Which Element Is Chemically Similar To Lithium
Hey there! So, you're curious about lithium, huh? That little powerhouse element. We all hear about it in our phones and electric cars, but have you ever stopped to wonder, "What else is like lithium?" It's like asking, "Who's the celebrity twin of my favorite actor?" right? Totally relatable.
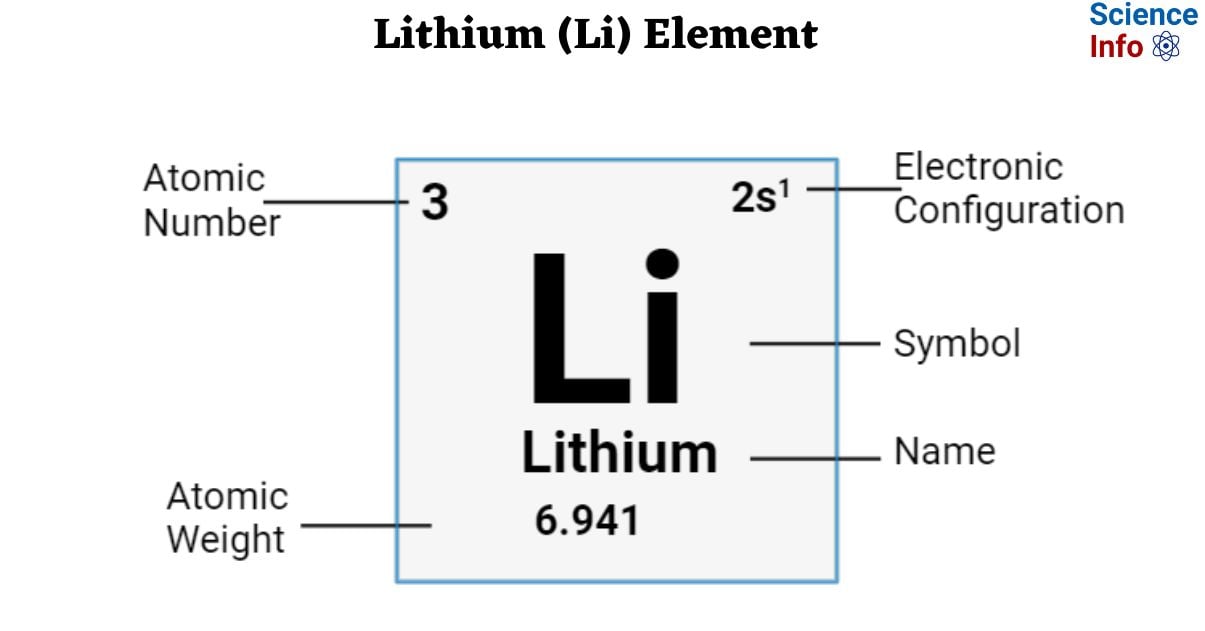
Well, let me tell you, when we talk about chemical similarities, we're not just pulling names out of a hat. It's all about where these elements hang out on that famous periodic table. You know, that giant chart with all the building blocks of the universe? It's basically the ultimate yearbook, grouping everyone by their personality, if you will. And lithium, bless its tiny heart, has some pretty cool buddies.
So, who’s the element that’s giving lithium a run for its money in the similarity department? Drumroll, please… It’s sodium! Yep, good old sodium. The stuff in your table salt. Kind of unexpected, maybe? I mean, one is all high-tech and the other is, well, salty. But trust me, they’re practically cousins from different planets.
Why sodium, though? It’s all about their position. See, lithium and sodium are both in the first column, the Alkali Metals. Think of it as the "Super Reactive Friends" club. These guys are like the life of the party, always wanting to jump into a chemical reaction. They’re not exactly wallflowers, are they?
What makes them so similar? It’s their outer electron. Each of these alkali metals has just one lonely electron chilling in its outermost shell. And that single electron? It's basically screaming, "Let me out! I want to bond with someone!" This makes them incredibly eager to give that electron away to another element. It’s like they’re constantly offering a helping hand, or in this case, a helping electron. Talk about generous!
This eagerness to shed that electron is what makes them so reactive. They love to team up with non-metals, especially those greedy halogens (think chlorine, fluorine – the ones that love to take electrons). It’s a classic give-and-take, a cosmic dance of electron exchange. They form ionic bonds like it's their job, which, technically, it is!
So, lithium has one electron to give. Sodium has one electron to give. See the pattern? It’s like they’re both wearing the same "I'm open for business" sign. This makes their chemical behavior remarkably alike. They’ll react with water with impressive enthusiasm, often with a little spark or even a flame. Don't try this at home, folks. Seriously. These guys are not to be messed with when they're in their pure form.
Think about it: both lithium and sodium readily lose that one outer electron to form a positive ion. Lithium becomes Li+, and sodium becomes Na+. It’s like they’ve both decided to become single and ready to mingle in the chemical world. This similarity in ion formation is HUGE in chemistry. It dictates how they interact with other elements and how they behave in compounds.
And it's not just about reacting with water or forming positive ions. Their general chemical properties are surprisingly aligned. They both tend to form compounds with similar structures. For instance, both lithium and sodium form oxides (when they react with oxygen) and chlorides (when they react with chlorine) that have very similar crystal structures. It’s like they have the same architectural taste in molecular buildings!
But are they exactly the same? Of course not! Life, and chemistry, would be boring if everything was identical. While sodium is chemically very similar to lithium, there are some key differences. And these differences, as you might expect, stem from lithium being the smallest of the alkali metals. It's the baby of the family, so to speak.
Because lithium is so small, its nucleus has a stronger grip on its electrons. This means that, while it still readily gives up its outer electron, it does so with a little more effort than its bigger sibling, sodium. This subtle difference in electron affinity can lead to some variations in their reactivity and the stability of their compounds.
For example, lithium hydroxide (LiOH) is actually less soluble in water than sodium hydroxide (NaOH). That’s a pretty big deal in the chemical world! It means sodium hydroxide is more likely to dissolve and spread out in water, becoming a more potent base. Lithium, being a bit more stubborn, likes to hang onto its solid form a bit better. Who knew such a tiny difference could matter so much?
Another fun fact: lithium is the only alkali metal that doesn't react vigorously with nitrogen gas (N2) at room temperature. Sodium? Oh, it'll happily team up with nitrogen. Lithium, however, is a bit of a diva and needs much higher temperatures to even consider that kind of partnership. It’s like sodium is a social butterfly and lithium is more of an introvert, preferring specific conditions for interaction.
And what about their metallic properties? Both are soft, silvery metals that can be easily cut with a knife. But again, that size thing. Lithium is denser than sodium. So, if you had a chunk of lithium and a chunk of sodium of the same size, the lithium would be heavier. It’s packed tighter, like a perfectly organized carry-on suitcase versus a casually stuffed duffel bag.
The reactivity with air is also slightly different. Both tarnish quickly when exposed to air, forming oxides. But lithium forms a nitride (Li3N) much more readily than sodium. Again, this nitrogen thing! It's like lithium has this special relationship with nitrogen that sodium doesn't quite share at the same ease.

So, we’ve established that sodium is lithium's chemical doppelganger, living in the same "Super Reactive Friends" group (Alkali Metals). They both have that one precious electron just begging to be shared, making them super eager to bond. They form similar types of compounds and behave in many analogous ways.
But, like any good friendship or family dynamic, there are nuances. Lithium’s smaller size gives it a bit more of a unique personality. It's the slightly less extreme version, the one who might think twice before jumping headfirst into every single situation. It's the friend who still loves a good party but might bring a book just in case.
Beyond sodium, are there any other elements that give lithium a nod in the similarity department? Well, if you stretch the definition a lot, you could maybe look at other elements in the same period (the horizontal rows) of the periodic table, but that’s a whole different ballgame. We're talking about very, very loose connections there.
When chemists talk about "chemically similar," they almost always mean elements in the same group (vertical column). And for lithium, that group is the Alkali Metals, with sodium leading the charge as its closest chemical relative. Think of it as siblings versus distant cousins. Sodium is definitely the sibling.

It’s fascinating, isn’t it? How just a few millimeters difference in atomic radius can lead to such distinct (though still fundamentally similar) chemical behaviors. It’s the subtle details that make the universe so interesting. It’s not just about the big, flashy reactions; it's about the tiny tugs and pulls of electrons.
So, next time you’re scrolling through your phone or thinking about an electric car, spare a thought for lithium and its salty friend, sodium. They're out there, quietly, or sometimes explosively, doing their chemical thing, proving that even the smallest differences can matter. And that, my friend, is the magic of chemistry.
It’s like they’re both saying, "We’re part of the same club, but I’ve got my own vibe." And that's perfectly okay. The periodic table is full of these stories. Groups of elements that share a core identity but still manage to carve out their own little niches. It’s a testament to the complexity and beauty of the elements that make up everything around us.
So, the short answer to your question, "Which element is chemically similar to lithium?" is unequivocally sodium. But the long answer, as we've seen, is a little more nuanced and a lot more fun to explore. It’s about understanding why they’re similar, where they diverge, and what that tells us about the fundamental nature of matter. Pretty cool, right?
And hey, if you ever find yourself needing to explain this to someone, just remember the "Super Reactive Friends" club. Everyone loves a good club analogy. It makes chemistry feel a lot less like a scary textbook and a lot more like a conversation. So, go forth and spread the lithium-sodium knowledge! You’re practically a chemistry guru now.
