Which Equation Represents A Combustion Reaction
Ever watched a campfire crackle and wondered what's actually going on? It's more than just pretty flames! There's a whole science show happening, and it's called a combustion reaction. Think of it as a fiery dance of molecules.
And guess what? There's an equation that perfectly captures this amazing show! It's like the script for the fire, telling us exactly what goes in and what comes out. It's a neat little puzzle that scientists figured out ages ago.
So, what makes this particular equation so darn entertaining? It's all about the drama! We start with certain ingredients, and then, BAM! Something entirely new and exciting appears. It’s like a magic trick, but with real chemistry.
The Superstar Ingredients
Every good show needs its stars, right? For combustion, our main stars are a fuel and an oxidizer. Usually, the fuel is something that can burn easily, like wood, gas, or even a tiny bit of candle wax. It’s the stuff that’s going to be transformed.
And the oxidizer? Most of the time, this is oxygen from the air all around us. Oxygen is super keen to react with things, and when it meets a fuel, sparks can fly! It’s like a perfect match made in the chemical world.
Imagine a tiny, eager oxygen molecule just waiting to grab some bits from the fuel. It's got that energetic vibe that makes things happen. Without oxygen, most of our everyday fires wouldn't even get started.
The Fiery Transformation
Now for the main event! When the fuel meets the oxygen, a chemical reaction kicks off. It's a bit like a molecular hug that goes a little too wild. Bonds break, and new bonds form, all happening at super-speed.
This is where the fun really begins. The old molecules are rearranged to create something completely different. It’s not just a little change; it’s a full-blown makeover! And the energy released? That’s what gives us heat and light.

Think about lighting a match. The chemicals on the match head are the fuel, and the oxygen in the air is the oxidizer. A tiny bit of friction gives them the nudge they need to start their fiery transformation.
The Spectacular Results
So, what are the results of this incredible show? We typically get two main things: carbon dioxide and water. Yes, the stuff we breathe out and the stuff that falls from the sky! It’s a reminder that even the most dramatic reactions can produce everyday substances.
But wait, there's more! Combustion also often releases a ton of energy. This is the heat that warms us and the light that lets us see. It’s the powerhouse of the reaction, making it so useful and, well, visually spectacular.
Sometimes, if the burning isn't perfect, you might also get other things like carbon monoxide (which is a bit sneaky and dangerous!) or tiny soot particles. It’s like the less glamorous side of the chemical performance.
The equation for combustion is like a recipe for fire!
The Star Equation Itself
Now, let's talk about the equation that pulls it all together. It's usually written in a specific format, showing the ingredients on one side and the results on the other. It’s like a balanced scorecard for the reaction.
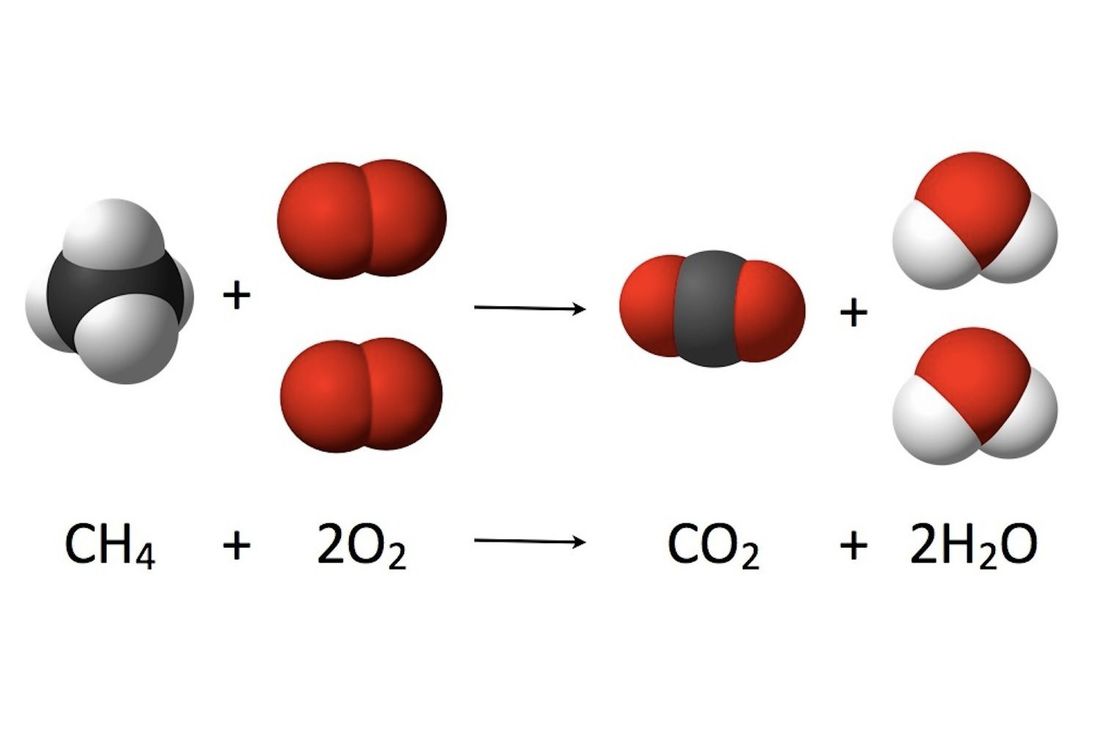
Let's take a common example: the combustion of methane, which is the main ingredient in natural gas. Methane has the chemical formula CH₄. It needs oxygen, which is O₂.
So, on the "ingredients" side, we have CH₄ + O₂. This is where the magic starts.
The Balancing Act
But here's where the "equation" part gets interesting. Chemists are all about keeping things balanced. They want to make sure that all the atoms that go into the reaction come out in the products, just in a different arrangement.
This means we often have to put numbers in front of the chemical formulas. These numbers are called coefficients. They tell us how many molecules of each substance are involved. It’s like saying, "We need two of these and one of those."
For our methane combustion, the balanced equation looks like this:
CH₄ + 2O₂ → CO₂ + 2H₂O
Isn't that neat? Let’s break it down! We start with one molecule of methane (CH₄) and two molecules of oxygen (2O₂).

And what do we get? One molecule of carbon dioxide (CO₂) and two molecules of water (2H₂O). If you count the atoms on each side, you’ll see they all match up! It's a perfect chemical accounting.
The arrow (→) is important too. It means "yields" or "produces." It’s the sign that the transformation has happened.
Why It's So Captivating
What makes this whole equation so captivating is its simplicity and power. It takes everyday elements and processes and describes them in a clear, concise way. It's a window into the hidden world of chemical transformations.
It’s also endlessly versatile. While we used methane as an example, the same principles apply to burning wood, gasoline, or even rocket fuel! The basic idea of fuel + oxygen → products + energy remains the same.
Plus, the visual aspect of combustion is undeniably engaging. Seeing flames leap and feeling the warmth is a direct connection to these invisible chemical reactions. The equation is the explanation behind the spectacle.

A Little Bit of Drama
There's a certain inherent drama in a combustion reaction. It's about change, transformation, and the release of energy. It’s the chemical equivalent of a fireworks display, but it happens every day in countless ways.
Think about a car engine. It’s a series of controlled combustion reactions happening at lightning speed, powering your journey. Or a gas stove, providing heat for your dinner. These are all thanks to this fundamental chemical process.
The beauty is that this seemingly simple equation unlocks understanding of so many phenomena around us. It's a key that opens doors to appreciating the world a little more deeply.
Sparking Curiosity
So, next time you see a flame, whether it's a candle or a bonfire, remember the equation behind it. Remember the fuel, the oxygen, and the incredible transformation that’s taking place. It’s a reminder that science is all around us, often in the most exciting and familiar forms.
This equation isn't just a string of letters and numbers; it's a story of energy, change, and the fundamental building blocks of our world. It’s an invitation to look closer and wonder about the fiery performances happening constantly.
Perhaps it will even inspire you to look up more about different types of combustion or the fascinating world of chemistry. Who knows what amazing reactions you might discover next!
