule, zipping and zooming with all its might, a testament to the fact that sometimes, being light and agile is the most energetic way to be. It’s a beautiful reminder of how even the smallest things can have the biggest impact, or in this case, the most spirited motion.
