Which Formula Represents A Polar Molecule Containing Polar Covalent Bonds
Ever feel like some things in life just have a split personality? Like that one friend who's super chill one minute and then suddenly gets all fired up about pineapple on pizza? Well, guess what? Molecules can be like that too! They can have a bit of a tug-of-war going on inside, and that, my friends, is where our topic for today struts onto the scene. We're diving into the wonderfully weird world of polar molecules, specifically those that are rocking some polar covalent bonds. Don't let the fancy science words scare you; we're going to break it down like a piñata at a birthday party – messy, fun, and ultimately rewarding.
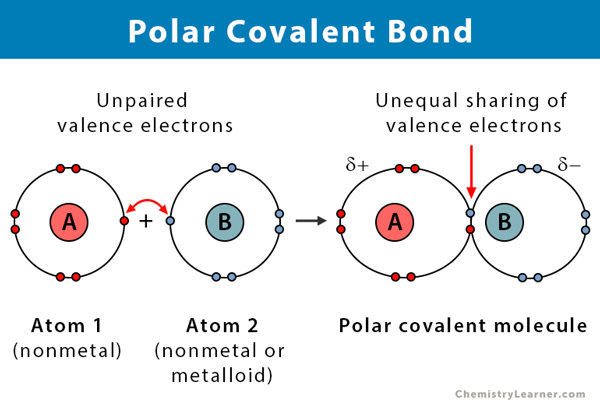
Think of a molecule like a tiny little team of atoms. They’re all linked up, like your family on a road trip. Now, in a covalent bond, atoms share electrons. It's like two siblings agreeing to share a video game controller. "You play for ten minutes, then I play for ten minutes." Fair enough, right? They're pretty much on equal footing.
But sometimes, in this molecule road trip, one atom is just a little bit greedier with the electrons. It's like the sibling who always hogs the charger, even when their phone is at 99%. This "greedier" atom has a higher electronegativity. Electronegativity is basically a fancy way of saying how much an atom wants to hog electrons. So, when two atoms with different appetites for electrons decide to share, things get a little unbalanced. This, my friends, is a polar covalent bond.
Imagine a tug-of-war. In a nonpolar covalent bond, it's like two evenly matched teams, maybe the Avengers vs. the Justice League. The rope (electrons) is pretty much in the middle. But in a polar covalent bond, it's like the Avengers vs. a lone, super-powered Groot. Groot's going to pull that rope a lot harder, right? The electrons are going to spend more time hanging out with the "Groot" atom.
This unequal sharing creates a little bit of an imbalance, like a seesaw where one side is a bit heavier. The atom that hogs the electrons gets a partial negative charge (we write this with a little Greek letter delta, δ-, which looks like a funky 'd'). The atom that's a bit less "electronegative" ends up with a partial positive charge (δ+). It’s not a full-on positive or negative like in an ionic bond (where one atom completely steals the electron, like a thief in the night), but it's enough to make a difference. It’s like one side of the seesaw has a permanent frown and the other has a permanent smile.
Now, for a molecule to be considered a polar molecule, it's not just about having polar covalent bonds. The shape of the molecule matters a whole lot. Think of it like this: you can have a bunch of grumpy people in a room, but if they're all spread out evenly, the overall mood might not feel that bad. But if they're all crammed into one corner, then yeah, it's going to feel pretty intense.
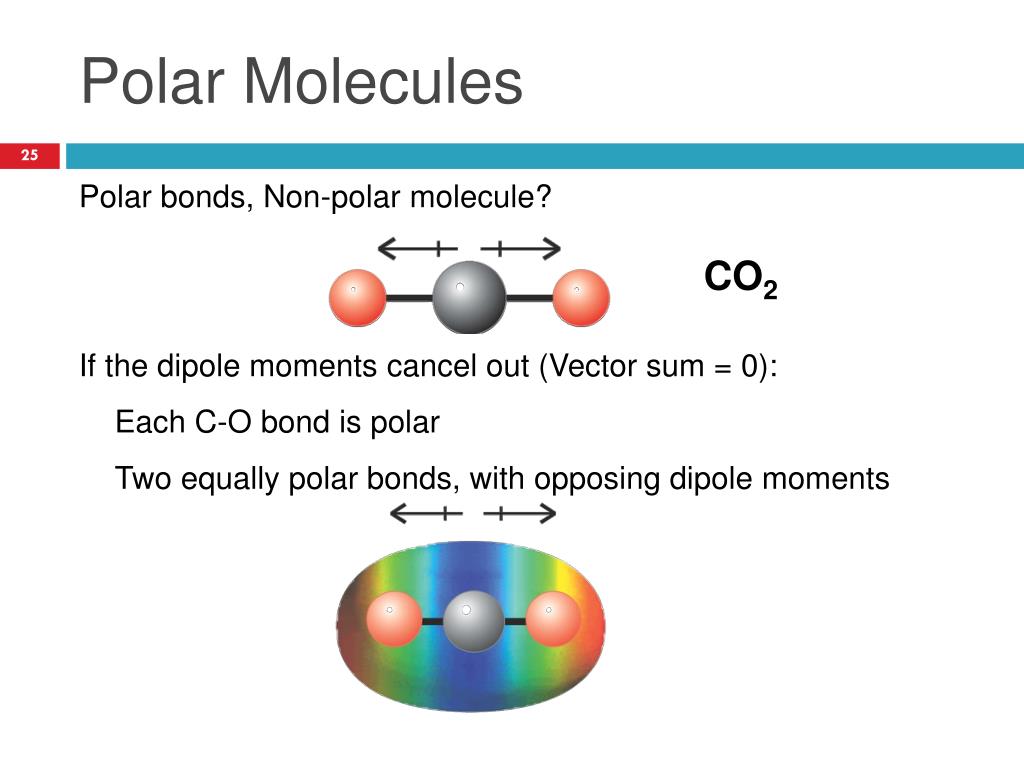
So, a molecule with polar covalent bonds can sometimes be nonpolar if its shape is symmetrical. Imagine a molecule like carbon dioxide (CO₂). It's got two polar covalent bonds, with oxygen being more electronegative than carbon. So, you've got these little electron tugs going on. But, the molecule is shaped like a straight line: O=C=O. The tugs are happening in opposite directions with equal force. It's like two strong people pulling a rope in a perfect line – the net effect is zero. The molecule as a whole doesn't have a distinct positive or negative end. It's like having two grumpy uncles on opposite ends of the room; the overall vibe isn't necessarily negative.
But then you have molecules where the polar covalent bonds are arranged in a way that doesn't cancel out. This is where our star, the polar molecule, shines (or rather, has a slight electrical charge). The classic example is water (H₂O).
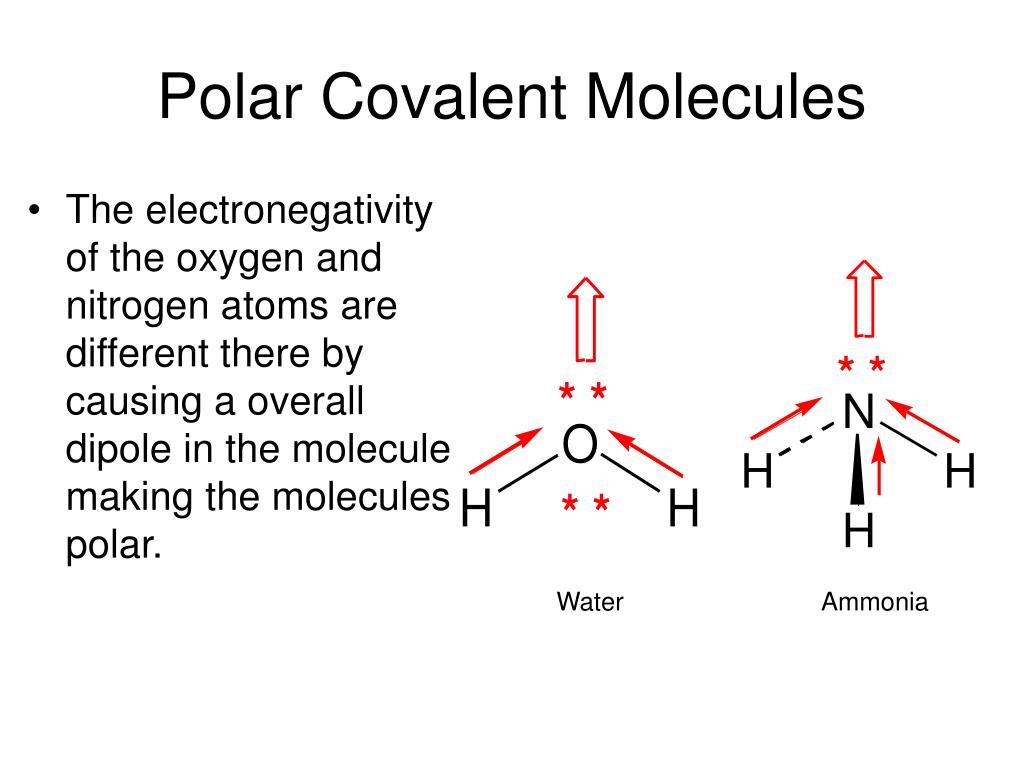
Water is like the life of the party. It's made of one oxygen atom and two hydrogen atoms. Oxygen is way more electronegative than hydrogen. So, in each O-H bond, oxygen is pulling those electrons closer. This creates a partial negative charge on the oxygen and partial positive charges on the hydrogens.
Now, the water molecule isn't a straight line. It's bent, like a little Mickey Mouse head (with the oxygen as the head and the hydrogens as the ears). Because of this bent shape, the partial negative charge is concentrated on one side (the oxygen side), and the partial positive charges are on the other side (the hydrogen side). It’s like having a magnet with a clear north pole and a clear south pole. This makes water a polar molecule!
This polarity is what makes water so amazing. It's like having a tiny little superhero with a good side and a slightly less good side. This makes water really good at dissolving things. Think about it: when you put salt (an ionic compound, which is very polar) in water, the positive ends of the water molecules are attracted to the negative chloride ions, and the negative ends of the water molecules are attracted to the positive sodium ions. It’s like water giving a big hug to the salt ions and pulling them apart. This is why water is often called the "universal solvent."

So, what formula represents a polar molecule containing polar covalent bonds? Well, it's not a single, magical formula like "ABC=Polar!" It's more about looking at the chemical formula and then understanding the molecular geometry. The chemical formula tells you what atoms are in the molecule and how many of each. For example, H₂O tells us we have two hydrogens and one oxygen. CCl₄ tells us we have one carbon and four chlorines.
But then you need to know the VSEPR theory (Valence Shell Electron Pair Repulsion theory). This is the science behind why molecules have the shapes they do. Basically, electron pairs around an atom want to get as far away from each other as possible, like toddlers on a bouncy castle. This repulsion dictates the molecule's geometry.
For our polar covalent bond and polar molecule discussion, we're looking for molecules where: 1. There are polar covalent bonds present. This means you need atoms with significantly different electronegativities bonded together. Think of common pairings like O-H, N-H, C-O, C-Cl, etc. If all the bonds are between identical atoms (like in O₂ or N₂), then there are no polar covalent bonds, and the molecule is nonpolar.
2. The molecular geometry is asymmetrical. This is the key to the whole "polar molecule" thing. Even if you have polar bonds, if the molecule is shaped symmetrically, the bond polarities will cancel each other out.
Let's look at some examples to really drive this home. We already talked about water (H₂O). It has two O-H polar covalent bonds, and its bent shape makes it asymmetrical, so it's a polar molecule.

How about ammonia (NH₃)? It has three N-H polar covalent bonds. Nitrogen is more electronegative than hydrogen. The molecule has a pyramidal shape, like a little tripod. Because of this shape, the bond polarities don't cancel out. The nitrogen atom has a partial negative charge, and the hydrogen atoms have partial positive charges. So, NH₃ is a polar molecule!
Now, consider methane (CH₄). Carbon and hydrogen have similar electronegativities, so the C-H bonds are considered largely nonpolar (or only slightly polar). Even if they were a bit polar, methane has a tetrahedral shape, which is very symmetrical. Imagine a perfectly balanced pyramid. Any slight pull in one direction is perfectly counteracted by a pull in the opposite direction. So, CH₄ is a nonpolar molecule.
What about sulfur dioxide (SO₂)? It has polar covalent bonds between sulfur and oxygen. It's a bent molecule, similar to water. The polarities of the S-O bonds don't cancel out. So, SO₂ is a polar molecule.
The general "formula" or rather, the representation that allows us to understand if a molecule is polar and has polar covalent bonds involves: * The chemical formula (e.g., H₂O, CO₂, NH₃). * Knowledge of the electronegativities of the atoms involved to determine if the bonds are polar covalent. * Understanding of the molecular geometry, often determined using VSEPR theory, to see if the polarities of the bonds cancel out.
So, when you see a chemical formula, you can't just say, "Yep, that's polar!" You have to do a little bit of detective work. It’s like looking at a recipe. You see the ingredients (the chemical formula), but you also need to know the cooking method (the molecular geometry) to know if the final dish will be a symphony of flavors (polar) or a bit of a bland mash-up (nonpolar).
The representation is really the combination of the chemical formula and the 3D model or understanding of its shape. For example, for water, the representation would be H₂O, and we'd know its bent shape. For carbon dioxide, it would be CO₂, and we'd know its linear shape.
The "formula" that describes a polar molecule containing polar covalent bonds is essentially the entire picture: the types of atoms, how many of them there are, and how they are arranged in three-dimensional space. It’s not a simple equation, but a concept built from understanding atomic properties and spatial arrangements.
So, next time you see a chemical formula, remember it’s not just letters and numbers. It’s a tiny universe with its own rules of sharing, tug-of-war, and personality. And understanding whether it’s a polar molecule or not can tell you a whole lot about how it’s going to interact with the world, or at least with other molecules. It's like knowing if your friend is going to share their snacks or hoard them all – it makes a big difference!
In essence, a polar molecule containing polar covalent bonds is one where the electron sharing is uneven (polar covalent bonds) and this unevenness isn't balanced out by symmetry (leading to a polar molecule overall). It's the molecular equivalent of a perfectly imperfect situation, and it’s what makes so much of chemistry, and life itself, so fascinating.
