Which Formula Represents A Nonpolar Molecule Containing Polar Covalent Bonds
Hey there, science curious folks! Ever wondered how some molecules can be made of bits that really like to share electrons unevenly, creating little electrical tug-of-wars, yet the whole molecule acts like it's totally chill and doesn't have a charge anywhere? It’s a bit like a really balanced team where everyone has their own strengths and weaknesses, but when you look at the team as a whole, they're just… a team, no standout hero or villain in sight. Pretty neat, right?
Today, we're diving into a fascinating corner of chemistry: the world of nonpolar molecules that contain polar covalent bonds. Sounds like a mouthful, I know, but stick with me! It's actually a super cool concept that explains a lot about how the world around us works.
The Nitty-Gritty of Covalent Bonds: Sharing is Caring (Mostly)
First things first, let's quickly touch upon what a covalent bond is. Imagine two atoms holding hands. In chemistry, that "hand-holding" is all about sharing electrons. They're like roommates sharing a pizza – everyone gets a slice, or in this case, an electron.
Now, sometimes, this sharing isn't exactly fair. One atom might be a bit greedier, or perhaps it's just better at attracting those shared electrons. This is where we get polar covalent bonds. Think of it like one roommate who really loves pepperoni and always grabs the biggest slices, leaving fewer for the other. So, one atom ends up with a slightly negative charge (because it has more electron love), and the other atom ends up with a slightly positive charge. This creates a little dipole, a tiny separation of charge, within that specific bond. It's like a mini-drama happening between two atoms.
Examples of these uneven sharers are everywhere. Oxygen is notorious for hogging electrons, which is why water (H₂O) has polar bonds. The oxygen atom pulls the electrons closer to itself, leaving the hydrogen atoms with a bit of a positive buzz.
So, Where Does Nonpolar Come In?
If we have these little charge imbalances (polar bonds), how can a whole molecule be nonpolar? This is the really mind-bending part! It all comes down to molecular geometry – how the atoms are arranged in three-dimensional space.

Imagine you have a group of people in a room, and each person is carrying a certain amount of something. If they're all standing in a scattered mess, you might be able to see who has more or less. But, if they all arrange themselves in a perfectly symmetrical circle, with people carrying equal amounts facing each other across the circle, the overall distribution of what they're carrying might be perfectly balanced. The individual differences cancel each other out.
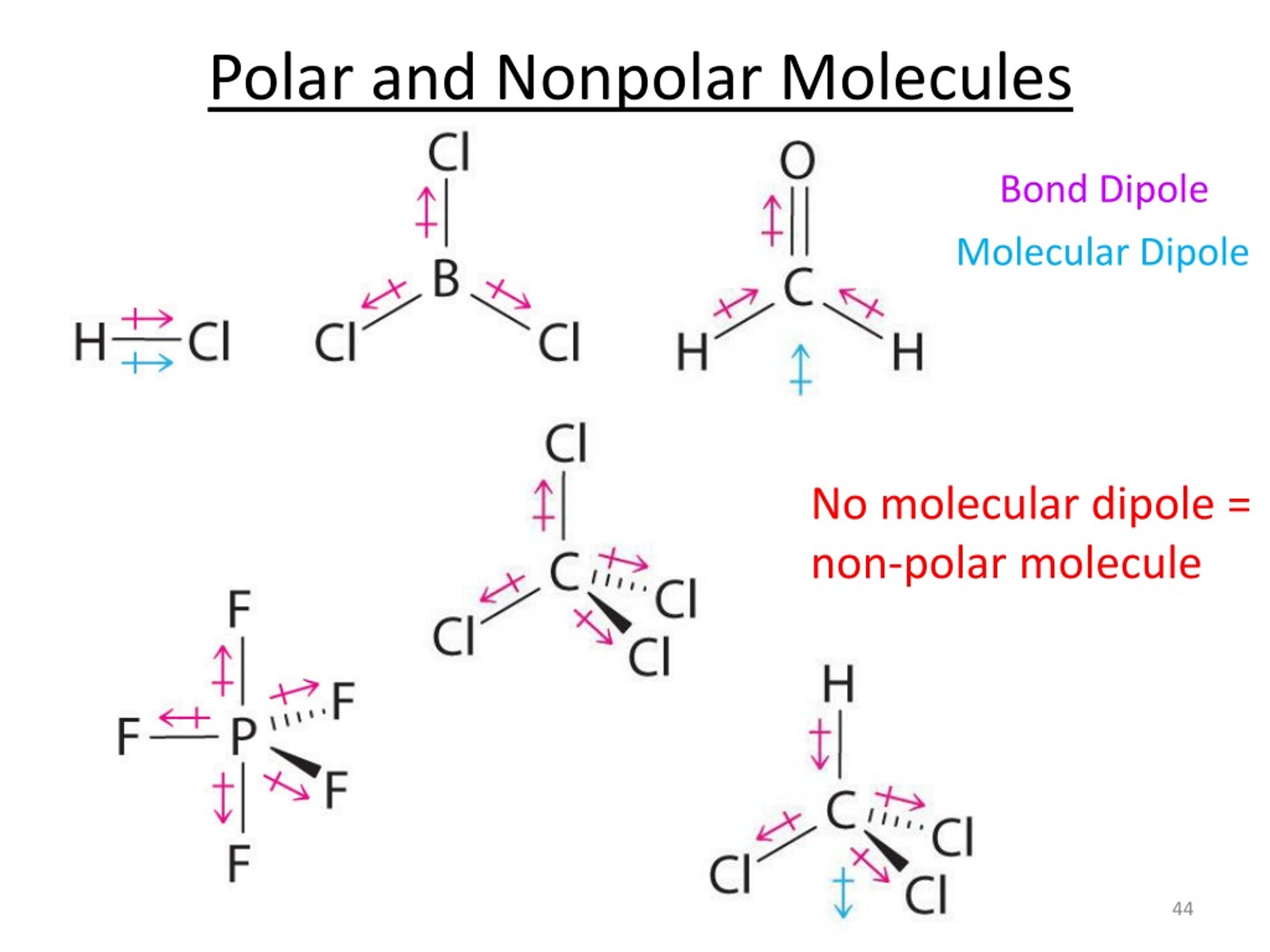
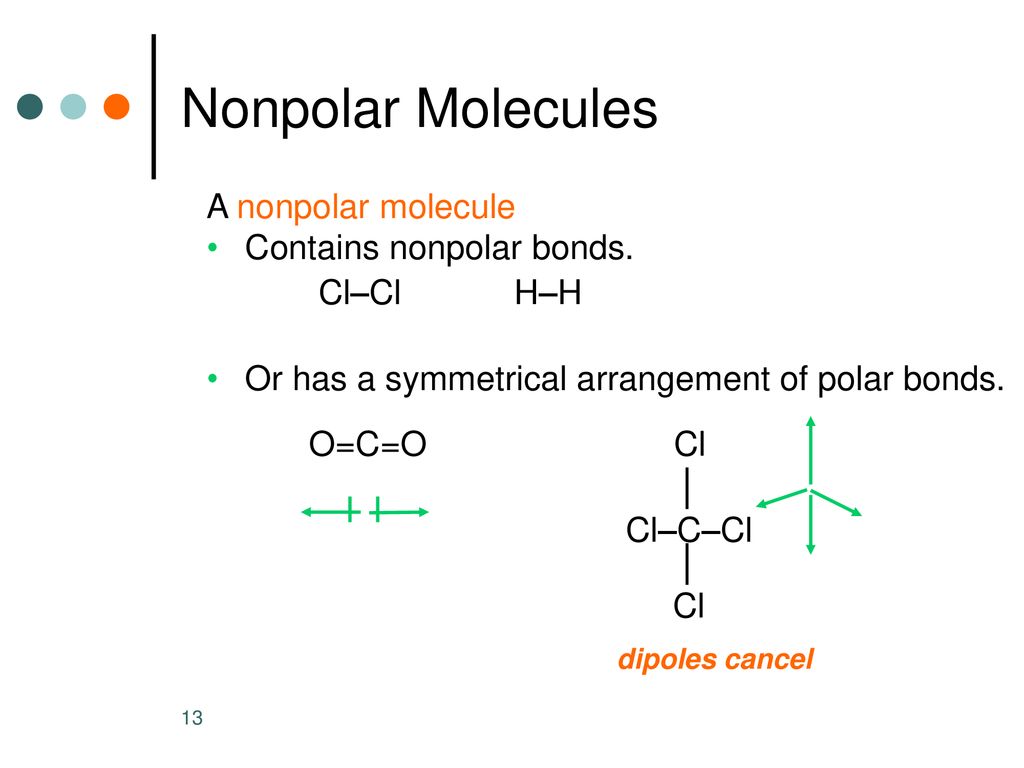
This is exactly what happens in molecules with polar covalent bonds. If the molecule is symmetrical enough, all those tiny dipoles, those little tugs of war, end up pointing in directions that perfectly cancel each other out. The positive ends are balanced by negative ends, and vice versa. So, even though there are polar bonds within the molecule, the molecule as a whole has no net charge. It’s like a perfectly balanced seesaw – each side is going up and down, but the whole thing stays level.
The Master of Symmetry: Carbon Dioxide (CO₂)
A classic example of this phenomenon is carbon dioxide (CO₂). You’ve heard of it, right? It’s what we exhale. Now, oxygen is super electronegative, meaning it likes to pull electrons towards itself. So, in CO₂, each carbon-oxygen bond is indeed polar. The oxygen atoms pull electrons away from the central carbon atom, making the oxygens slightly negative and the carbon slightly positive.
But here’s the magic: the CO₂ molecule is linear. The carbon atom is in the middle, and the two oxygen atoms are on either side, perfectly spaced out. So, you have a dipole pointing towards one oxygen, and an identical dipole pointing in the exact opposite direction towards the other oxygen. These two opposing dipoles are equal in strength and opposite in direction, so they perfectly cancel each other out. Poof! The entire CO₂ molecule is nonpolar, even though it's packed with polar covalent bonds.
It’s like having two identical balloons, each filled with slightly different colored water. If you tie them together at the ends and let them float, the colors might mix in the balloons, but the overall balloon system remains balanced and doesn't drift towards one color over the other.
Other Cool Examples
What about methane (CH₄)? Carbon forms covalent bonds with four hydrogen atoms. Carbon is a bit more electronegative than hydrogen, so the C-H bonds are technically slightly polar. However, the methane molecule has a beautiful tetrahedral shape. Imagine the carbon atom at the center of a pyramid, with the four hydrogen atoms at the four corners. This symmetrical arrangement means that the individual bond polarities all point outwards and cancel each other out. So, methane is another great example of a nonpolar molecule with polar bonds.
Another one is boron trifluoride (BF₃). The B-F bonds are polar because fluorine is very electronegative. But, BF₃ has a trigonal planar shape, with the boron in the center and the three fluorine atoms arranged in an equilateral triangle around it. Again, the symmetry is key! The three polar bonds, equally spaced and pointing away from the center, perfectly neutralize each other. The molecule is nonpolar.
Why Does This Even Matter?
You might be thinking, "Okay, cool symmetry, but why should I care?" Well, this property of molecules being nonpolar, despite having polar bonds, has huge implications!
Solubility is a big one. Remember the saying, "like dissolves like"? Polar molecules tend to dissolve in other polar substances (like salt in water), and nonpolar molecules tend to dissolve in other nonpolar substances (like oil dissolving in gasoline). If a molecule looks polar because of its bonds, but is actually nonpolar due to symmetry, it will behave differently when you try to mix it with things. This affects everything from how medicines are absorbed by our bodies to how paints mix and dry.

Intermolecular forces – the "sticky" forces between molecules – are also affected. Nonpolar molecules generally have weaker intermolecular forces (called London dispersion forces) compared to polar molecules (which have dipole-dipole interactions and sometimes hydrogen bonding). This means nonpolar substances often have lower melting and boiling points. Think about how easily propane gas (a nonpolar molecule) vaporizes compared to water!
It’s also crucial for understanding things like cell membranes, which are made of lipids (fats) that are largely nonpolar. The nonpolar nature allows them to form barriers that separate different environments within our cells and in our bodies. The way these membranes function relies heavily on the nonpolar character of their building blocks, which often contain polar covalent bonds within their structure.
So, the next time you encounter a molecule, don't just look at the individual bonds. Take a step back and consider the overall shape. It’s the geometry that often holds the secret to whether a molecule, with its tiny internal charge dramas, can still present a perfectly neutral, nonpolar face to the world. Pretty cool, right? It's a beautiful illustration of how, in science as in life, the big picture can sometimes be very different from the sum of its parts.
