Which Equation Correctly Represents The Electron Affinity Of Calcium

Hey there, science curious folks! Ever wondered about the tiny, zippy things that make up everything around us? Yep, we’re talking about atoms, and specifically, the electrons whizzing around their centers. Today, we're going to dive into something called electron affinity, and figure out which equation is the real deal for our friend, Calcium.
Now, what in the world is electron affinity, you ask? Think of it like this: imagine an atom is a bit like a house. Electrons are like little guests who want to join the party. Electron affinity is basically the energy change that happens when a neutral atom decides to invite an extra electron into its "house." Does it get excited to welcome the new guest (releasing energy), or does it have to expend energy to make room (absorbing energy)? It's like figuring out if your house naturally has space and welcomes an extra roommate, or if you have to shuffle furniture and spend some cash to make it work.
So, why is this concept so cool? Well, it tells us a lot about how atoms behave, how they interact, and ultimately, how they form all the stuff we see and touch. It’s a fundamental piece of the puzzle in understanding chemistry.
Calcium: A Friendly Metal
Now, let's zoom in on our star for today: Calcium. You know, the stuff in milk that’s good for your bones? Calcium is a metal, and metals tend to be pretty generous with their electrons. They often like to lose them to become stable, like a kid finally getting rid of a toy they’re tired of playing with.
But what happens when Calcium is faced with the opportunity to gain an electron? Does it eagerly snatch it up, or is it a bit hesitant? This is where electron affinity comes in. We're trying to quantify that decision, that energy transaction.
The Grand Equation Hunt
When we talk about electron affinity, especially in the context of a chemical equation, we’re looking for a representation that accurately shows this energy change. It's like trying to find the perfect recipe for a cake – there might be a few variations, but only one is truly the most accurate and universally accepted for a specific outcome.
Generally, electron affinity is represented as the energy change when an electron is added to a gaseous atom. So, you’ll see a neutral atom in its gaseous state, with an electron floating nearby, and then what happens when they come together. It’s like a little atomic meet-cute!
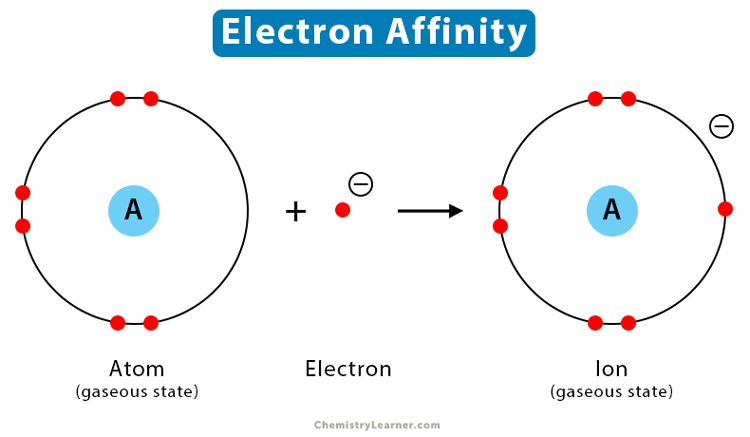
Let’s imagine a generic scenario. If an atom wants an electron, it will likely release energy when it gains one. This is because the electron finds a more stable, lower-energy state within the atom's electron cloud. Think of it like finding a really comfy spot on the couch – you just relax into it, and that relaxation is like releasing energy. This process is usually described as exothermic, meaning heat (or energy) is given off.
On the other hand, if an atom really, really doesn’t want an electron, it might actually require energy to be forced to accept one. This is like trying to squeeze a square peg into a round hole – it takes effort! This process is described as endothermic, meaning energy is absorbed.
So, What About Calcium?
Here’s where it gets interesting. Calcium, being a metal, is generally quite reluctant to gain electrons. It’s already got a couple of valence electrons it's happy to shed to achieve a stable electron configuration. So, when you try to force an extra electron onto a calcium atom, it’s not exactly a warm welcome.
Instead of happily accepting the electron and releasing energy, Calcium actually needs to absorb energy to accommodate this new, unwanted guest. It’s like trying to add another person to a small, cozy apartment when everyone’s already feeling a bit crowded. You might need to do some work, rearrange things, and maybe even pay a little extra to make it happen.

Therefore, the electron affinity of Calcium is typically considered to be a positive value. This positive sign is a crucial indicator. It tells us that energy must be added to the system for Calcium to gain an electron. It's not a spontaneous, energy-releasing event.
Let's Look at the "Equations"
When we write equations to represent electron affinity, we're essentially showing the atomic process and the associated energy change. For Calcium, the correct representation will reflect this energy absorption.
You might see something like this:
Ca (g) + e- → Ca- (g) ΔE = + value

Let's break that down.
- Ca (g): This represents a neutral calcium atom in its gaseous state. We specify gaseous because we're looking at the atom in isolation, free from the interactions it might have in a solid or liquid.
- + e-: This is the electron that's being offered.
- →: This arrow shows the transformation, the "after" picture.
- Ca- (g): This is the calcium atom after it has gained an electron, forming a negative ion (an anion).
- ΔE = + value: This is the critical part! ΔE stands for the change in energy. The "+" sign indicates that the process is endothermic, meaning energy is absorbed by the system. The "value" would be the specific amount of energy in units like kilojoules per mole (kJ/mol).
So, this equation tells us that when a gaseous calcium atom accepts an electron, it requires an input of energy. It's not a process that happens on its own with a puff of energy. It’s more like a gentle nudge that requires a bit of a push.
You might sometimes see electron affinity described with a negative value for exothermic processes. In those cases, the equation might look like:
Atom (g) + e- → Atom- (g) ΔE = - value (for exothermic)

But for Calcium, we're firmly in the positive ΔE territory. It’s like the difference between a hug that releases warmth and a handshake that requires a bit of effort to initiate.
Why Does This Matter?
Understanding this positive electron affinity for Calcium is super important in chemistry. It helps us predict how Calcium will react with other elements. For instance, elements with highly negative electron affinities (meaning they readily accept electrons) will tend to react vigorously with metals like Calcium, which are reluctant to gain them but happy to lose them. It’s a dance of electron giving and taking!
This fundamental concept plays a role in everything from designing new materials to understanding biological processes. It’s a small piece of information, but it unlocks a whole lot of understanding about the universe at its most basic level.
So, the next time you’re sipping on some milk, you can marvel at the fascinating electron affinity of Calcium – a metal that, while great for your bones, isn’t exactly jumping at the chance to welcome an extra electron without a little energy investment!
Keep those curious minds buzzing, and until next time, happy exploring!
